Abstract
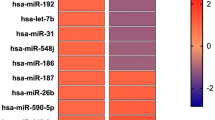
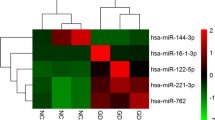
The aim of this study is to characterize the microRNA (miRNA) expression signatures in patients with thyroid eye disease (TED) and identify miRNA biomarkers of disease activity. Total RNA was isolated from the sera of patients with TED (n = 10) and healthy controls (HCs, n = 5) using the miRNeasy Serum/Plasma Kit. The NanoString assay was used for the comprehensive analysis of 798 miRNA expression profiles. Analysis of specific miRNA signatures, mRNA target pathway analysis, and network analysis were performed. Patients with TED were divided into two groups according to disease activity: active and inactive TED groups. Differentially expressed circulating miRNAs were identified and tested using quantitative reverse transcription-polymerase chain reaction (qRT-PCR) tests in the validation cohort. Among the 798 miRNAs analyzed, 173 differentially downregulated miRNAs were identified in TED patients compared to those in the HCs. Ten circulating miRNAs were differentially expressed between the active and inactive TED groups and regarded as candidate biomarkers for TED activity (one upregulated miRNA: miR-29c-3p; nine downregulated miRNAs: miR-4286, miR-941, miR-571, miR-129-2-3p, miR-484, miR-192-5p, miR-502-3p, miR-597-5p, and miR-296-3p). In the validation cohort, miR-484 and miR-192-5p showed significantly lower expression in the active TED group than in the inactive TED group. In conclusion, the expression levels of miR-484 and miR-192-5p differed significantly between the active and inactive TED groups, suggesting that these miRNAs could serve as circulating biomarkers of TED activity, however, these findings need to be validated in further studies.
Similar content being viewed by others
Introduction
Thyroid eye disease (TED) is the most common orbital inflammatory disease, and extrathyroidal manifestation of Grave’s disease (GD)1. Most patients with TED show distinct clinical features, including eyelid retraction, proptosis, extraocular muscle movement limitation, and inflammatory signs of eyelid and conjunctiva, which enable easy diagnosis. Although enormous progress has been made in elucidating the pathogenesis of TED, the exact mechanism of TED remains elusive, and therapeutic challenges and dilemmas remain.
Assessment of disease activity is crucial for designing treatment plans for patients with TED. Immunosuppressive agents, including corticosteroids, radiation treatment, and insulin-like growth factor-1 receptor (IGF-1R) antibodies are effective only in patients with active TED2,3,4. Surgical rehabilitation is the conventional treatment modality for inactive TED. The clinical activity score (CAS) is the most commonly used to measure the activity of TED in a clinical setting5. However, CAS contains subjective items such as spontaneous retrobulbar pain and pain on attempted eye movement. Furthermore, fine, quantitative analysis is not possible because each item of the CAS is binary6. These drawbacks warrant the development of novel biomarkers for measuring TED activity.
MicroRNAs (miRNAs) are a class of noncoding RNAs that post-transcriptionally regulate gene expression and are involved in a wide range of physiological and pathological processes36 investigated the association between the changes in levels of miR-146a and IL-7 with the clinical activity of TED using qRT-PCR and ELISA and reported lower miR-146a levels and higher IL-7 levels in active TED than in inactive TED. Shen et al.33 previously evaluated the level of circulating miRNAs according to the steroid responsiveness in TED patients. They used miScript PCR arrays for screening and qPCR for validation, like in case of the present study. However, they only included patients with active TED and compared the level of 84 miRNAs between steroid responders and non-responders. The strength of this study is that we performed the analysis of the expression of a wide panel of miRNAs. Here, we explored the levels of 798 miRNAs and used a very sensitive method to detect miRNAs in serum.
This study has some limitations. First, the sample size was limited; thus, the results from this study should ideally be validated in a much larger study population. Furthermore, the exact function of miR-484 and miR-192-5p in the pathogenesis of TED remains unclear, and in vitro or in vivo experimental studies regarding these miRNAs are warranted. Second, heterogeneous demographic, endocrinologic, and ophthalmologic factors can affect the expression of circulating miRNAs. Therefore, we plan to conduct further studies on the comparative analysis of miRNA expression in sequential serum samples collected from the same patient. Third, we performed only traditional biological experiments involving the screening of a broad panel of miRNAs, and validation of candidate miRNAs. Recently, there is a trend of develo** computational models to identify the associations between miRNAs and various human diseases37,38,39. These computational models can effectively integrate heterogeneous biological data and predict potential associations between miRNAs and human diseases. Future studies are expected to develop a computational model that can predict miRNAs related to TED. Forth, we identified 10 miRNAs as potential biomarkers of TED activity using the NanoString nCounter system, but we successfully amplified only 6 of 10 miRNAs using qRT-PCR. We speculated that miRNAs that exist in scarce amounts in the serum might fail to be amplified by qRT-PCR, although they were detected using an amplification-free technique.
In conclusion, we investigated the expression of a wide panel of 798 miRNAs in patients with TED and identified 10 miRNAs which were differentially expressed between active and inactive TED. Validation studies revealed that the serum levels of miR-484 and miR-192-5p were significantly lower in active TED than in inactive TED. These miRNAs have the potential to serve as serum biomarkers of disease activity in TED. However, studies with larger sample sizes are needed to verify the roles of these miRNAs in the pathogenesis of TED.
Data availability
The datasets generated and analyzed during the present study are available in the Gene Expression Omnibus repository (GSE207995).
References
Bahn, R. S. Graves’ ophthalmopathy. N. Engl. J. Med. 362, 726–738 (2010).
Rao, R., MacIntosh, P. W., Yoon, M. K. & Lefebvre, D. R. Current trends in the management of thyroid eye disease. Curr. Opin. Ophthalmol. 26, 484–490 (2015).
Smith, T. J. et al. Teprotumumab for thyroid-associated ophthalmopathy. N. Engl. J. Med. 376, 1748–1761 (2017).
Strianese, D. Update on Graves disease: Advances in treatment of mild, moderate and severe thyroid eye disease. Curr. Opin. Ophthalmol. 28, 505–513 (2017).
Mourits, M. P. et al. Clinical criteria for the assessment of disease activity in Graves’ ophthalmopathy: A novel approach. Br. J. Ophthalmol. 73, 639–644 (1989).
Dolman, P. J. Grading severity and activity in thyroid eye disease. Ophthalmic Plast. Reconstr. Surg. 34, S34-s40 (2018).
**ao, C. & Rajewsky, K. MicroRNA control in the immune system: Basic principles. Cell 136, 26–36 (2009).
Chen, X., **e, D., Zhao, Q. & You, Z. H. MicroRNAs and complex diseases: From experimental results to computational models. Brief. Bioinform. 20, 515–539 (2019).
Zen, K. & Zhang, C. Y. Circulating microRNAs: A novel class of biomarkers to diagnose and monitor human cancers. Med. Res. Rev. 32, 326–348 (2012).
Zeng, L., Cui, J., Wu, H. & Lu, Q. The emerging role of circulating microRNAs as biomarkers in autoimmune diseases. Autoimmunity 47, 419–429 (2014).
Mi, S., Zhang, J., Zhang, W. & Huang, R. S. Circulating microRNAs as biomarkers for inflammatory diseases. MicroRNA 2, 63–71 (2013).
Bahn, R. S., Garrity, J. A., Bartley, G. B. & Gorman, C. A. Diagnostic evaluation of Graves’ ophthalmopathy. Endocrinol. Metab. Clin. North Am. 17, 527–545 (1988).
Yao, Q. et al. Circulating microRNA-144-3p and miR-762 are novel biomarkers of Graves’ disease. Endocrine 65, 102–109 (2019).
Pritchard, C. C., Cheng, H. H. & Tewari, M. MicroRNA profiling: Approaches and considerations. Nat. Rev. Genet. 13, 358–369 (2012).
Vlachos, I. S. et al. DIANA-miRPath v3.0: Deciphering microRNA function with experimental support. Nucleic acids Res. 43, W460 (2015).
Huang, H. Y. et al. miRTarBase 2020: Updates to the experimentally validated microRNA-target interaction database. Nucleic Acids Res. 48, D148-d154 (2020).
Kanehisa, M., Goto, S., Sato, Y., Furumichi, M. & Tanabe, M. KEGG for integration and interpretation of large-scale molecular data sets. Nucleic Acids Res. 40, D109–D114 (2012).
Li, Y. & Kowdley, K. V. Method for microRNA isolation from clinical serum samples. Anal. Biochem. 431, 69–75 (2012).
Yin, L., Huang, C. H. & Ni, J. Clustering of gene expression data: Performance and similarity analysis. BMC Bioinform. 7(Suppl 4), S19 (2006).
Permuth, J. B. et al. A pilot study to troubleshoot quality control metrics when assessing circulating miRNA expression data reproducibility across study sites. Cancer Biomark. 33, 467–478 (2022).
Jang, S. Y., Chae, M. K., Lee, J. H., Lee, E. J. & Yoon, J. S. Role of miR-146a in the regulation of inflammation in an in vitro model of Graves’ orbitopathy. Invest. Ophthalmol. Vis. Sci. 57, 4027–4034 (2016).
Jang, S. Y. et al. Role of microRNA-146a in regulation of fibrosis in orbital fibroblasts from patients with Graves’ orbitopathy. Br. J. Ophthalmol. 102, 407–414 (2018).
Tong, B. D., **ao, M. Y., Zeng, J. X. & **ong, W. MiRNA-21 promotes fibrosis in orbital fibroblasts from thyroid-associated ophthalmopathy. Mol. Vis. 21, 324–334 (2015).
Zhang, W. et al. Low serum exosomal miR-484 expression predicts unfavorable prognosis in ovarian cancer. Cancer Biomark. 27, 485–491 (2020).
Li, Y., Liu, Y., Yao, J., Li, R. & Fan, X. Downregulation of miR-484 is associated with poor prognosis and tumor progression of gastric cancer. Diagn. Pathol. 15, 25 (2020).
Lee, D., Tang, W., Dorsey, T. H. & Ambs, S. miR-484 is associated with disease recurrence and promotes migration in prostate cancer. Biosci. Rep. https://doi.org/10.1042/BSR20191028 (2020).
Konaka, T., Kawami, M., Yamamoto, A., Yumoto, R. & Takano, M. miR-484: A possible indicator of drug-induced pulmonary fibrosis. J. Pharm. Pharm. Sci. 23, 486–495 (2020).
El-Maraghy, S. A. et al. Circulatory miRNA-484, 524, 615 and 628 expression profiling in HCV mediated HCC among Egyptian patients; implications for diagnosis and staging of hepatic cirrhosis and fibrosis. J. Adv. Res. 22, 57–66 (2020).
Pan, D. et al. Inhibition of TGF-β repairs spinal cord injury by attenuating EphrinB2 expressing through inducing miR-484 from fibroblast. Cell Death Discov. 7, 319 (2021).
Ren, F. J., Yao, Y., Cai, X. Y. & Fang, G. Y. Emerging role of MiR-192-5p in human diseases. Front. Pharmacol. 12, 614068 (2021).
Mysore, R. et al. MicroRNA-192* impairs adipocyte triglyceride storage. Biochem. Biophys. Acta. 1861, 342–351 (2016).
Liu, X. L. et al. miR-192-5p regulates lipid synthesis in non-alcoholic fatty liver disease through SCD-1. World J. Gastroenterol. 23, 8140–8151 (2017).
Shen, L. et al. Circulating microRNA predicts insensitivity to glucocorticoid therapy in Graves’ ophthalmopathy. Endocrine 49, 445–456 (2015).
Bartalena, L. et al. Consensus statement of the European group on Graves’ orbitopathy (EUGOGO) on management of Graves’ orbitopathy. Thyroid 18, 333–346 (2008).
Jeon, H., Lee, J. Y., Kim, Y. J. & Lee, M. J. Clinical relevance of thyroid-stimulating immunoglobulin as a biomarker of the activity of thyroid eye disease. Eye https://doi.org/10.1038/s41433-022-01981-z (2022).
Wei, H. et al. Circulating levels of miR-146a and IL-17 are significantly correlated with the clinical activity of Graves’ ophthalmopathy. Endocr. J. 61, 1087–1092 (2014).
Chen, X., Sun, L. G. & Zhao, Y. NCMCMDA: miRNA-disease association prediction through neighborhood constraint matrix completion. Brief. Bioinform. 22, 485–496 (2021).
Chen, X., Li, T. H., Zhao, Y., Wang, C. C. & Zhu, C. C. Deep-belief network for predicting potential miRNA-disease associations. Brief. Bioinform. https://doi.org/10.1093/bib/bbaa186 (2021).
Chen, X., Zhu, C. C. & Yin, J. Ensemble of decision tree reveals potential miRNA-disease associations. PLoS Comput. Biol. 15, e1007209 (2019).
Funding
This research was supported by the National Research Foundation (NRF) grant funded by the Korea government (No. 2019R1G1A1100257). The funding organization had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
N.K., H-K.C, S.I.K and M.J.L conceived and designed the study. S.E.W and M.K.Y. collected the data, analyzed the experimental data. Y.J.K and S.E.W. did part of the experiments. All authors designed the work, read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, N., Choung, H., Kim, Y.J. et al. Serum microRNA as a potential biomarker for the activity of thyroid eye disease. Sci Rep 13, 234 (2023). https://doi.org/10.1038/s41598-023-27483-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-27483-w
- Springer Nature Limited