Abstract
Background:
CYP2C8/9 polymorphisms may influence breast cancer-free survival after diagnosis due to their role in the metabolism of tamoxifen, paclitaxel, and other chemotherapy. cytochrome P450 (CYP)2C8/9 metabolise arachidonic acid to epoxyeicosatrienoic acids, which enhance migration and invasion in vitro and promote angiogenesis in vivo. We aimed to investigate the frequency of CYP2C8/9 polymorphisms in relation to breast tumour characteristics and disease-free survival.
Methods:
A prospective series of 652 breast cancer patients from southern Sweden was genotyped for CYP2C8*3, CYP2C8*4, CYP2C9*2, and CYP2C9*3. Blood samples and questionnaires were obtained pre- and postoperatively. Clinical information and tumour characteristics were obtained from patients' charts and pathology reports.
Results:
Frequencies of CYP2C8/9 polymorphisms were similar to healthy European populations. Significantly less node involvement (P=0.002) and fewer PR+ tumours (P=0.012) were associated with CYP2C8*4. Median follow-up was 25 months and 52 breast cancer-related events were reported. In a multivariate model, CYP2C8/9*3/*1*/*2/*1 was the only factor associated with increased risk for early events in 297 tamoxifen-treated, ER-positive patients, adjusted HR 2.54 (95%CI 1.11–5.79). The effect appeared to be driven by CYP2C8*3, adjusted HR 8.56 (95%CI 1.53–51.1).
Conclusion:
Polymorphic variants of CYP2C8/9 may influence breast tumour characteristics and disease-free survival in tamoxifen-treated patients.
Similar content being viewed by others
Main
In Sweden, approximately 7000 women are diagnosed with breast cancer annually and 1500 die of their disease. Up to 25% of breast cancer patients considered to be at low risk for recurrence, that is, stage-I or II without lymph node involvement, recur within 5 years (Malmström et al, 2001, 2003). Approximately 15% of all breast cancer patients in Sweden die from their disease within 5 years and 30% die within 10 years of diagnosis. Adjuvant therapies such as radiation, tamoxifen, aromatase inhibitors (AIs), and chemotherapy improve the prognosis, but also confer a risk of adverse side effects (Early breast cancer trialists' collaborative group, 2005; Forbes et al, 2008). Moreover, many patients receive adjuvant therapy without any impact on survival, as most are already cured by surgery alone or the adjuvant therapy chosen does not work as intended. Markers, which would help to better tailor adjuvant therapy to each patient, are urgently needed.
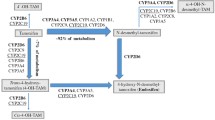
Several genetic polymorphisms in genes such as cytochrome-P450 (CYP)2C8 and CYP2C9, may influence survival after cancer diagnosis due to their role in the metabolism of various breast cancer drugs, including tamoxifen and chemotherapy (** et al, 2005). CYP2C8 and CYP2C9 are polymorphic enzymes. CYP2C8*3 and CYP2C9*2 are the major variant alleles in Caucasian populations (Yasar et al, 2002). Approximately 96% of subjects with the CYP2C8*3 allele also carried a CYP2C9*2 and 85% of subjects who had the CYP2C9*2 variant also carried a CYP2C8*3. CYP2C8*3 is defective in the metabolism of two important CYP2C8 substrates: the anticancer drug paclitaxel (Bahadur et al, 2002) and the physiologically important compound arachidonic acid (AA) (Dai et al, 2001). In addition, variants CYP2C8*4 and CYP2C9*2 and CYP2C9*3 also have a lower metabolic activity than the wild-type variants (Bahadur et al, 2002; Griskevicius et al, 2003; King et al, 2004; Sandberg et al, 2004).
Arachidonic acid is metabolised via three major pathways: the cyclooxygenase pathway, which produces prostaglandins; the lipoxygenase pathway, and finally the CYP epoxygenase pathway (Belton and Fitzgerald, 2003).
CYP2C8 and 2C9 are CYP epoxygenases, which metabolise AA to epoxyeicosatrienoic acids (EETs) (Zeldin et al, 1995; Michaelis et al, 2005), with the most abundant product being 14,15-EET, which promotes angiogenesis in vivo (Medhora et al, 2003). In vitro studies have shown that overexpression of CYP2C9 elicits angiogenesis via activation of the epidermal growth factor receptor (EGFR) (Michaelis et al, 2003). Yasar et al (1999) from a Swedish population of 430 unrelated healthy people.
This is the first study to report an association between decreased odds for lymph node involvement and CYP2C8*4 and an over twofold increased odds for lymph node involvement among CYP2C8/9 *3/*1/*2/*1 carriers with invasive tumour sizes over 20 mm. CYP2C8*3 and CYP2C8*4 have both been reported to have lower metabolic activity than the wild type (Bahadur et al, 2002), but the associations between these polymorphisms and axillary nodal involvement went in opposite directions in the current study. Since CYP2C8*3 was in strong LD with CYP2C9*2, it is likely that the association between CYP2C8*3 and nodal status is actually reflecting the association between the CYP2C9*2 polymorphism and nodal status.
We found that an increasing number of CYP2C8/9 *3/*1/*2/*1 haplotype alleles and especially CYP2C8*3 alleles was associated with increased hazard of early breast cancer-related events in tamoxifen-treated patients. Tamoxifen is a moderate CYP2C8 inhibitor (Walsky et al, 2005), a CYP2C9 substrate (** et al, 2005), and significantly inhibits CYP2C9 activity in breast cancer patients (Boruban et al, 2006). CYP2C9 is involved in tamoxifen activation, although neither CYP2C9*2 nor CYP2C9*3, which have lower activity than the wild type, were significantly associated with the levels of the potent tamoxifen metabolite endoxifen in one study (** et al, 2005). The effect of CYP2C8 polymorphisms on endoxifen levels has, to our knowledge, not been investigated. Since the LD between CYP2C8*3 and CYP2C9*2 was incomplete, we also examined the effect of each SNP separately in the model, and while CYP2C8*3 was associated with significant increased hazard of early breast cancer-related events, CYP2C9*2 was associated with non-significant decreased hazard. If our finding is replicated, the CYP2C8*3 may be used to identify patients who may recur early when treated with tamoxifen and who should be offered additional or different treatment.
Currently, no genotype data are used prior to selection of tamoxifen or aromatase inhibitors for breast cancer patients with ER-positive tumours warranting adjuvant endocrine treatment. CYP2D6 has been shown to significantly affect the levels of endoxifen and clinical outcome after tamoxifen treatment (** et al, 2005; Goetz et al, 2008). To our knowledge there is no association between CYP2D6 and CYP2C8/9, and these genes are located on different chromosomes. It is, therefore, unlikely that the increased risk for early recurrences observed in CYP2C8*3 carriers would be explained by CYP2D6 polymorphisms. Schroth et al. studied CYP2D6, CYP2C19, CYP3A5, CYP2B6, and CYP2C9 in tamoxifen-treated women. They reported that the CYP2C19*17 allele partly compensates for non-functioning CYP2D6 alleles and showed that carriers of one CYP2D6-null allele could be further stratified according to their CYP2C19*17 genotype with respect to tamoxifen response (Schroth et al, 2007). The genotypes of CYP3A5, CYP2B6, and CYP2C9 were not significantly associated with tamoxifen response, which is in line with our finding with respect to CYP2C9*2 and CYP2C9*3, but they did not examine CYP2C8.
The St Gallen guidelines recommend that extensive peritumoral vascular invasion be used as a prognostic factor (Goldhirsch et al, 2007). The degree of angiogenesis is not yet routinely evaluated in the clinical setting in Sweden, and we could, therefore, not evaluate whether there was any association between peritumoral vascular invasion and CYP2C8/9 haplotypes. It is plausible that we could have found an association, since CYP2C9-derived EETs stimulate angiogenesis (Michaelis et al, 2003). CYP2C9-derived EETs do so by a mechanism involving activation of the EGFR (Michaelis et al, 2003). One study reported high expression of EGFR to be inversely correlated with nodal metastases and shorter, distant disease-free survival in a group of breast cancer patients who had received 2 years of adjuvant tamoxifen treatment (Dihge et al, 2008). This is not fully compatible with our finding, as we found both a higher frequency of nodal involvement with the CYP2C8/9 *3/*1/*2/*1 genotype (in tumours larger than 20 mm) and a higher risk of any type of breast cancer recurrence, especially in women treated with tamoxifen. In small tumours, angiogenesis is also a predictor of nodal status (Arisio et al, 2000), and it is thus possible that women with the CYP2C8/9 *3/*1/*2/*1 genotype have tumours that are more likely to metastasise through increased angiogenesis.
The enzyme activity of CYP2C8 and CYP2C9 is not only determined by polymorphic variants, but also by use of CYP2C8- and CYP2C9-inhibiting compounds, including several drugs, as reviewed by Ingelman-Sundberg et al (2007). In the present study we enquired only about the use of all concomitant medications during the past week and lack information on long-term use of any of the medications. However, this information is not relevant in relation to tumour characteristics as the tumours were formed many years prior to diagnosis.
HER-2/neu status was not analysed routinely before November 2005, and we have therefore not been able to evaluate the correlation between HER-2 overamplification and CYP2C8/9 haplotypes. Histological grade was assessed according to the procedure of Elston and Ellis (1991). In the current study histological grade was not associated with early recurrences in either group (data not shown), but the median follow-up time is still short.
Our material consisted of a series of primary breast cancer patients, where the only exclusion criteria were any previous breast cancer diagnosis and other cancer diagnosed within the past 10 years. Approximately 60% of the patients operated on in Lund and 46% of the patients operated on in Helsingborg during the study's enrolment period were included. Our sample from Lund was similar to all patients from Lund with respect to age and ER and PR status. The patients from Helsingborg were somewhat older and had fewer PR-positive tumours as compared with the region as a whole, while the subset of patients who were included in this study was comparable to those included from Lund and the whole South Swedish region with respect to age and hormone-receptor status. There are several potential explanations for why receptor status may be different in Helsingborg patients. The patients in Helsingborg are somewhat older and different sets of antibodies are used at different Departments of pathology. ER and PR status was independently re-assessed by 22 pathologists from nine hospitals and the kappa values were 0.78 for ER and 0.72 for PR in 2003 (Chebil et al, 2003). Provided that these markers are used for selection of breast cancer treatment, quality assurance is ongoing. The frequency of HRT use may also differ between the two cities and HRT treatment is associated with a higher frequency of ER-positive tumours (Glass et al, 2007). As patients who were included did not differ with respect to ER, PR, or prior HRT use, we were unable to explain why patients from Helsingborg in general would have fewer PR-positive tumours.
In conclusion, we found that the frequencies of CYP2C8*3 and CYP2C8*4, and CYP2C9*2 and CYP2C9*3 were comparable to those of healthy European populations. Each copy of the CYP2C8/9 *1/*4/*1/*1 allele was associated with significantly lower risk for nodal involvement, while each copy of the CYP2C8/9 *3/*1/*2/*1 allele was associated with increased risk for nodal involvement in tumours larger than 20 mm. Moreover, the CYP2C8*3 allele was associated with early breast cancer-related events in women treated with tamoxifen. Since this is the first study reporting an association between CYP2C8 and tamoxifen response, and the median follow-up time is still short, the finding warrants confirmation. If confirmed, it is possible that CYP2C8*3 can be used as a genetic marker for prediction of treatment response to tamoxifen.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Arisio R, Sapino A, Cassoni P, Accinelli G, Cuccorese MC, Mano MP, Bussolati G (2000) What modifies the relation between tumour size and lymph node metastases in T1 breast carcinomas? J Clin Pathol 53: 846–850
Bahadur N, Leathart JB, Mutch E, Steimel-Crespi D, Dunn SA, Gilissen R, Houdt JV, Hendrickx J, Mannens G, Bohets H, Williams FM, Armstrong M, Crespi CL, Daly AK (2002) CYP2C8 polymorphisms in Caucasians and their relationship with paclitaxel 6alpha-hydroxylase activity in human liver microsomes. Biochem Pharmacol 64: 1579–1589
Belton O, Fitzgerald DJ (2003) Cyclooxygenase isoforms and atherosclerosis. Expert Rev Mol Med 5: 1–18
Boruban MC, Yasar U, Babaoglu MO, Sencan O, Bozkurt A (2006) Tamoxifen inhibits cytochrome P450 2C9 activity in breast cancer patients. J Chemother 18: 421–424
Chebil G, Bendahl PO, Ferno M (2003) Estrogen and progesterone receptor assay in paraffin-embedded breast cancer—reproducibility of assessment. Acta Oncol 42: 43–47
Dai D, Zeldin DC, Blaisdell JA, Chanas B, Coulter SJ, Ghanayem BI, Goldstein JA (2001) Polymorphisms in human CYP2C8 decrease metabolism of the anticancer drug paclitaxel and arachidonic acid. Pharmacogenetics 11: 597–607
Dihge L, Bendahl PO, Grabau D, Isola J, Lovgren K, Ryden L, Ferno M (2008) Epidermal growth factor receptor (EGFR) and the estrogen receptor modulator amplified in breast cancer (AIB1) for predicting clinical outcome after adjuvant tamoxifen in breast cancer. Breast Cancer Res Treat 109: 255–262
Dorado P, Cavaco I, Caceres MC, Piedade R, Ribeiro V, Llerena A (2008) Relationship between CYP2C8 genotypes and diclofenac 5-hydroxylation in healthy Spanish volunteers. Eur J Clin Pharmacol 64: 967–970
Early breast cancer trialists' collaborative group (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365: 1687–1717
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19: 403–410
Forbes JF, Cuzick J, Buzdar A, Howell A, Tobias JS, Baum M (2008) Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the ATAC trial. Lancet Oncol 9: 45–53
Glass AG, Lacey Jr JV, Carreon JD, Hoover RN (2007) Breast cancer incidence, 1980–2006: combined roles of menopausal hormone therapy, screening mammography, and estrogen receptor status. J Natl Cancer Inst 99: 1152–1161
Goetz MP, Kamal A, Ames MM (2008) Tamoxifen pharmacogenomics: the role of CYP2D6 as a predictor of drug response. Clin Pharmacol Ther 83: 160–166
Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thurlimann B, Senn HJ (2007) Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann Oncol 18: 1133–1144
Griskevicius L, Yasar U, Sandberg M, Hidestrand M, Eliasson E, Tybring G, Hassan M, Dahl ML (2003) Bioactivation of cyclophosphamide: the role of polymorphic CYP2C enzymes. Eur J Clin Pharmacol 59: 103–109
Ingelman-Sundberg M, Sim SC, Gomez A, Rodriguez-Antona C (2007) Influence of cytochrome P450 polymorphisms on drug therapies: pharmacogenetic, pharmacoepigenetic and clinical aspects. Pharmacol Ther 116: 496–526
Jiang JG, Ning YG, Chen C, Ma D, Liu ZJ, Yang S, Zhou J, **ao X, Zhang XA, Edin ML, Card JW, Wang J, Zeldin DC, Wang DW (2007) Cytochrome P450 epoxygenase promotes human cancer metastasis. Cancer Res 67: 6665–6674
** Y, Desta Z, Stearns V, Ward B, Ho H, Lee KH, Skaar T, Storniolo AM, Li L, Araba A, Blanchard R, Nguyen A, Ullmer L, Hayden J, Lemler S, Weinshilboum RM, Rae JM, Hayes DF, Flockhart DA (2005) CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst 97: 30–39
King BP, Khan TI, Aithal GP, Kamali F, Daly AK (2004) Upstream and coding region CYP2C9 polymorphisms: correlation with warfarin dose and metabolism. Pharmacogenetics 14: 813–822
Malmström P, Bendahl PO, Boiesen P, Brünner N, Idvall I, Fernö M, South Sweden Breast Cancer Group (2001) S-phase fraction and urokinase plasminogen activator are better markers for distant recurrences than Nottingham Prognostic Index and histologic grade in a prospective study of premenopausal lymph node-negative breast cancer. J Clin Oncol 19: 2010–2019
Malmström P, Holmberg L, Anderson H, Mattsson J, Jönsson PE, Tennvall-Nittby L, Balldin G, Loven L, Svensson JH, Ingvar C, Möller T, Holmberg E, Wallgren A, Swedish Breast Cancer Group (2003) Breast conservation surgery, with and without radiotherapy, in women with lymph node-negative breast cancer: a randomised clinical trial in a population with access to public mammography screening. Eur J Cancer 39: 1690–1697
Medhora M, Daniels J, Mundey K, Fisslthaler B, Busse R, Jacobs ER, Harder DR (2003) Epoxygenase-driven angiogenesis in human lung microvascular endothelial cells. Am J Physiol Heart Circ Physiol 284: H215–H224
Michaelis UR, Fisslthaler B, Barbosa-Sicard E, Falck JR, Fleming I, Busse R (2005) Cytochrome P450 epoxygenases 2C8 and 2C9 are implicated in hypoxia-induced endothelial cell migration and angiogenesis. J Cell Sci 118: 5489–5498
Michaelis UR, Fisslthaler B, Medhora M, Harder D, Fleming I, Busse R (2003) Cytochrome P450 2C9-derived epoxyeicosatrienoic acids induce angiogenesis via cross-talk with the epidermal growth factor receptor (EGFR). FASEB J 17: 770–772
NCBI SNP homepage (2008) NCBI SNP homepage Access date 2008-07-15 http://www.ncbi.nlm.nih.gov/SNP/snp_ref.cgi?rs=1058930
Sandberg M, Johansson I, Christensen M, Rane A, Eliasson E (2004) The impact of CYP2C9 genetics and oral contraceptives on cytochrome P450 2C9 phenotype. Drug Metab Dispos 32: 484–489
Schroth W, Antoniadou L, Fritz P, Schwab M, Muerdter T, Zanger UM, Simon W, Eichelbaum M, Brauch H (2007) Breast cancer treatment outcome with adjuvant tamoxifen relative to patient CYP2D6 and CYP2C19 genotypes. J Clin Oncol 25: 5187–5193
Walsky RL, Gaman EA, Obach RS (2005) Examination of 209 drugs for inhibition of cytochrome P450 2C8. J Clin Pharmacol 45: 68–78
Yasar U, Eliasson E, Dahl ML, Johansson I, Ingelman-Sundberg M, Sjoqvist F (1999) Validation of methods for CYP2C9 genoty**: frequencies of mutant alleles in a Swedish population. Biochem Biophys Res Commun 254: 628–631
Yasar U, Lundgren S, Eliasson E, Bennet A, Wiman B, de Faire U, Rane A (2002) Linkage between the CYP2C8 and CYP2C9 genetic polymorphisms. Biochem Biophys Res Commun 299: 25–28
Zeldin DC, DuBois RN, Falck JR, Capdevila JH (1995) Molecular cloning, expression and characterization of an endogenous human cytochrome P450 arachidonic acid epoxygenase isoform. Arch Biochem Biophys 322: 76–86
Acknowledgements
We thank the participants of this study. We also thank the following research nurses for data collection: Annette Möller, Karin Henriksson, Anna Weddig, Linda Ågren, Ulrika Midelund, Arnhild Nilsson, and Karina Sandström. We would like to acknowledge Maj-Britt Hedenblad and Sol-Britt Olsson for data entry and DNA-extraction. We thank Dr Eric T Dryver for proofreading the paper. This study was supported by grants from The Swedish Cancer Society, The Mrs Berta Kamprad Cancer Foundation, Lund University Hospital Fund, The Crafoord Foundation, The G Nilsson Foundation, The Swedish Research Council (K2001-27GX-14120-01A and K2008-68X-20802-01-3), The GA's Donation for Breast Cancer Research, the 1049 Fund at the Lund Oncology Clinic, the Region Skåne ALF, the Medical Faculty of Lund University, and an unrestricted grant by Novartis. Erika Bågeman's position was supported by the by grants from The Swedish Cancer Society, The G Nilsson Foundation, The Swedish Research Council (K2001-27GX-14120-01A), and the Medical Faculty of Lund University. Drs Christian Ingvar, Carsten Rose, and Per-Ebbe Jönsson were financially supported through their clinical positions. Dr Helena Jernström's position was supported by The Swedish Research Council (K2002-27GP-14104-02B), the Medical Faculty of Lund University, The Crafoord Foundation, and the Faculty of Health and Society of Malmö University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Jernström, H., Bågeman, E., Rose, C. et al. CYP2C8 and CYP2C9 polymorphisms in relation to tumour characteristics and early breast cancer related events among 652 breast cancer patients. Br J Cancer 101, 1817–1823 (2009). https://doi.org/10.1038/sj.bjc.6605428
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6605428
- Springer Nature Limited
Keywords
This article is cited by
-
Role of Genetic Polymorphisms in Drug-Metabolizing Enzyme-Mediated Toxicity and Pharmacokinetic Resistance to Anti-Cancer Agents: A Review on the Pharmacogenomics Aspect
Clinical Pharmacokinetics (2022)
-
Metabolism pathways of arachidonic acids: mechanisms and potential therapeutic targets
Signal Transduction and Targeted Therapy (2021)
-
Coffee consumption and breast cancer risk: a narrative review in the general population and in different subtypes of breast cancer
European Journal of Nutrition (2021)
-
Association of CYP gene polymorphisms with breast cancer risk and prognostic factors in the Jordanian population
BMC Medical Genetics (2019)
-
The impact of body size changes on recurrence risk depends on age and estrogen receptor status in primary breast cancer
Cancer Causes & Control (2019)