Abstract
This study aimed to evaluate the changes in intraocular pressure (IOP) before and after femtosecond laser capsulorhexis and lens fragmentation for cataract surgery. We measured the IOP before, immediately, 30 min, and 1 h after the laser procedure in 47 eyes of 47 patients who underwent the femtosecond laser procedure. The mean IOP was 17.51 ± 3.28 mmHg, 30.23 ± 6.70 mmHg, 17.96 ± 3.75 mmHg, and 21.77 ± 5.88 mmHg before, immediately after, 30 min after, and 1 h after the laser procedure, respectively. The mean IOP significantly increased immediately (adjusted P < 0.001) and 1 h (adjusted P = 0.001) after the laser procedure compared with the pre-laser IOP. The mean IOP at 30 min after the laser procedure was significantly lower than that immediately after the procedure (adjusted P < 0.001). However, the IOP 1 h after the laser procedure became higher than that 30 min after the laser procedure. Additionally, the IOP 1 h after the laser procedure was positively correlated with the baseline IOP and negatively correlated with the axial length. In conclusion, this study demonstrated that cataract surgery should be commenced within 30 min after the femtosecond laser procedure to ensure a safe cataract surgery that reduces the risk of increased intraocular pressure.
Similar content being viewed by others
Introduction
The femtosecond laser has recently been used in cataract surgery for corneal incision, continuous curvilinear capsulorhexis (CCC), and lens fragmentation, as well as in astigmatism correction1,2,3,4,5. The advantages of the femtosecond laser include accurate CCC with good intraocular lens (IOL) centration, minimal loss of endothelium, and a short phacoemulsification time6,7,8,9,10,11. Femtosecond laser-assisted cataract surgery (FLACS) had an advantage over conventional methods in minimizing endothelial cell loss after cataract surgery12.
Contrastingly, complications of using the femtosecond laser, such as suction loss, conjunctival injection, conjunctival hemorrhage, CCC tag, CCC tear, and miosis, have also been reported13. In addition, intraocular pressure (IOP) elevation occurs in porcine eyes when applying the suction ring and vacuum of the femtosecond laser14,15,16. Preoperative IOP elevation during cataract surgery could be a risk factor for suprachoroidal hemorrhage, a rare but significant complication of cataract surgery17. Therefore, IOP evaluation is crucial to preventing this serious complication during cataract surgery when using the femtosecond laser.
Several studies have reported changes in the IOP before and after performing femtosecond laser procedures in humans18,19,20,21,22. However, to the best of our knowledge, no published studies have evaluated the changes in IOP at certain intervals before and after femtosecond laser procedures. Because the femtosecond laser device is often located separately because of the lack of space in the operating room, monitoring the changes in IOP over time between the surgery and the laser procedure is necessary to ensure safety. Therefore, this study aimed to evaluate the changes in IOP before, immediately after, 30 min after, and 1 h after a femtosecond laser procedure.
Results
In this study, 47 eyes of 47 patients were included. Table 1 shows the demographic and clinical characteristics of the study participants. The mean nuclear sclerosis grade was 3.67 ± 0.94, and the preoperative corneal astigmatism, based on the autokeratometry, was 0.92 ± 0.85 D. Based on slit-lamp microscopy examinations, the nuclear sclerosis grades (graded according to the LOCS III) were grade 2 in 17.0% (8 eyes), grade 3 in 29.8% (14 eyes), grade 4 in 38.3% (18 eyes), and grade 5 in 14.9% (7 eyes) of the eyes. The laser energy used for CCC was 0.3 or 0.4 J, and that for lens fragmentation was approximately 5.0 to 11.5 J, according to the laser time. Arcuate incision (AI) was performed according to the patient’s corneal astigmatism, and the average laser energy for AI was 1.47 ± 1.01 J.
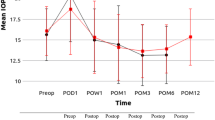
The mean IOPs before, immediately after, at 30 min after, and at 1 h after the laser procedure were 17.51 ± 3.28 mmHg, 30.23 ± 6.70 mmHg, 17.96 ± 3.75 mmHg, and 21.77 ± 5.88 mmHg, respectively (Table 2). Compared with the IOP before the laser procedure, the mean IOP immediately after (adjusted P < 0.001) and at 1 h after the laser procedure (adjusted P = 0.001) was significantly increased. In contrast, the mean IOP 30 min after the laser procedure (adjusted P < 0.001) was significantly decreased compared with that immediately after the laser procedure. Furthermore, there was a significant increase in the mean IOP at 1 h after the laser procedure (adjusted P < 0.001) compared with the IOP at 30 min after the laser procedure. The mean IOP before and 30 min after the laser procedure was not significantly different (Fig. 1). The mean IOPs per 10 min for up to 1 h after the femtosecond laser procedure for 16 eyes are shown in Table 3 and Fig. 2. The IOP remained similar from 10 to 30 min after the femtosecond laser procedure and then gradually increased until 1 h after the femtosecond laser procedure.
Regarding the regression analysis, data for 47 eyes and 47 patients’ personal data were analyzed. In the simple regression analysis, age (R2 = 0.008, P = 0.554), nuclear sclerosis grade (R2 = 0.000, P = 0.967), sex (R2 = 0.017, P = 0.385), anterior chamber depth (R2 = 0.032, P = 0.229), and laser energy (R2 = 0.007, P = 0.567) were not independent factors affecting the IOP at 1 h after the laser procedure. The factors that were significant in the simple regression were the axial length (R2 = 0.128, P = 0.014) and the baseline IOP (R2 = 0.148, P = 0.008). In the multiple regression analyses for these factors, the IOP at 1 h after the laser procedure was positively correlated with the baseline IOP (B = 0.637, P = 0.011) and negatively correlated with the axial length (B = -1.031, P = 0.019) (Table 4). In addition, the mean IOP at each time point between the low nuclear sclerosis grade (grades 2 and 3) and high nuclear sclerosis grade (grades 4 and 5) groups (Table 5) was not significantly different. Moreover, there was no significant difference in the mean IOP at each time point between the low total laser energy (mean energy less than 8.45 J) and high total laser energy (mean energy greater than 8.45 J) groups (Table 6).
Discussion
In the present study, we evaluated the changes in IOP before and after a femtosecond laser procedure and thus demonstrated that the IOP significantly increased immediately after the femtosecond laser procedure, normalized at 30 min after the laser procedure, but increased again at 1 h after the femtosecond laser procedure. FLACS has been reported to make cataract surgery easier and IOL centration more precise by ensuring a precise CCC. Therefore, a high quality of vision can be expected after FLACS24. In addition, FLACS reduces the phacoemulsification power and effective phacoemulsification time by preoperative lens fragmentation, accompanied by minimal loss of endothelial cells7,8,9,10.
The interface of the femtosecond laser and the cornea of the patient should be strongly attached by a vacuum before applying the femtosecond laser. Moreover, CCC can lead to the release of fragmented lens materials into the anterior chamber after a femtosecond laser procedure. Few studies have investigated the effects of these situations on ocular parameters such as the IOP. In experimental researches, IOP was increased in porcine eyes during the suction ring application of the femtosecond laser device14,15,16. In addition, during the vacuum application, the anterior and posterior structures of the eye could be changed25. Consequently, binocular vitreous detachment, retinal hemorrhage, and changes in ocular blood supply have been reported26,27,28. Although changes in the IOP before and after the femtosecond laser procedure have been reported, no published report has evaluated the changes in the IOP at certain time intervals before and after the laser procedure in humans18,19,20,21,22. Therefore, we sought to evaluate the IOP before and after a laser procedure at 30-min intervals for up to 1 h to ensure the safety of the femtosecond laser procedure.
Overall, in the present study, IOP significantly changed with time; the IOP immediately after the laser procedure was significantly higher than that before the laser procedure, and the IOP subsequently decreased significantly. However, it increased again over time. One study reported that increased IOP normalized immediately after the removal of the interface of the femtosecond laser22. However, in this study, the IOP remained higher than that before the laser procedure, even after removing the interface of the femtosecond laser. This result is similar to those of previous studies, which reported that the IOPs immediately after laser procedures were significantly higher than those before laser procedures, even after suction ring removal18,19,20,21. We hypothesized that this difference in changes in the IOP may be due to differences in femtosecond laser systems. Baig et al. used the Victus platform (Bausch & Lomb, Germany), which uses both the fluid-filled interface and the curved contact lens docking system during FLACS in two steps22. However, in all the others, as well as in our study, we used the Catalys Precision system with only a liquid optics interface18,19,20. De Giacinato et al. compared IOP changes between the Catalys Precision system and LenSx Laser (Alcon Laboratories, Inc., Fort Worth, TX, USA) with a curved interface and soft contact lens. The results showed a marked increase in IOP with the Catalys system than with the LenSx Laser21. The pressure of the suction ring and the vacuum force of the liquid optics interface probably contributed to the significant IOP increase immediately after the laser procedure.
In our study, the IOP decreased to a value similar to the pre-laser IOP 30 min after the laser procedure. This may have been caused by an aqueous humor turnover rate of approximately 1.0–1.5% per minute29. The number of tiny lens particles or macrophages released into the anterior chamber and inflammatory cytokines may have been diluted by the replacement of the intraocular volume29. The mean IOP at 1 h after the laser procedure was not as high as that immediately after the laser procedure, but the IOP increased significantly compared to that before and that at 30 min after the laser procedure. We suggest that fragmented lens particles may have blocked the trabecular meshwork, and CO2 bubbles produced after the femtosecond laser procedure may have increased the anterior chamber volume, both leading to the increased IOP. Based on the pathophysiology of phacolytic glaucoma, tiny lens particles or macrophages, even though invisible, could temporarily enter the anterior chamber, leading to an increased IOP30,31. In addition, inflammatory cytokines that were upregulated by the laser procedure could induce acute inflammation of the trabecular meshwork, leading to its blockage32,33.
We also hypothesized that increased IOP at 1 h after the femtosecond laser procedure may be affected by age, nuclear sclerosis grade, axial length, the IOP before the laser procedure, and the total laser energy. Among these, the IOP before the laser procedure was positively correlated with the IOP at 1 h after the laser procedure, and axial length was negatively correlated with the IOP at 1 h after the laser procedure. Previous studies also reported that an increase in the IOP during a laser procedure in porcine eyes was more correlated with the baseline IOP than the level of applied vacuum16,34. Additionally, we found a negative correlation between axial length and the IOP 1 h after the femtosecond laser procedure. This may be because a patient with a long axial length has a large eyeball volume, and the large volume plays a role in buffering the force exerted by suction during the laser procedure. Our results suggest that surgeons should look out for increases in the IOP of patients with a high preoperative IOP and a short axial length during laser procedures. Further evaluation with more patients is required to clarify this.
We measured the IOP every 10 min until 1 h after the laser procedure in our study. The IOP was usually lowered to the pre-laser level 10 min after the procedure and maintained at the same level until 30 min after the laser procedure. However, considering that the reduced IOP increased again 1 h after the laser procedure, we recommend that cataract surgery be performed within 30 min after a femtosecond laser procedure, during the time of stable IOP. Although the average IOP at 1 h after the femtosecond laser procedure was 21.77 mmHg, which is not high enough to cause optic nerve damage, the IOP ranged from 11 to 44 mmHg. Moreover, there were 12 eyes (26%) where the IOP was 25 mmHg or higher. Furthermore, even transient increases in the IOP can cause optic nerve damages and serious complications17,35,36. Therefore, we suggest focusing on those eyes with a tendency to develop high IOP rather than on their average IOP value. Some studies recommend using topical NSAIDs to reduce the inflammatory cytokine reflex, including prostaglandins, which can induce miosis33,35,37. Further research that investigates the ideal timing of performing cataract surgery after a femtosecond laser procedure is required to ensure patient safety and procedural efficiency.
We used Tono-Pen AVIA (Reichert Inc., Buffalo, NY, USA) to measure the IOP in the supine position, as it was necessary to measure the IOP quickly while maintaining the supine position. Because an intrastromal arcuate keratotomy was performed, there was no change in the corneal surface. Furthermore, it can be assumed that corneal edema resulting from the femtosecond laser procedure may cause a slight error in the IOP measurement. However, in a similar situation, a previous study showed that the IOP measurement obtained with the Tono-Pen was more accurate than that obtained with the Goldmann applanation tonometry owing to its small contact area with the corneal surface38. Therefore, we believe the measurement to be comparatively valid. However, because this is a hand-held device, the effect of instability should also be considered.
This study is limited by its relatively small number of patients and its retrospective design. IOP can be affected by the patient's anxiety about surgery and emotional stress, which were not included in the analysis in this study. In addition, our results are only applicable to situations where the same device, the Catalys system, is used; the study of IOP changes over time in cases where other femtosecond laser devices are used is required. Further prospective studies with a larger sample size and serial time interval IOP measurements in various femtosecond laser systems are required.
In summary, this is the first study that has serially evaluated IOP changes after a femtosecond laser procedure for cataract surgery. The mean IOP significantly increased immediately after the femtosecond laser procedure and then decreased significantly after 30 min. The IOP increased again 1 h after the laser procedure. The IOP 30 min after the laser procedure was not significantly different from that before the laser procedure. Therefore, we recommend that cataract surgery be performed within 30 min after a femtosecond laser procedure to ensure a safe and successful cataract surgery.
Methods
We conducted this observational case series with the approval of the Institutional Review Board of the Asan Medical Center and the University of Ulsan College of Medicine, Seoul, South Korea (2019–0774). Informed consent was obtained from all patients, and the study adhered to the tenets of the Declaration of Helsinki and followed good clinical practice guidelines. The study was registered with WHO/ICTRP, registration number: KCT0004710.
This study included patients who underwent femtosecond laser-assisted cataract surgery (FLACS) performed by the same surgeon at the Asan Medical Center. Patients who met the following criteria were included: (1) aged > 18 years; (2) have pre-existing corneal astigmatism of less than + 2.50 D; and (3) have a visual acuity of greater than 0.1 logMAR as measured using a potential acuity meter. Patients were excluded from the analyses if they had (1) optical opacities or pathology detected on slit-lamp examination; (2) previous corneal surgeries; (3) ocular trauma; (4) intraocular surgery; (5) severe dry eyes; (6) corneal disease; (7) glaucoma and ocular hypertension; (8) ocular infection; or (9) collagen vascular disease or other autoimmune diseases.
Preoperative assessments included autokeratometry (Canon R-50, Canon USA Inc., Huntington, NY, USA), slit-lamp examinations (Haag-Streit, Gartenstadtstrasse, Köniz, Switzerland), and corneal topography (Orbscan, Bausch & Lomb, Rochester, NY, USA). The nuclear sclerosis grade was measured based on the Lens Opacities Classification System (LOCS) III during slit-lamp examinations23. The anterior chamber depth was measured using anterior segment optical coherence tomography (Visante OCT, Carl Zeiss Meditec, Germany). Anterior chamber depth was defined as the vertical distance from the internal border of the central corneal endothelium to the line connecting both iris recesses. The IOP was measured using Tono-Pen AVIA (Reichert Inc., Buffalo, NY, USA) in the supine position before, immediately after, 30 min after, and 1 h after the femtosecond laser procedure. Each measurement was repeated three times, and the average was taken.
FLACS was performed using the Catalys Precision Laser System (Johnson & Johnson, Milpitas, CA, USA) for CCC, cataract fragmentation, and intrastromal arcuate keratotomy. The same method and parameters were used for CCC and lens fragmentation despite differences in the nuclear sclerosis grade of each patient. An AI was performed to correct astigmatism for all patients based on the degree of corneal astigmatism of each patient. For CCC, the “Scanned Capsule” mode was used, the diameter was 4.9 mm, the horizontal spot spacing was 5 µm, and the vertical spot spacing was 10 µm. Regarding lens fragmentation, the “Quadrants Softened” mode was used, the segmentation repetitions were 5, the horizontal spot spacing was 10 µm, and the vertical spot spacing was 40 µm. Regarding the AI, the “intrastromal” mode was used, the horizontal spot spacing was 5 µm, and the vertical spot spacing was 10 µm. In addition, 0.1 J of energy was used for each 5 degrees of the arc length.
Cataract surgery commenced 1 h after the femtosecond laser procedure. The Whitestar Signature Pro System (Johnson & Johnson, Milpitas, CA, USA) was used for the cataract surgery. The cornea was manually incised with an incision knife. After the insertion of the ocular viscoelastic devices (OVD), hydrodissection was performed through the CCC margin that was already made by the femtosecond laser. Fragmented lens materials were removed using a phacoemulsifier, after which the cortex was removed, the IOL was implanted, and the OVD was removed. The surgery ended after corneal hydration was performed. A single course of a ketorolac 0.45%, proparacaine 0.5%, and mixture of phenylephrine 0.5% and tropicamide 0.5% were administered 30 min before the femtosecond laser procedure. Topical antibiotics, steroids, and non-steroidal anti-inflammatory drugs were used until 1 month postoperatively.
Statistical analysis
A Shapiro–Wilk test was used to assess the distribution of numerical data. The Friedman test with post-hoc Bonferroni correction was used to investigate changes in IOP over time within a group of patients. The power analysis of the Friedman test showed that the statistical power of this study with 47 subjects was 99% and that sufficient participants were included. The Mann–Whitney test was used to compare the IOP between two different nuclear sclerosis grade groups and between two different total laser energy groups. A simple regression analysis was conducted to evaluate the correlation of age, nuclear sclerosis grade, sex, anterior chamber depth, laser energy, axial length, baseline IOP with IOP at 1 h after the femtosecond laser procedure. Multiple regression analyses were performed for factors with a P value of less than 0.05 in the simple regression analysis. There were no missing values among the variables included in the analysis. All statistical analyses were performed using SPSS 25.0 (IBM Corp., Armonk, NY, USA). A P value < 0.05 was considered statistically significant.
Ethics declarations
This study was approved by the Institutional Review Board of the Asan Medical Center and the University of Ulsan College of Medicine, Seoul, South Korea (2019–0774). Informed consent was obtained from all patients, and the study adhered to the tenets of the Declaration of Helsinki and followed good clinical practice guidelines.
Data availability
The data used in this study are available from the corresponding author upon request.
References
Nagy, Z., Takacs, A., Filkorn, T. & Sarayba, M. Initial clinical evaluation of an intraocular femtosecond laser in cataract surgery. J. Refract. Surg. 25(12), 1053–1060. https://doi.org/10.3928/1081597X-20091117-04 (2009).
Masket, S., Sarayba, M., Ignacio, T. & Fram, N. Femtosecond laser-assisted cataract incisions: Architectural stability and reproducibility. J. Cataract Refract. Surg. 36(6), 1048–1049. https://doi.org/10.1016/j.jcrs.2010.03.027 (2010).
Friedman, N. J. et al. Femtosecond laser capsulotomy. J. Cataract Refract. Surg. 37(7), 1189–1198. https://doi.org/10.1016/j.jcrs.2011.04.022 (2011).
Chang, J. S. et al. Initial Evaluation of a femtosecond laser system in cataract surgery. J. Cataract Refract. Surg. 40(1), 29–36. https://doi.org/10.1016/j.jcrs.2013.08.045 (2014).
Baharozian, C. J., Song, C., Hatch, K. M. & Talamo, J. H. A novel nomogram for the treatment of astigmatism with femtosecond-laser arcuate incisions at the time of cataract surgery. Clin. Ophthalmol. 11, 1841–1848. https://doi.org/10.2147/OPTH.S141255 (2017).
Kranitz, K. et al. Femtosecond laser capsulotomy and manual continuous curvilinear capsulorrhexis parameters and their effects on intraocular lens centration. J. Refract. Surg. 27(8), 558–563. https://doi.org/10.3928/1081597X-20110623-03 (2011).
Conrad-Hengerer, I., Al Juburi, M., Schultz, T., Hengerer, F. H. & Dick, H. B. Corneal endothelial cell loss and corneal thickness in conventional compared with femtosecond laser-assisted cataract surgery: Three-month follow-up. J. Cataract Refract. Surg. 39(9), 1307–1313. https://doi.org/10.1016/j.jcrs.2013.05.033 (2013).
Conrad-Hengerer, I., Hengerer, F. H., Schultz, T. & Dick, H. B. Effect of femtosecond laser fragmentation of the nucleus with different softening grid sizes on effective phaco time in cataract surgery. J. Cataract Refract. Surg. 38(11), 1888–1894. https://doi.org/10.1016/j.jcrs.2012.07.023 (2012).
Conrad-Hengerer, I., Hengerer, F. H., Schultz, T. & Dick, H. B. Effect of femtosecond laser fragmentation on effective phacoemulsification time in cataract surgery. J. Refract Surg. 28(12), 879–883. https://doi.org/10.3928/1081597X-20121116-02 (2012).
Abell, R. G., Kerr, N. M. & Vote, B. J. Toward zero effective phacoemulsification time using femtosecond laser pretreatment. Ophthalmology 120(5), 942–948. https://doi.org/10.1016/j.ophtha.2012.11.045 (2013).
Pantanelli, S. M. et al. Anterior capsulotomy outcomes: A comparison between two femtosecond laser cataract surgery platforms. J. Refract Surg. 31(12), 821–825. https://doi.org/10.3928/1081597X-20151118-01 (2015).
Puzo, P. et al. Live surgery outcomes in cataract surgery. Eur. J. Ophthalmol. 32(6), 3444–3450. https://doi.org/10.1177/11206721221089172 (2022).
Nagy, Z. Z. et al. Complications of femtosecond laser-assisted cataract surgery. J. Cataract Refract. Surg. 40(1), 20–28. https://doi.org/10.1016/j.jcrs.2013.08.046 (2014).
Vetter, J. M. et al. Intraocular pressure during corneal flap preparation: Comparison among four femtosecond lasers in porcine eyes. J. Refract. Surg. 27(6), 427–433. https://doi.org/10.3928/1081597X-20101210-01 (2011).
Sperl, P. et al. Intraocular pressure course during the femtosecond laser-assisted cataract surgery in porcine cadaver eyes. Investig. Ophthalmol. Vis. Sci. 58(14), 6457–6461. https://doi.org/10.1167/iovs.17-21948 (2017).
Mariacher, S. et al. Impact of baseline iop, vacuum, and different docking mechanisms, and their interaction on iop rise in femtosecond laser-assisted refractive and cataract surgery. J. Cataract Refract. Surg. 45(12), 1818–1825. https://doi.org/10.1016/j.jcrs.2019.07.020 (2019).
Obuchowska, I. & Mariak, Z. Risk factors of massive suprachoroidal hemorrhage during extracapsular cataract extraction surgery. Eur. J. Ophthalmol. 15(6), 712–717. https://doi.org/10.1177/112067210501500609 (2005).
Darian-Smith, E. et al. Intraocular pressure during femtosecond laser pretreatment: Comparison of glaucomatous eyes and nonglaucomatous eyes. J. Cataract Refract. Surg. 41(2), 272–277. https://doi.org/10.1016/j.jcrs.2014.10.026 (2015).
Kerr, N. M., Abell, R. G., Vote, B. J. & Toh, T. Intraocular pressure during femtosecond laser pretreatment of cataract. J. Cataract Refract. Surg. 39(3), 339–342. https://doi.org/10.1016/j.jcrs.2012.12.008 (2013).
Schultz, T., Conrad-Hengerer, I., Hengerer, F. H. & Dick, H. B. Intraocular pressure variation during femtosecond laser-assisted cataract surgery using a fluid-filled interface. J. Cataract Refract. Surg. 39(1), 22–27. https://doi.org/10.1016/j.jcrs.2012.10.038 (2013).
De Giacinto, C. et al. Intraocular pressure changes during femtosecond laser-assisted cataract surgery: A comparison between two different patient interfaces. J. Ophthalmol. 2019, 5986895. https://doi.org/10.1155/2019/5986895 (2019).
Baig, N. B. et al. Intraocular pressure profiles during femtosecond laser-assisted cataract surgery. J. Cataract Refract. Surg. 40(11), 1784–1789. https://doi.org/10.1016/j.jcrs.2014.04.026 (2014).
Karbassi, M., Khu, P. M., Singer, D. M. & Chylack, L. T. Jr. evaluation of lens opacities classification system iii applied at the slitlamp. Optom. Vis. Sci. 70(11), 923–928. https://doi.org/10.1097/00006324-199311000-00009 (1993).
Mihaltz, K. et al. Internal aberrations and optical quality after femtosecond laser anterior capsulotomy in cataract surgery. J. Refract. Surg. 27(10), 711–716. https://doi.org/10.3928/1081597X-20110913-01 (2011).
Davis, R. M. & Evangelista, J. A. Ocular structure changes during vacuum by the hansatome microkeratome suction ring. J. Refract. Surg. 23(6), 563–566. https://doi.org/10.3928/1081-597X-20070601-05 (2007).
Smith, R. J. et al. Complete bilateral vitreous detachment after lasik retreatment. J. Cataract Refract. Surg. 30(6), 1382–1384. https://doi.org/10.1016/j.jcrs.2003.10.021 (2004).
Moshfeghi, A. A., Harrison, S. A., Reinstein, D. Z. & Ferrone, P. J. Valsalva-like retinopathy following hyperopic laser in situ keratomileusis. Ophthalmic Surg. Lasers Imaging 37(6), 486–488. https://doi.org/10.3928/15428877-20061101-08 (2006).
Conway, M. L., Wevill, M., Benavente-Perez, A. & Hosking, S. L. Ocular blood-flow hemodynamics before and after application of a laser in situ keratomileusis ring. J. Cataract. Refract. Surg. 36(2), 268–272. https://doi.org/10.1016/j.jcrs.2009.09.013 (2010).
Goel, M., Picciani, R. G. & Lee, R. K. Bhattacharya SK. aqueous humor dynamics: A review. Open. Ophthalmol. J. 4, 52–59. https://doi.org/10.2174/1874364101004010052 (2010).
Yoo, W. S. et al. A case of phacolytic glaucoma with anterior lens capsule disruption identified by scanning electron microscopy. BMC Ophthalmol. 14, 133. https://doi.org/10.1186/1471-2415-14-133 (2014).
Mavrakanas, N., Axmann, S., Issum, C. V., Schutz, J. S. & Shaarawy, T. Phacolytic glaucoma: Are there 2 forms?. J. Glaucoma. 21(4), 248–249. https://doi.org/10.1097/IJG.0b013e31820d7d2e (2012).
Dick, H. B., Gerste, R. D. & Schultz, T. Laser cataract surgery: Curse of the small pupil. J. Refract. Surg. 29(10), 662. https://doi.org/10.3928/1081597X-20130920-01 (2013).
Schultz, T., Joachim, S. C., Szuler, M., Stellbogen, M. & Dick, H. B. Nsaid pretreatment inhibits prostaglandin release in femtosecond laser-assisted cataract surgery. J. Refract. Surg. 31(12), 791–794. https://doi.org/10.3928/1081597X-20151111-01 (2015).
Mariacher, S., Laubichler, P., Wendelstein, J., Mariacher, M. & Bolz, M. Preoperative intraocular pressure as a strong predictive factor for intraocular pressure rise during vacuum application in femtosecond laser-assisted cataract surgery. Acta Ophthalmol. 97(8), e1123–e1129. https://doi.org/10.1111/aos.14149 (2019).
Levy, N. S. & Crapps, E. E. Displacement of optic nerve head in response to short-term intraocular pressure elevation in human eyes. Arch. Ophthalmol. 102(5), 782–786. https://doi.org/10.1001/archopht.1984.01040030630037 (1984).
Jiang, R. et al. Optic nerve head changes after short-term intraocular pressure elevation in acute primary angle-closure suspects. Ophthalmology 122(4), 730–737. https://doi.org/10.1016/j.ophtha.2014.11.008 (2015).
Diakonis, V. F. et al. Effects of short-term preoperative topical ketorolac on pupil diameter in eyes undergoing femtosecond laser-assisted capsulotomy. J. Refract. Surg. 33(4), 230–234. https://doi.org/10.3928/1081597X-20170111-02 (2017).
Neuburger, M., Maier, P., Bohringer, D., Reinhard, T. & J, J. F. The impact of corneal edema on intraocular pressure measurements using goldmann applanation tonometry, tono-pen Xl, icare, and Ora: An in vitro model. J. Glaucoma. 22(7), 584–590. https://doi.org/10.1097/IJG.0b013e31824cef11 (2013).
Acknowledgements
We would like to thank Editage (www.editage.co.kr) for the English language editing.
Funding
This research was supported by the Research and Business Development Program through the Korea Institute for the Advancement of Technology (KIAT) funded by the Ministry of Trade, Industry, and Energy (MOTIE) (grant number: P0014063) and by a grant from the Asan Institute for Life Sciences, Asan Medical Center, Seoul, Korea (2021IP0061-3).
Author information
Authors and Affiliations
Contributions
H.S.C., H.L., and S.Y.P. contributed equally to this study and should be regarded as co-first authors. All authors contributed to the concept, design, supervision, and final approval of this study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chung, H.S., Lee, H., Park, S.y. et al. Intraocular pressure changes before and after a femtosecond laser procedure for cataract surgery. Sci Rep 14, 9020 (2024). https://doi.org/10.1038/s41598-024-55961-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-55961-2
- Springer Nature Limited