Abstract
The medicinal value of the Ferula L. has been recognized for more than a thousand years. Wild stocks of Ferula have declined dramatically because high economic value has led to overharvesting. The objective of this study was to compare the rhizosphere microbial community of four Ferula species [F. syreitschikowii K.-Pol., F. gracilis (Ledeb.) Ledeb., F. ferulaeoides (Steud.) Korov., and F. lehmannii Boiss.] in the northern part of **njiang, China. The 16S rRNA sequences of rhizosphere bacteria were obtained with an Illumina paired-end sequence platform. Analysis was conducted to determine the richness and diversity of the rhizosphere bacterial communities. Two-way ANOVA indicated that plant species and soil depth had no significant effect on the alpha diversity of rhizobacteria. Linear discriminant analysis effect size showed that F. lehmannii followed by F. ferulaeoides had the most biomarkers and the highest taxon level, F. syreitschikowii and F. gracilis the least, while F. syreitschikowii and F. gracilis had the least property. This trend is consistent with reports that the medicinal value of F. lehmannii and F. ferulaeoides is greater than that of F. gracilis and F. syreitschikowii. The results of this study provide information that could be used for the commercial cultivation of Ferula spp.
Similar content being viewed by others
Introduction
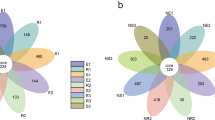
Twenty-six Ferula species and one Ferula variant are present in China1. Twenty of these species are found in the ** histories and environmental conditions. Microbial Ecology 68, 794–806 (2014)." href="/article/10.1038/s41598-018-22802-y#ref-CR37" id="ref-link-section-d262096482e1078">37,38. Two-way ANOVA indicated that Ferula species significantly affected the relative abundance of Actinobacteria (P < 0.01), Gemmatimonadetes (P < 0.01), Bacteroidetes (P < 0.01), Acidobacteria (P < 0.01), Verrucomicrobia (P < 0.05) and Thaumarchaeota (P < 0.01). Soil depth significantly affected the relative abundance of Proteobacteria (P < 0.05), Acidobacteria (P < 0.05), Firmicutes (P < 0.05) and Verrucomicrobia (P < 0.01).
Figure 2b shows the top ten bacterial classes in the rhizosphere soil. Gammaproteobacteria had the highest abundance (15.28%), followed by unidentified_Gemmatimonadetes (8.90%) and Thermoleophilia (8.89%). The relative abundance of unidentified_Gemmatimonadetes (P < 0.01), Thermoleophilia(P < 0.05), unidentified_Acidobacteria (P < 0.01), unidentified_Actinobacteria (P < 0.05), Acidimicrobiia (P < 0.01), Cytophagia (P < 0.05) and Sphingobacteriia (P < 0.01) were significantly affected by Ferula species. The abundance of Sphingobacteriia was significantly affected by soil depth (P < 0.01).
Soil Physicochemical Properties
Soil physicochemical properties vary among soil types and with soil depth. Therefore, we measured 12 common soil physiochemical properties (Table S1) and then analyzed the correlation between these properties and the relative abundance of bacteria at the phylum and class levels. Two-way ANOVA indicated that Ferula species and soil depth significantly affected all 12 Soil physicochemical properties (P < 0.01).
By factor analysis, we reduced 12 physicochemical factors to 4 principal components. The results of the correlation analysis of these four components with the relative abundance of bacteria in the rhizosphere are shown in Table S2 (phylum level) and Table S3 (class levels). We found that 12 physicochemical factors in rhizobacteria also have an impact on each other. For example, nitrate nitrogen and ammonium nitrogen are closely related, and they have impact on certain bacteria consistently.
Overall, these results suggest that soil properties were positively correlated with the relative abundance of bacteria.
Alpha Diversity
Alpha Diversity is used to analyze the diversity of species within community samples39. In general, sequences with greater than 97% identity are clustered into one operational taxonomic unit. The Alpha Diversity analysis index for different samples at a 97% identity are shown in Table 2. Two-way ANOVA indicated no significant differences among soil samples in observed species, Shannon, Simpson, Chao1, and ACE. Good’s coverage ranged from 97 to 98%, indicating that the measurement depth has met the requirements.
In summary, we propose a hypothesis that the reason that some of the soil bacteria are sensitive to soil depth and medicinal value is that these microorganisms are sensitive to volatile oil from plant.
As the rhizosphere soil volatile substances can not only come from the roots, but also from the ground parts of Ferula. For example, shoots can emit volatile substances in gaseous form which can diffuse into the surface soil, the rainwater can dissolve the volatile material to bring it into the soil. These processes gradually become less pronounced with the soil depth increased. So the abundance of some volatile-sensitive bacteria is higher in the upper soil (3 cm) and lower in the deeper soil (20 and 40 cm).
The results of LEfSe suggest that Ferula species with greater medicinal efficacy have a more unique microbial community (biomarkers) in the rhizosphere. The likely reason is that Ferula species with high medicinal efficacy produce more and better Ferula gum and volatile substances than species with low medicinal efficacy. That is to say, the medicinal value of Ferula is closely related to this volatile substance. The more volatile material the flavor has, the higher medicinal value is. We hypothesize that the differences in microbial populations among Ferula species were primarily due to differences in the production of volatile substances which can increase microbial diversity and abundance.
One shortcoming in our study is that the four Ferula species in this study were collected from areas which differed in geographical environment and climatic conditions. Ecological and geographical conditions greatly influence the terpenoid content of Ferula plants43. Therefore, the plant species factor in this study also includes the effects of the different environments in which the plants were growing. However, it is rare to find two Ferula species in the same field. We are not aware of any places where Ferula is being cultivated, so we could not obtain Ferula plants that had been grown under identical conditions. Each Ferula species has its unique habitat. Thus to a certain extent, the four Ferula species represent the environment in which they were growing.
Previous studies have examined the rhizosphere microbial community of F. sinkiangensis and F. fukanensis24,25. This study was the first to describe the rhizosphere microbial community of F. syreitschikowii, F. gracilis, F. ferulaeoides, and F. lehmannii. Many studies have compared the rhizosphere microbial community among species within a genus. Liu proposed that tree species influenced microbial diversity and nitrogen availability in rhizosphere soil44. Ladygina and Hedlun observed that plant species influenced microbial diversity and carbon allocation in the rhizosphere soil45. Cleary reported that root depth and plant species influenced microbiological parameters and bacterial composition in a mercury contaminated salt marsh46.
In conclusion, the results indicate that soil depth had some effect on bacterial community structure. And some rhizosphere bacteria are sensitive to soil depth and medicinal value of Ferula. The abundance of specific rhizosphere bacteria become higher as the medicinal value of the Ferula species increased and the soil depth decreased. This discovery provides insight about the ecological characteristics of Ferula. More information is still needed, but perhaps we can increase the survival rate of cultivated Ferula by artificially increasing the abundance of certain microorganisms.
References
Editorial Board for flora of China, C. A. o. & Sciences. Flora of China. Vol. 55 (1990).
Alkhatib, R. et al. Activity of elaeochytrin A from Ferula elaeochytris on leukemia cell lines. Phytochemistry 69, 2979–2983 (2008).
Suzuki, K. et al. Sesquiterpene lactones from the roots of Ferula varia and their cytotoxic activity. Journal of Natural Products 70, 1915–1918 (2007).
Motai, T. & Kitanaka, S. Sesquiterpene chromones from Ferula fukanensis and their nitric oxide production inhibitory effects. Journal of Natural Products 67, 432–436 (2004).
Jahansooz, F. et al. Composition and Antifungal Activity of the Oil of Ferula Gummosa Samples from Iran. Journal of Essential Oil Bearing Plants 11, 284–291 (2008).
Lin, N. et al. Preliminary study on bacteriostatic activity of essential oils in vitro of Ferula plant. **njiang Agricultural Science and Technology 4, 71–71 (2008).
Kartal, N. et al. Investigation of the antioxidant properties of Ferula orientalis L. using a suitable extraction procedure. Food Chemistry 100, 584–589 (2007).
Lee, C. L. et al. Influenza A (H(1)N(1)) Antiviral and Cytotoxic Agents from Ferula assa-foetida. Journal of Natural Products 72, 1568 (2009).
El‐Razek, M. H. A., Ohta, S. & Hirata, T. Terpenoid Coumarins of the Genus Ferula. Cheminform 34 (2003).
Heijden, M. G. A. V. D., Bardgett, R. D. & Straalen, N. M. V. The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecology Letters 11, 296–310 (2010).
Ghini, R., Hamada, E. & Bettiol, W. Climate change and plant diseases. Sci Agric. Scientia Agricola 65, 98–107 (2008).
Bell, C. W. et al. Linking microbial community structure and function to seasonal differences in soil moisture and temperature in a Chihuahuan desert grassland. Microbial Ecology 58, 827–842 (2009).
Berg, G. & Smalla, K. Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. Fems Microbiology Ecology 68, 1–13 (2009).
Marschner, P., Yang, C. H., Lieberei, R. & Crowley, D. E. Soil and plant specific effects on bacterial community composition in the rhizosphere. Soil Biology & Biochemistry 33, 1437–1445 (2001).
İnceoğlu, Ö., Salles, J. F. & Elsas, J. D. V. Soil and Cultivar Type Shape the Bacterial Community in the Potato Rhizosphere. Microbial Ecology 63, 460–470 (2012).
Singh, B. K., Munro, S., Potts, J. M. & Millard, P. Influence of grass species and soil type on rhizosphere microbial community structure in grassland soils. Applied Soil Ecology 36, 147–155 (2007).
Lima, A. B. et al. Amazonian Dark Earth and Plant Species from the Amazon Region Contribute to Shape Rhizosphere Bacterial Communities. Microbial Ecology 69, 855–866 (2015).
Aira, M., Gómez-Brandón, M., Lazcano, C., Bååth, E. & Domínguez, J. Plant genotype strongly modifies the structure and growth of maize rhizosphere microbial communities. Soil Biology & Biochemistry 42, 2276–2281 (2010).
Paterson, E., Gebbing, T., Abel, C., Sim, A. & Telfer, G. Rhizodeposition shapes rhizosphere microbial community structure in organic soil. New Phytologist 173, 600–610 (2007).
Bais, H. P., Weir, T. L., Perry, L. G., Gilroy, S. & Vivanco, J. M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annual Review of Plant Biology 57, 233–266 (2006).
Jackson, L. E., Bowles, T. M., Hodson, A. K. & Lazcano, C. Soil microbial-root and microbial-rhizosphere processes to increase nitrogen availability and retention in agroecosystems. Current Opinion in Environmental Sustainability 4, 517–522 (2012).
Newton, A. C., Fitt, B. D., Atkins, S. D., Walters, D. R. & Daniell, T. J. Pathogenesis, parasitism and mutualism in the trophic space of microbe-plant interactions. Trends in Microbiology 18, 365–373 (2010).
Fu-suo, L. Y.-h. Z. The Advances in Rhizosphere Microbiology. Soils 38, 113–121 (2006).
Zhu, J., Sun, L., Fan, C. & Li, X. Study on the dynamic variation of the population and the number of rhizosphere microorganism communities of Ferula fukanensis K. M. Shen. Lishizhen Medicine and Materia Medica Research 1, 203–205 (2015).
Zhu, J., Sun, L., Zhao, L. & Li, X. Study on the dynamic variation of the population and the number of rhizosphere microorganism communities of Ferula sinkiangensis K. M. Shen. Journal of Chinese Medicinal Materials 38, 265–266 (2015).
Riley, D. & Barber, S. A. Bicarbonate Accumulation and pH Changes at the Soybean (Glycine max (L.) Merr.) Root-Soil Interface. Soil Science Society of America Journal 33, 905–908 (1969).
Riley, D. & Barber, S. A. Salt accumulation at the soybean (Glycine max (L.) Merr) root-soil interface. Soil Science Society of America Journal 34, 154–155 (1970).
Mago, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957 (2011).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature methods 7, 335 (2010).
Bokulich, N. A. et al. Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nature Methods 10, 57 (2012).
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C. & Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27, 2194–2200 (2011).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Methods 10, 996 (2013).
Desantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Applied & Environmental Microbiology 72, 5069–5072 (2006).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied & Environmental Microbiology 73, 5261–5267 (2007).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research 32, 1792–1797 (2004).
Bao, S. D. Soil Agro-chemistrical Analysis. 22–196 (China Agriculture Press, 2008).
Tian, Y. & Gao, L. Bacterial diversity in the rhizosphere of cucumbers grown in soils covering a wide range of cucumber crop** histories and environmental conditions. Microbial Ecology 68, 794–806 (2014).
Jiang, X. T. et al. Illumina Sequencing of 16S rRNA Tag Revealed Spatial Variations of Bacterial Communities in a Mangrove Wetland. Microbial Ecology 66, 96 (2013).
Li, B., Zhang, X., Guo, F., Wu, W. & Zhang, T. Characterization of tetracycline resistant bacterial community in saline activated sludge using batch stress incubation with high-throughput sequencing analysis. Water Research 47, 4207–4216 (2013).
Segata, N. et al. Metagenomic biomarker discovery and explanation. 12, R60 (2011).
Uren, N. Types, Amounts, and Possible Functions of Compounds Released into the Rhizosphere by Soil-Grown Plants. English, 1–21 (2000).
Rajanikanth, B., Ravindranath, B. & Shankaranarayana, M. L. Volatile polysulphides of asafoetida. Phytochemistry 23, 899–900 (1984).
Saidkhodzhaev, A. I. & Mamatkhanov, A. U. Terpenoids of plants of the Ferula genus. Chemistry of Natural Compounds 31, 645–656 (1995).
Liu, D., Liu, Y., Fang, S. & Tian, Y. Tree species composition influenced microbial diversity and nitrogen availability in rhizosphere soil. Plant Soil & Environment 61, 438–443 (2015).
Ladygina, N. & Hedlund, K. Plant species influence microbial diversity and carbon allocation in the rhizosphere. Soil Biology & Biochemistry 42, 162–168 (2010).
Cleary, D. F. R. et al. Impact of sampling depth and plant species on local environmental conditions, microbiological parameters and bacterial composition in a mercury contaminated salt marsh. Marine Pollution Bulletin 64, 263 (2012).
Acknowledgements
This work was financially supported by the Chinese National Basic Research Program (2014CB954203) and the National Natural Science Foundation of China (31360139, 41561010, 31560177).
Author information
Authors and Affiliations
Contributions
Professor Li Zhuang and **uling Wang designed the experiment. **uling Wang and Zhongke Wang analyzed the data. **uling Wang and ** Jiang wrote the article. Yaling He, Yudi Mu and **nhua Lv modified the article. **uling Wang collected the samples.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, X., Wang, Z., Jiang, P. et al. Bacterial diversity and community structure in the rhizosphere of four Ferula species. Sci Rep 8, 5345 (2018). https://doi.org/10.1038/s41598-018-22802-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-22802-y
- Springer Nature Limited
This article is cited by
-
Meta-analysis of root-associated bacterial communities of widely distributed native and invasive Poaceae plants in Antarctica
Polar Biology (2024)
-
Diversity and structure of the rhizosphere microbial communities of wild and cultivated ginseng
BMC Microbiology (2022)
-
High-throughput sequencing analysis of the rhizosphere arbuscular mycorrhizal fungi (AMF) community composition associated with Ferula sinkiangensis
BMC Microbiology (2020)
-
Isolation, functional characterization and efficacy of biofilm-forming rhizobacteria under abiotic stress conditions
Antonie van Leeuwenhoek (2019)
-
Amplicon sequencing reveals different microbial communities in living poplar wetwood and sapwood
Trees (2019)