Abstract
Dopaminergic systems play a major role in reward-related behavior and dysregulation of dopamine (DA) systems can cause several mental disorders, including depression. We previously reported that dopamine D2 receptor knockout (D2R−/−) mice display increased anxiety and depression-like behaviors upon chronic stress. Here, we observed that chronic stress caused myelin loss in wild-type (WT) mice, while the myelin level in D2R−/− mice, which was already lower than that in WT mice, was not affected upon stress. Fewer mature oligodendrocytes (OLs) were observed in the corpus callosum of stressed WT mice, while in D2R−/− mice, both the control and stressed group displayed a decrease in the number of mature OLs. We observed a decrease in the number of active β-catenin (ABC)-expressing and TCF4-expressing cells among OL lineage cells in the corpus callosum of stressed WT mice, while such regulation was not found in D2R−/− mice. Administration of lithium normalized the behavioral impairments and myelin damage induced by chronic stress in WT mice, and restored the number of ABC-positive and TCF4-positive OLs, while such effect was not found in D2R−/− mice. Together, our findings indicate that chronic stress induces myelin loss through the Wnt/β-catenin signaling pathway in association with DA signaling through D2R.
Similar content being viewed by others
Introduction
Dopamine (DA) regulates emotional and motivational behavior, and changes in dopaminergic neurotransmission have been found to modify behavioral responses to various environmental stimuli such as stress1,2. Stress is thought to be a key factor in the development of depression, as reported by numerous animal and human studies3,4. It has been suggested that the dopamine system is highly susceptible to stress and is altered by stressful stimuli; this alteration contributes to the pathophysiology of stress-induced depressive disorders2,4,5,6,7. Increasing evidence suggests that dopamine agonists are effective for treating depression in humans8,9,10.
The dopamine D2 receptor (D2R), which is one of the major DA receptor subtypes, is known to be critically involved in the pathology of depression and is associated with stress11,12,13. We have previously shown that anxiety-like and depression-like behaviors after chronic stress are more pronounced in D2R knockout mice (D2R−/− mice) than in wild type (WT) mice14.
Depression is known to be associated with not only neuronal alteration but also glial dysfunction. In particular, it has been reported that patients with major depressive disorder display oligodendroglial abnormalities and alterations in oligodendrocyte (OL) structure, and that glial function can produce behavioral changes associated with mood regulation15,16. Transcriptional profiling analysis of patients with major depressive disorder revealed that changes in intercellular communication and signal transduction mechanisms that contribute to abnormalities in oligodendroglia and synaptic function may contribute to depressive disorder regulation15. Other studies have also shown significant reductions in the expression of myelin-related and OL-related genes in the brain of patients with depression15,17,18,19.
It has been reported that DA can affect myelin formation through OL development and function, in particular by D2Rs or D3Rs20,21, suggesting that a D2R agonist could increase the number of oligodendrocyte progenitor cells (OPCs). Notably, D2R agonism has been shown to provide significant protection of oligodendrocytes against oxidative injury22.
The current study addressed whether DA signals via D2R regulate myelination in stress-induced depression by examining myelination changes in response to chronic stress in WT and D2R−/− mice. We found that stress induced a loss of myelination in WT mice which was protected with treatment of the antidepressant, lithium. D2R−/− mice showed alterations in this stress-induced myelin loss; however, they were not responsive to lithium treatment. Our results show that the D2R has a key role in altering myelination, which can be associated with pathology of DA-related neuropsychiatric diseases.
Results
Chronic stress-induced myelin loss in WT and D2R−/− mice
WT and D2R−/− mice assigned to “stressed” groups were subjected to restraint stress by immobilization in a restrainer for 2 h daily for 14 d, as described previously14 (Fig. 1A). Control groups of mice were left undisturbed. After stress exposure, WT and D2R−/− mice subjected to chronic stress were evaluated in the forced swim test. In this test, immobility time is considered an indicator of a depressive state. The stress-induced increase in immobility time in D2R−/− mice was significantly longer than that of WT mice (stress × genotype interaction: F1,30 = 5.43, P = 0.027; Fig. 1B). This finding is in accordance with previous data showing increased levels of depressive behaviors in D2R−/− compared to WT animals following stress14.
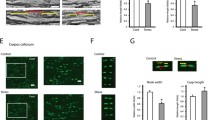
Effects of chronic stress on myelination in WT and D2R−/− mice. (A) Schematic illustration of the chronic immobilization stress model. For chronic stress, WT and D2R−/− mice were immobilized for 2 h once daily for 14 days. Control groups of mice were left undisturbed. (B) Immobilization time after chronic stress. Mice were subjected to the forced swim test after exposure to chronic restraint stress (St). Non-stressed (NSt) animals were examined as a control (WT-NSt n = 11, WT-St n = 7, D2R−/−-NSt n = 8, D2R-St n = 8). (C–F) Visualization of myelin by luxol fast blue (LFB) staining (C) and myelin basic protein (MBP) immunohistochemistry (E). WT mice following 5 weeks of cuprizone feeding (CPZ) were examined as the myelin loss control. (D,F) Quantitative analysis of LFB staining and MBP immunohistochemistry by integrated optical densitometry within the middle corpus callosum (region from bregma 0.02 mm to −0.82 mm). Scale bars: 50 μm (D, WT-NSt n = 6, WT-St n = 5, D2R−/−-NSt n = 6, D2R-St n = 5, CPZ n = 6; (F) WT-NSt n = 7, WT-St n = 6, D2R−/–-NSt n = 6, D2R-St n = 5, CPZ = 6) (G–K), Transmission electron micrographs of transverse sections at the middle corpus callosum (bregma −0.46 mm, mid-sagittal 0 mm) (n = 5, scale bars represent 2 μm). Approximately 1500 axons from 5 mice were investigated in each group. Average myelin thickness (H) and g-ratio (I) in the middle corpus callosum were measured and grouped by axon diameter (J,K) (n = 5 mice for each group). White bars indicate non-stressed animals, black bars indicate stressed animals and gray bars indicate cuprizone-fed animals. Data are presented as mean ± SEM. **P < 0.01, ***P < 0.001 versus NSt mice; #P < 0.05, ##P < 0.01, ###P < 0.001 versus WT-NSt mice; +P < 0.01 versus genotype (Two-way ANOVA post hoc test). §P < 0.05, §§P < 0.01 versus WT-NSt mice (Two-tailed Student’s t-test).
We determined whether chronic stress affects myelination in WT and D2R−/− mice. Luxol fast blue (LFB) myelin staining was used for measuring the extent of myelin loss23. As a myelin loss control, we used the copper chelator cuprizone (CPZ), which can induce severe demyelination with oligodendrocyte damage in animals after 5 to 6 weeks of CPZ feeding24,25 (Fig. 1C). We observed severe myelin loss of the corpus callosum of stressed WT mice (WT-St) compared to non-stressed control WT mice (WT-NSt) (Fig. 1C,D). By analyzing the density of LFB staining, the white matter of the corpus callosum spanning from bregma 1.10 mm to −2.30 mm was evaluated. We found that most of the corpus callosum between 0.14 mm and −0.82 mm was significantly demyelinated in the WT-St group mice as compared to the control group. This loss of white matter was comparable with the cuprizone (CPZ)-fed (for 5 weeks) animals, where the LFB staining intensity was decreased up to 55% in the corpus callosum. Interestingly, a low level of LFB staining density was already observed in non-stressed D2R−/− mice (D2R−/−-NSt) (51% versus WT-NSt group) and stressed D2R−/− mice (D2R−/−-St) group displayed a similar level of LFB staining density (Fig. 1C,D, stress × genotype interaction: F1,18 = 5.65, P = 0.029). In parallel, we have also analyzed the representative myelin-specific protein, Myelin Basic Protein (MBP)26 in the corpus callosum area by immunohistochemistry and observed a significant decrease of MBP expression in the WT-St mice compared to the WT-NSt control group (71% of WT-NSt group). In D2R−/− mice, the non-stressed control group showed a low level of MBP which was not affected by stress subjection (78% of control WT mice, stress × genotype interaction: F1,20 = 11.33, P = 0.003; Fig. 1E,F). These findings suggest that myelin is decreased by stress in WT mice while the basal myelin level is low in D2R−/− mice, and that there is no change of myelin status by chronic stress in these mice.
Ultrastructural analysis of myelinated axons in the corpus callosum by electron microscopy revealed a significant decrease in myelin thickness of axons within different diameter scales (223 ± 11 nm in WT-NSt control, n = 5 and 156 ± 10 nm, in WT-St, n = 5) and an increase in the g-ratio (0.741 ± 0.010 in WT-NSt, n = 5 and 0.816 ± 0.013, WT-St, n = 5, g-ratio: axon diameter/outer myelin sheath diameter) in the WT-St group compared to the WT-NSt group (Fig. 1G–K). Approximately 1500 axons from 5 mice were investigated in each group. In the corpus callosum of D2R−/−-NSt control mice; however, basal myelin thickness was already low (153 ± 8 nm, n = 5, 69% of control WT mice, myelin thickness: stress × genotype interaction: F1,16 = 11.83, P = 0.003; Fig. 1H) and the g-ratio was increased as compared to WT control mice (0.827 ± 0.009, n = 5, 112% of control WT mice, stress × genotype interaction: F1,16 = 16.23, P = 0.001; Fig. 1I). A similar pattern of myelin level was observed in D2R−/−-St mice (myelin thickness: 148 ± 3 nm, n = 5, g-ratio: 0.823 ± 0.007, n = 5). This myelin level in D2R−/− mice was comparable to that of CPZ-fed animals (myelin thickness: 157 ± 13 nm, n = 5; g-ratio, 0.833 ± 0.010, n = 5). These myelin thickness and g-ratio changes were observed across the different axon diameter scales (stress × genotype interaction: myelin thickness, category < 0.5, F1,16 = 8.8, P = 0.009; category 0.5 < 1.0, F1,16 = 15.02, P = 0.001; category 1.0 < 1.5, F1,16 = 13.46, P = 0.002; category > 1.5, F1,16 = 6.74, P = 0.02; g-ratio, category < 0.5, F1,16 = 8.75, P = 0.009; category 0.5 < 1.0, F1,16 = 18.92, P = 0.0005; category 1.0 < 1.5, F1,16 = 22.13, P = 0.0002; category > 1.5, F1,16 = 7.07, P = 0.017; paired, two-tailed Student’s t-test: myelin thickness, category < 0.5, P = 0.0048; category 0.5 < 1.0, P = 0.0291; category 1.0 < 1.5, P = 0.0168; category > 1.5, P = 0.046; for WT non-stress versus CPZ: g-ratio, category < 0.5, P = 0.051; category 0.5 < 1.0, P = 0.0151; category 1.0 < 1.5, P = 0.0142; category > 1.5, P = 0.002, Fig. 1J,K). These results strongly indicate that chronic stress can cause severe myelin loss in adult WT mice and that the absence of D2R results in loss of myelin and similar level of myelin loss was observed in both D2R−/−-NSt and D2R−/−-St mice.
Stress-induced myelin loss involves a decrease in mature oligodendrocytes in WT mice
To examine in more detail the altered myelination in stressed WT and D2R−/− mice, we identified OL cell populations in the corpus callosum of each group and determined the number of oligodendrocyte precursor cells (OPCs) and mature OLs among all the OL lineage cells. PDGFRα (platelet-derived growth factor receptor, alpha polypeptide) is a known marker of oligodendrocyte precursors27, and it has been shown that the adenomatous polyposis coli (APC) protein is a marker for mature OLs28. Oligodendrocyte transcription factor (Olig2) is a basic helix-loop-helix (bHLH) transcription factor encoded by the Olig2 gene and Olig2 labels OPCs and mature OLs, and is thus indicative of all OL lineage cells29. The number of OPCs (PDGFRα+), APC-positive cells (APC+, representing mature OLs), and Olig2-positive cells (Olig2+, representing all OL lineage cells) were enumerated in control and stressed animals. By double immunofluorescence analysis, we found that in the corpus callosum of WT-St mice, Olig2+ and APC+ cells were decreased and APC+/Olig2+ cells were decreased by 47% of the WT-NSt control group (Fig. 2A,C). In the corpus callosum of D2R−/− mice, both D2R−/−-NSt and D2R−/−-St groups of mice displayed a significant decrease of Olig2+ and APC+ cells, and APC+/Olig2+ cells by 48–53% of WT control mice (D2R−/−-NSt mice, 48% of WT-NSt mice; D2R−/−-St mice, 53% of WT-NSt mice, stress × genotype interaction: F1,24 = 4.55, P = 0.043, Fig. 2A,C). In contrast, the number of PDGFRα+ cells was comparable across the corpus callosum of both WT and D2R−/− mice, and was not changed by stress (stress × genotype interaction: F1,28 = 0.54, P = 0.47, Fig. 2B,D). These results suggest that chronic stress-induced myelin loss involves a decrease in the number of mature OLs in WT mice and in D2R−/− mice, the latter which already had decreased number of mature OLs and similar level of OLs number is observed in D2R−/−-St mice.
Quantitative analysis of mature oligodendrocytes in stress-imposed WT and D2R−/− mice. (A,B) Identification of mature oligodendrocytes and oligodendrocyte precursor cells after chronic stress. Immunolabeling performed using antibodies to mature oligodendrocyte marker APC (CC1 clone), oligodendrocyte lineage marker Olig2, and premature oligodendrocyte marker PDGFRα. Representative low and high magnification immunofluorescence images of APC(green)/Olig2(red) (A) and PDGFRα (red)- positive cells (B) in the middle corpus callosum of stress-imposed WT and D2R−/− mice (St). Non-stressed (NSt) animals were examined as a control. White dashed line indicates corpus callosum region and white dashed box shows magnified image presented at far right panel. Scale bars: 50 μm in low magnification images and 10 μm in high magnification images. cc: corpus callosum, cg: cingulate gyrus, LSD: lateral septal nucleus. (C,D) The percentage of APC/Olig2 and PDGFRα positive cells in middle corpus callosum for non-stressed and stressed animals in WT and D2R−/− mice (C) WT-NSt n = 8, WT-St n = 7, D2R−/−-NSt n = 7, D2R-St n = 6; (D) n = 8 mice for each group). White bars indicate non-stressed animals, black bars indicate stressed animals. Data are presented as mean ± SEM. *P < 0.05 versus NSt mice; #P < 0.05 versus WT mice (Two-way ANOVA post hoc test).
Wnt/β-catenin-TCF signaling is associated with stress-induced myelin loss
Further, we examined the mechanism by which stress-induced myelin loss is mediated and determine why a low level of myelin is observed in D2R−/− mice despite similar level of OPC cells as WT mice.
Recent studies have shown that the Wnt/β-catenin-TCF/LEF signaling pathway is critical in the regulation of oligodendrogenesis, OL differentiation, and myelination29,30,31,32,33,52,53,54. Benedetti et al., for example, reported that adverse childhood experiences alter white matter microstructure including corpus callosum, in patients with bipolar disorder55. As well, brain imaging studies with major depressive disorder (MDD) patients showed that MDD patients have widespread abnormalities in the corpus callosum56,57,58. Therefore, together with other animal studies59,60,61, these observations indicate a strong association between alterations of the myelination of the corpus callosum with chronic stress-induced depressive behaviors.
In the present study, we demonstrated that chronic stress induced-myelin loss was regulated by Wnt/β-catenin signaling, and that this regulation was dependent on the DA system through D2R. Recent findings identified a role of the Wnt/β-catenin signaling in myelination, with initial reports suggesting, rather, an inhibitory role for Wnt signaling in OPC differentiation and OL maturation during early postnatal development29,31,62. Other studies using genetic and pharmacological antagonists to inhibit components of Wnt/β-catenin signaling also demonstrated that inhibition promoted OPC differentiation and remyelination in both in vitro and in vivo models63,64,65,66. In contrast, numerous reports have shown that activation of Wnt signaling induces myelination. For instance, Tawk et al.32, have demonstrated a key role for Wnt/β-catenin signaling in the expression of myelin-related genes and myelin sheath compaction in both in the peripheral and central nervous systems using loss-of-function analyses in zebrafish embryos. Inhibition of Wnt/β-catenin signaling resulted in hypomyelination without affecting Schwann cell and OL generation, or axonal integrity. It has also been reported that in vivo activation or inhibition of canonical Wnt signaling, respectively, increased or decreased the number of Olig2-positive and PDGFRα-positive cells in the adult subependymal zone, thereby showing the contribution of Wnt/β-catenin pathway to oligodendrogliogenesis, myelin gene expression, and myelinogenesis67. Furthermore, it was demonstrated that in Tcf4 gene KO mice, there are few changes in the number of OPCs; however, there is severe defects in oligodendrocyte maturation62,68. It has been reported that Tcf4 is expressed during remyelination stages in patients with multiple sclerosis and in the cuprizone mouse model of myelin loss, thereby supporting a role for Tcf4 in remyelination62,68,69,70. These contradictory findings indicate that Wnt pathway plays an important role in OL maturation and myelin formation, most likely in a complex manner, which depends on developmental stage and corresponding molecular networks. One possible explanation is that interaction of the Wnt signaling with other pathways can tightly adjust the balance between the negative and positive effects on myelination. In this context, our present finding suggests a possible link between DA and Wnt system through D2R regulation of remyelination, particularly under the stressful stimuli.
Given that lithium, generally known as a glycogen synthase kinase-3β inhibitor, treatment normalized chronic stress-induced Wnt signaling inactivation we also examined whether lithium treatment in the modulation of myelin in corpus callosum would involve glycogen synthase kinase-3β (GSK3β) signaling. However, immunofluorescent histochemistry performed using antibody to GSK3β revealed no signal of GSK3β in the corpus callosum (Supplementary Fig. 1A) while robust labeling of GSK3β could be detected in the cingulate gyrus (cg), caudate putamen (CPu) and primary somatosensory cortex (S1 cortex) (Supplementary Fig. 1B,C). Therefore, based on our data, we believe that the regulation we observed here involves rather a link between dopamine/lithium and Wnt/ β-catenin/Tcf4 signaling without direct involvement of GSK3β in this regulation of myelin in the corpus callosum.
Dopamine systems are considered important modulators of stress-related depressive illness which is associated with altered response to reward, thereby contributing to the pathophysiology of stress-evoked depression2,3,4,6,71. This deficiency in DA transmission involves not only reduced DA release but also abnormal density and sensitivity of dopamine receptors, resulting in dysfunction of DA receptors and disrupted DA transmission in response to external stimuli72,73. How dopamine systems with dysfunctional DA receptors contribute to the pathophysiology of depression is not yet known.
It has been reported that the dopamine system can regulate OL functions. For example, D2R expression was observed in interfascicular OLs around the peak of myelination, from P15 to P20, in rat brain21 and in differentiated OLs from rat cortical OL cultures22,74. In addition, a protective role of D2R against oxidative glutamate toxicity and oxygen-glucose deprivation injury in OLs22 has been demonstrated as chronic treatment with the D2R antagonist haloperidol reduced expression of myelin protein genes in mice75. Disrupted integrity in myelination observed in the corpus callosum of D2R−/− mice in the present study thus raises the possibility that D2R is a potential regulator of OL development and myelination formation. This is consistent with the observations made in this study, in which quinpirole treatment in stressed WT mice protected stress-induced myelin damage, while no such effect is observed in D2R−/− mice.
Another interesting finding of the present study is that in D2R−/− mice, upon chronic stress, Wnt singling pathway-mediated regulation of myelination was blunted, which supports the hypothesis that DA signaling through D2R, in association with the Wnt pathway, is critical for stress-induced myelin loss. Since stress can affect axon-myelin interaction, myelin formation and remyelination in response to stress must be tightly controlled by neurotransmission. Based on our present findings, we suggest that DA, via D2R, can modulate the Wnt pathway, which in turn controls stress-induced myelin loss, thereby contributing to the regulation of white matter integrity in stress-induced neurological disorders. Our findings may explain the complexity of antidepressant treatment resistance, known to be related a hypodopaminergic state, which can result in lack of effect owing to the absence of DA system activation. Indeed, it has been demonstrated that some D2R agonists such as pramipexole and aripiprazole are effective in patients with depression who have failed to respond to previous medications9,10,76,77. Our finding that the effect of lithium treatment was dependent on the presence of D2R may be related to this clinical situation. Consideration of DA signaling would not only be particularly important for synaptic modification in the regulation of myelination associated with the Wnt/β-catenin axis but also be promising as effective antidepressant medication.
Methods
Mice
D2R−/− mice (B6.129S2- Drd2tm1Low/J) were obtained from the Induced Mutant Resource at the Jackson Laboratory (Bar Harbor, ME). The D2R−/− mice and WT littermates originated from the breeding of D2R+/− heterozygotes. Mice were maintained in a specific pathogen-free barrier facility under constant conditions of temperature and humidity, and on a 12:12-h light-dark schedule. Animal care and handling were performed in accordance with standards approved by the Institutional Animal Care and Use Committee of Korea University.
Chronic Immobilization Stress
Behavioral experiments were performed with male D2R−/− and WT control mice at 11–13 weeks of age. Age-matched WT and D2R−/− mice were housed individually and allowed to acclimatize to the cage for 1 week. Mice assigned to stress groups were restrained in a ventilated acrylic restrainer fit to allow the animal to breathe but not to move otherwise. For chronic stress, they were immobilized for 2 h once daily for 14 days. WT and D2R−/− mice were placed in the home cage for restraint stress. Mice assigned to non-stressed groups were untouched in the home cage, whereas their counterparts were subjected to restraint stress.
Cuprizone-Induced Myelin loss
7-week-old WT male mice were fed 0.2% (w/w) cuprizone (bis-cyclohexanone oxaldihydrazone, 14690, Sigma-Aldrich, St. Louis, MI) mixed into a regular diet of ground standard chow (Purina Certified Rodent Diet, USA). The cuprizone diet was maintained for 5 weeks. Cuprizone ingestion results in a reproducible pattern of corpus callosum myelin loss over this 5-week period. Control “non-treated” mice were maintained on normal chow pellets.
Lithium treatment
Mice received either a lithium-supplemented diet (0.2% LiCO3; TD.08019, Teklad, Harlan) or the control diet. Since chronic lithium treatment can result in polyuria and increased excretion of sodium78, all mice were provided a second drinking bottle containing 0.9% saline to counteract potential ion imbalance79.
Quinpirole treatment
(−)-Quinpirole hydrochloride (Tocris, Bristol, UK) was dissolved in 0.9% saline and was administered intraperitoneally once daily (one hour before restraint) at doses of 5 mg/kg/day during chronic stress. Control group received an equivalent volume of 0.9% saline.
Tissue preparation
Animals were anesthetized by intraperitoneal (i.p.) injection of 1.6 μl of Zoletil and 0.05 μl of xylazine (Rompun, Bayer) per gram of body weight and transcardially perfused with 4% paraformaldehyde in PBS. Brains were removed and postfixed 4 h at 4 °C. The brains were cryoprotected in 30% sucrose-PBS solution for 2 days. Brains were then frozen and 20 µm thick consecutive coronal sections were obtained on a Cryotome (Leica CM 1900, Germany).
Luxol fast blue stain
Six sections were prepared from specific regions within the middle corpus callosum that showed severe myelin loss in LFB stain (region from bregma 0.14 mm to −0.82 mm, 0.16-mm intervals). Brain sections were dehydrated with ethanol grades, incubated in 0.1% luxol fast blue stain solution (solvent blue 38 0.1 g, ethyl alcohol 95% 100 ml, glacial acetic acid 0.5 ml) at 56 °C overnight, and washed with distilled water. Sections were dipped in 0.05% lithium carbonate 30 sec and 70% regent alcohol for gray and white matter differentiation. The slices were incubated in cresyl violet stain for 1 min, dipped in 70% regent alcohol, dehydrated through 2 changes of absolute alcohol, and mounted with Permount (Fisher Scientific). The myelin loss area and integrated optical density (IOD) were analyzed with MetaMorph 6.1 software (Universal Imaging Corp.; Downingtown, PA).
Electron microscope histological analysis (TEM)
After anesthesia, mice were perfused with 4% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M phosphate buffer. Dissected tissues containing the corpus callosum (bregma −0.46 mm) were prepared from 1 mm parasagittal serial brain sections. The tissues were buffered washed, fixed in 1% osmium tetroxide, dehydrated in graded ethanol series, transitioned in propylene oxide, and embedded in TAAB resin (TAAB Laboratories). Semi-thin sections (1 µm mid-sagittal, lateral 0 mm) were cut, mounted on glass slides, and stained in toluidine blue for general assessment in the light microscope. Subsequently, 50~60-nm ultra-thin sections were cut with a diamond knife (Diatome), mounted on copper slot grids coated with parlodion, and stained with uranyl acetate and lead citrate for examination on an electron microscope (Hitachi; H7650). Ten random images within each field were collected at 12000× magnification. The digitalized images were then analyzed on a MetaMorph image analysis system (Universal Imaging Corp.).
G-ratio analysis
Digitized, non-overlap** electron micrographs of corpus callosum were analyzed for axon diameter and g-ratio. Images were analyzed using MetaMorph 6.1 software. The g-ratio measurements were obtained by manually drawing lines across 2 perpendicular diameters for both the axons and for the axons plus myelin sheath. At least 300 axons per brain were measured. The analysis was modified such that the inner diameter of compact myelin (instead of the axon diameter) was divided by the outer diameter of the myelin sheath (because the inner tongue was frequently enlarged in demyelinated animals, but not in controls). Axons with diameters typical of unmyelinated fibers (<0.3 mm) were excluded from analysis. Fibers with prominent outfoldings in the plane of section were also excluded.
MBP Immunohistochemistry
Six sections were prepared within the middle corpus callosum that showed severe myelin loss in LFB stain (region from bregma 0.14 mm to −0.82 mm, 0.16-mm intervals). The 20-µm free-floating sections were washed in 0.1 M PBS (pH 7.4) three times for 10 min and placed in 0.1 M PBS solution containing 0.3% H2O2 and 50% methanol for 30 min. Sections were incubated in blocking solution (0.3% Triton X-100, 1% BSA in 0.1 M PBS) for 30 min and then in primary antibody a-MBP (1:2000; Abcam, 7349) at 4 °C overnight. This was followed by incubations at room temperature in biotinylated secondary antibody (anti-rat IgG 1:1000; Vector Laboratories, Burlingame, CA) for 30 min and in an ABC kit (Vector Laboratories, Burlingame, CA) for 30 min. Sections were reacted with DAB (Vector Laboratories, Burlingame, CA). The 0.1 M PBS rinses were performed between each step. Sections were mounted, dehydrated, and cover slipped.
Double-labeled immunofluorescence
Three sections were prepared from the middle corpus callosum that showed severe myelin loss in LFB stain (region from bregma −0.22 mm, −0.46 mm and −0.70 mm). The 20 µm free-floating sections were pre-treated with 1% NaOH and 1% H2O2 for 20 min, incubated in 0.3% glycine in PBS for 10 min, and blocked with a blocking solution (3% bovine serum albumin, and 0.3% Triton X-100 in PBS, pH 7.4) for 1 h at room temperature. Tissue sections were incubated overnight at 4 °C with a-TCF4 (1:500; Santacruz, sc-13027), a-APC(CC1) (1:250; Millipore, OP80), a-Olig2 (1:250; Santacruz, sc-19969), a-Olig2 (1:350; Millipore, AB9610), ABC (Anti-Active-β-Catenin Antibody clone 8E7; 1:100; Millipore, 05–665), and PDGFRα (1:200; rat 558774, BD Biosciences). After rinsing in PBS, the double-stained sections were incubated at room temperature for 1 h with Alexa Fluor 488 anti-Rabbit (donkey IgG) (1:500; Life Technologies, A-21206), Alexa Fluor 568 anti-mouse (donkey IgG) (1:1000; Life Technologies, A-10037), and Alexa Fluor 488 anti-Goat (donkey IgG) (1:500; Life Technologies, A-11057). After rinsing in PBS, the sections were mounted in Vectashield (Vector Laboratories) to prevent fading of the immunofluorescence stain. Sections were examined on a confocal laser scanning system, (LSM 700, Zeiss, Berlin, Germany).
Fluorescent in situ hybridization (FISH) and Olig2 immunofluorescence-histochemistry
Animals were anesthetized and transcardially perfused with 4% paraformaldehyde in PBS. Brains were removed and postfixed 4 h at 4 °C. Dehydrate the tissue in 10%, 20%, 30% sucrose in 1X PBS at 4 °C until the tissue sinks to the bottom of the container. Tissue were frozen at −20 °C in the Optimal Cutting Temperature (OCT) embedding media and 14 μm-thick coronal sections were placed on SuperFrost® Plus slides (Fisher Scientific #12-550-15) and air dry the slides for 20 min at −20 °C. FISH was performed according to RNAscope® ISH Tissue instructions (Advanced Cell Diagnostics (ACD), ACD 320850, Hayward, CA) for fixed-frozen tissue. Slides were digested with protease (1:15, diluted in PBS) for 30 min at 40 °C. Sections were incubated for 2 h at 40 °C in a hybridization oven (HybEZ™, ACD) with RNAscope® Probe- mm-Drd2-C3 (ACD 406501-C3) probes to detect mouse D2R mRNA. Ppib probe (ACD 313911-C3), a mouse housekee** gene, was used as a positive control probe, and a bacterial dapB probe (ACD 310043-C3) was used as a negative control. Sections were then incubated with Amp 1- FL, Amp 2- FL, Amp 3- FL, and 4-FL–Alt A solutions according to instructions. Wash with 0.1 M PBS twice, blocked with a blocking solution (5% bovine serum albumin, and 0.2% Triton X-100 in PBS, pH 7.4) for 1 h at room temperature. Tissue sections were incubated overnight at 4 °C with a-Olig2 (1:500; Millipore, AB9610). After rinsing in PBS, the double-stained sections were incubated at room temperature for 1 h with Alexa Fluor 488 anti-Rabbit (donkey IgG) (1:1000; Life Technologies, A-21206). After counterstaining with DAPI for 30 sec, slides were cover-slipped with Vectashield (Vector Laboratories) to prevent fading. Slides were imaged using confocal laser scanning system, (LSM 700, Zeiss, Berlin, Germany). Probe channel C3 was observed with a filter for Atto 647. To count D2R expressing cells in Olig2-positive cells in WT mice (Fig. 6A), cells were counted in the corpus callosum region within the range from bregma −0. 22 mm to −0.70 mm (4 sections per mouse) and summed data were used for quantification. Approximately 400 cells expressing D2R or Olig2 were counted and only cells with a D2R mRNA puncta fluorescent signals or Olig2 fluorescent signals that filled the cell body were included for analysis. Individual puncta were counted using MetaMorph image analysis system (Universal Imaging Corp.). In a similar way, the expression levels of D2R mRNA transcripts in stress-imposed WT mice with and without lithium treatment (Fig. 6C) was analyzed, puncta were counted in the corpus callosum region within the region from bregma −0.22 mm, −0.46 mm and −0.70 mm (3 sections per mouse) and summed data were used for quantification.
Forced swim test
Mice were transferred to the experimental room 60 min before the onset of the experiment to allow for habituation and reduce stress (brightness of the experimental room was 70 lux). The forced swim test was performed by drop** a mouse in water and recording its behavior. The apparatus consisted of a plastic cylinder (internal diameter, 10 cm; height, 25 cm) containing a 19 cm column of water maintained at 22 ± 2 °C. The mouse was thus not able to support itself by touching the bottom of the apparatus with its paws or tail. An animal was considered to be immobile when it floated in an upright position and made only small movements to maintain its head above water. The total duration of immobility, which is thought to reflect a depressive state, was determined during a 6-min test session.
Statistical analysis
Data are presented as means ± s.e.m. and were analyzed with one-way or two-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc test. A P-value of < 0.05 was considered statistically significant.
Change history
11 April 2018
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has not been fixed in the paper.
References
Baik, J. H. et al. Parkinsonian-like locomotor impairment in mice lacking dopamine D2 receptors. Nature 377, 424–428 (1995).
Baik, J. H. Dopamine signaling in reward-related behaviors. Front Neural Circuits 7, 152 (2013).
Zacharko, R. M. & Anisman, H. Stressor-induced anhedonia in the mesocorticolimbic system. Neurosci Biobehav Rev 15, 391–405 (1991).
Cabib, S. & Puglisi-Allegra, S. Stress, depression and the mesolimbic dopamine system. Psychopharmacology (Berl) 128, 331–342 (1996).
Pani, L., Porcella, A. & Gessa, G. L. The role of stress in the pathophysiology of the dopaminergic system. Mol Psychiat 5, 14–21 (2000).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).
Pittenger, C. & Duman, R. S. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology 33, 88–109 (2008).
Chiba, S., Numakawa, T., Ninomiya, M., Yoon, H. S. & Kunugi, H. Cabergoline, a dopamine receptor agonist, has an antidepressant-like property and enhances brain-derived neurotrophic factor signaling. Psychopharmacology (Berl) 211, 291–301 (2010).
Hori, H. & Kunugi, H. The efficacy of pramipexole, a dopamine receptor agonist, as an adjunctive treatment in treatment-resistant depression: an open-label trial. Sci World J 2012, 372474 (2012).
Kunugi, H., Hori, H. & Ogawa, S. Biochemical markers subty** major depressive disorder. Psychiat Clin Neurosci 69, 597–608 (2015).
Lawford, B. R., Young, R., Noble, E. P., Kann, B. & Ritchie, T. The D2 dopamine receptor (DRD2) gene is associated with co-morbid depression, anxiety and social dysfunction in untreated veterans with post-traumatic stress disorder. Eur Psychiat 21, 180–185 (2006).
Zhu, X., Peng, S., Zhang, S. & Zhang, X. Stress-induced depressive behaviors are correlated with Par-4 and DRD2 expression in rat striatum. Behav Brain Res 223, 329–335 (2011).
Zurawek, D. et al. Mesolimbic dopamine D2 receptor plasticity contributes to stress resilience in rats subjected to chronic mild stress. Psychopharmacology (Berl) 227, 583–593 (2013).
Sim, H. R. et al. Role of dopamine D2 receptors in plasticity of stress-induced addictive behaviours. Nat Commun 4, 1579 (2013).
Aston, C., Jiang, L. & Sokolov, B. P. Transcriptional profiling reveals evidence for signaling and oligodendroglial abnormalities in the temporal cortex from patients with major depressive disorder. Mol Psychiat 10, 309–322 (2005).
Edgar, N. & Sibille, E. A putative functional role for oligodendrocytes in mood regulation. Transl Psychiat 2, e109 (2012).
Klempan, T. A. et al. Altered expression of genes involved in ATP biosynthesis and GABAergic neurotransmission in the ventral prefrontal cortex of suicides with and without major depression. Mol Psychiat 14, 175–189 (2009).
Kim, S. & Webster, M. J. Correlation analysis between genome-wide expression profiles and cytoarchitectural abnormalities in the prefrontal cortex of psychiatric disorders. Mol Psychiat 15, 326–336 (2010).
Rajkowska, G. et al. Oligodendrocyte morphometry and expression of myelin - Related mRNA in ventral prefrontal white matter in major depressive disorder. J Psychiatr Res 65, 53–62 (2015).
Bongarzone, E. R., Howard, S. G., Schonmann, V. & Campagnoni, A. T. Identification of the dopamine D3 receptor in oligodendrocyte precursors: potential role in regulating differentiation and myelin formation. J Neurosci 18, 5344–5353 (1998).
Howard, S. et al. Postnatal localization and morphogenesis of cells expressing the dopaminergic D2 receptor gene in rat brain: expression in non-neuronal cells. J Comp Neurol 391, 87–98 (1998).
Rosin, C., Colombo, S., Calver, A. A., Bates, T. E. & Skaper, S. D. Dopamine D2 and D3 receptor agonists limit oligodendrocyte injury caused by glutamate oxidative stress and oxygen/glucose deprivation. Glia 52, 336–343 (2005).
Kluver, H. & Barrera, E. A method for the combined staining of cells and fibers in the nervous system. J Neuropathol Exp Neurol 12, 400–403 (1953).
Matsushima, G. K. & Morell, P. The neurotoxicant, cuprizone, as a model to study demyelination and remyelination in the central nervous system. Brain Pathol 11, 107–116 (2001).
Kipp, M., Clarner, T., Dang, J., Copray, S. & Beyer, C. The cuprizone animal model: new insights into an old story. Acta Neuropathol 118, 723–736 (2009).
Miller, R. H. Regulation of oligodendrocyte development in the vertebrate CNS. Prog Neurobiol 67, 451–467 (2002).
Pringle, N. P., Mudhar, H. S., Collarini, E. J. & Richardson, W. D. PDGF receptors in the rat CNS: during late neurogenesis, PDGF alpha-receptor expression appears to be restricted to glial cells of the oligodendrocyte lineage. Development 115, 535–551 (1992).
Bhat, R. V. et al. Expression of the APC tumor suppressor protein in oligodendroglia. Glia 17, 169–174 (1996).
Fancy, S. P. et al. Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Genes Dev 23, 1571–1585 (2009).
Fancy, S. P. et al. Overcoming remyelination failure in multiple sclerosis and other myelin disorders. Exp Neurol 225, 18–23 (2010).
Feigenson, K., Reid, M., See, J., Crenshaw, E. B. 3rd & Grinspan, J. B. Wnt signaling is sufficient to perturb oligodendrocyte maturation. Mol Cell Neurosci 42, 255–265 (2009).
Tawk, M. et al. Wnt/beta-catenin signaling is an essential and direct driver of myelin gene expression and myelinogenesis. J Neurosci 31, 3729–3742 (2011).
Dai, Z. M. et al. Stage-specific regulation of oligodendrocyte development by Wnt/β-catenin signaling. J Neurosci 34, 8467–8473 (2014).
**e, C., Li, Z., Zhang, G. X. & Guan, Y. Wnt signaling in remyelination in multiple sclerosis: friend or foe? Mol Neurobiol 49, 1117–1125 (2014).
Guo, F. et al. Canonical Wnt signaling in the oligodendroglial lineage–puzzles remain. Glia 63, 1671–1693 (2015).
Yoon, S., Choi, M. H., Chang, M. S. & Baik, J. H. Wnt5a-dopamine D2 receptor interactions regulate dopamine neuron development via extracellular signal-regulated kinase (ERK) activation. J Biol Chem 286, 15641–15651 (2011).
Yoon, S., Choi, M. H. & Baik, J. H. Wnt Ligand Binding to and Regulation of Dopamine D2 Receptors. In: Dopamine Receptor Technologies: Neuromethods vol. 96 (ed. Tiberi, M.) 65–78 (Springer, 2015).
Price, L. H. & Heninger, G. R. Lithium in the treatment of mood disorders. N Engl J Med 331, 591–598 (1994).
Klein, P. S. & Melton, D. A. A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci USA 93, 8455–8459 (1996).
Gould, T. D. & Manji, H. K. Glycogen synthase kinase-3: a putative molecular target for lithium mimetic drugs. Neuropsychopharmacology 30, 1223–1237 (2005).
Bartzokis, G. Neuroglialpharmacology: myelination as a shared mechanism of action of psychotropic treatments. Neuropharmacology 62, 2137–2153 (2012).
Rajkowska, G. et al. Morphometric evidence for neuronal and glial prefrontal cell pathology in major depression. Biol Psychiat 45, 1085–1098 (1999).
Tkachev, D. et al. Oligodendrocyte dysfunction in schizophrenia and bipolar disorder. Lancet 362, 798–805 (2003).
Katsel, P., Davis, K. L. & Haroutunian, V. Variations in myelin and oligodendrocyte-related gene expression across multiple brain regions in schizophrenia: a gene ontology study. Schizophr Res 79, 157–173 (2005).
Mahon, K., Burdick, K. E. & Szeszko, P. R. A role for white matter abnormalities in the pathophysiology of bipolar disorder. Neurosci Biobehav Rev 34, 533–554 (2010).
Tham, M. W., Woon, P. S., Sum, M. Y., Lee, T. S. & Sim, K. White matter abnormalities in major depression: evidence from post-mortem, neuroimaging and genetic studies. J Affect Disord 132, 26–36 (2011).
Banasr, M. et al. Chronic unpredictable stress decreases cell proliferation in the cerebral cortex of the adult rat. Biol Psychiat 62, 496–504 (2007).
Czéh, B. et al. Chronic social stress inhibits cell proliferation in the adult medial prefrontal cortex: hemispheric asymmetry and reversal by fluoxetine treatment. Neuropsychopharmacology 32, 1490–1503 (2007).
Miyata, S. et al. Association between chronic stress-induced structural abnormalities in Ranvier nodes and reduced oligodendrocyte activity in major depression. Sci Rep 6, 23084 (2016).
Brambilla, P. et al. Corpus callosum signal intensity in patients with bipolar and unipolar disorder. J. Neurol. Neurosurg. Psychiatry 75, 221–225 (2004).
Matsuo, K. et al. Anterior genu corpus callosum and impulsivity in suicidal patients with bipolar disorder. Neuroscience Letters 469, 75–80 (2010).
Bearden, C. E. et al. Map** corpus callosum morphology in twin pairs discordant for bipolar disorder. Cerebral Cortex 21, 2415–2424 (2011).
Benedetti, F. et al. Disruption of white matter integrity in bipolar depression as a possible structural marker of illness. Biological Psychiatry 69, 309–317 (2011).
Saxena, K. et al. A preliminary investigation of corpus callosum and anterior commissure aberrations in aggressive youth with bipolar disorders. Journal of Child and Adolescent Psychopharmacology 22, 112–119 (2012).
Benedetti, F. et al. Adverse childhood experiences influence white matter microstructure in patients with bipolar disorder. Psychol Med 44, 3069–3082 (2014).
Kumar, A. et al. Biophysical changes in normal-appearing white matter and subcortical nuclei in late-life major depression detected using magnetization transfer. Psychiatry Res 130, 131–40 (2004).
Kieseppä, T. et al. Major depressive disorder and white matter abnormalities: a diffusion tensor imaging study with tract-based spatial statistics. J Affect Disord 120, 240–4 (2010).
Chen, G. et al. Disorganization of white matter architecture in major depressive disorder: a meta-analysis of diffusion tensor imaging with tract-based spatial statistics. Sci Rep 6, 21825 (2016).
Miyata, S. et al. Plasma corticosterone activates SGK1 and induces morphological changes in oligodendrocytes in corpus callosum. PLoS One 6, e19859 (2011).
Howell, B. R. et al. Brain white matter microstructure alterations in adolescent rhesus monkeys exposed to early life stress: associations with high cortisol during infancy. Biol Mood Anxiety Disord. 3, 21 (2013).
Hemanth Kumar, B. S. et al. Demyelinating evidences in CMS rat model of depression: a DTI study at 7T. Neuroscience 275, 12–21 (2014).
Ye, F. et al. HDAC1 and HDAC2 regulate oligodendrocyte differentiation by disrupting the beta-catenin-TCF interaction. Nat Neurosci 12, 829–838 (2009).
Huang, S. M. et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461, 614–620 (2009).
Fancy, S. P. et al. Axin2 as regulatory and therapeutic target in newborn brain injury and remyelination. Nat Neurosci 14, 1009–1016 (2011).
Lee, H. K. et al. Daam2-PIP5K is a regulatory pathway for Wnt signaling and therapeutic target for remyelination in the CNS. Neuron 85, 1227–1243 (2015).
Lee, H. K. et al. Apcdd1 stimulates oligodendrocyte differentiation after white matter injury. Glia 63, 1840–1849 (2015).
Ortega, F. et al. Oligodendrogliogenic and neurogenic adult subependymal zone neural stem cells constitute distinct lineages and exhibit differential responsiveness to Wnt signalling. Nat Cell Biol 15, 602–613 (2013).
Fu, H. et al. A genome-wide screen for spatially restricted expression patterns identifies transcription factors that regulate glial development. J Neurosci 29, 11399–11408 (2009).
Fu, H., Kesari, S. & Cai, J. Tcf7l2 is tightly controlled during myelin formation. Cell Mol Neurobiol 32, 345–352 (2012).
Lürbke, A. et al. Limited TCF7L2 expression in MS lesions. PLoS One 8, e72822 (2013).
Puglisi-Allegra, S. & Cabib, S. Effects of defeat experiences on dopamine metabolism in different brain areas of the mouse. Aggressive Behavior 16, 271–284 (1990).
Klimek, V., Schenck, J. E., Han, H., Stockmeier, C. A. & Ordway, G. A. Dopaminergic abnormalities in amygdaloid nuclei in major depression: a postmortem study. Biol Psychiat 52, 740–748 (2002).
Leggio, G. M. et al. Dopamine D(3) receptor as a new pharmacological target for the treatment of depression. Eur J Pharmacol 719, 25–33 (2013).
Lee, P. R. & Fields, R. D. Regulation of myelin genes implicated in psychiatric disorders by functional activity in axons. Front Neuroanat 3, 4 (2009).
Narayan, S., Kass, K. E. & Thomas, E. A. Chronic haloperidol treatment results in a decrease in the expression of myelin/oligodendrocyte-related genes in the mouse brain. J Neurosci Res 85, 757–765 (2007).
Aiken, C. B. Pramipexole in psychiatry: a systematic review of the literature. J Clin Psychiat 68, 1230–1236 (2007).
Kamijima, K. et al. Aripiprazole augmentation to antidepressant therapy in Japanese patients with major depressive disorder: a randomized, double-blind, placebo-controlled study (ADMIRE study). J Affect Disord 151, 899–905 (2013).
Grünfeld, J. P. & Rossier, B. C. Lithium nephrotoxicity revisited. Nat Rev Nephrol 5, 270–276 (2009).
Gould, T. D. et al. Involvement of AMPA receptors in the antidepressant-like effects of lithium in the mouse tail suspension test and forced swim test. Neuropharmacology 54, 577–587 (2008).
Acknowledgements
We thank staff of Gyerim Experimental Animal Resource Center for animal care and technical assistance. This study was supported by the Brain Research Program (2013M3C7A1056101), Bio & Medical Technology Development Program (2013M3A9D5072550 and 2016M3A9D5A01952412), Mid-Career Researcher Program (2017R1A2B4008875), Science Research Center (2015R1A5A1009024) of National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, Future Planning of the Republic of Korea and by a Korea University grant.
Author information
Authors and Affiliations
Contributions
J.-H.B. conceived and supervised the study and wrote the manuscript. M.H.C., Y.R.Y., H.J.L., and S.Y. performed the molecular and cellular studies, the behavioral experiments and analyzed the results. J.E.N. and I.J.R. designed and performed the electron microscopic analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Choi, MH., Na, J.E., Yoon, Y.R. et al. Role of Dopamine D2 Receptor in Stress-Induced Myelin Loss. Sci Rep 7, 11654 (2017). https://doi.org/10.1038/s41598-017-10173-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-10173-9
- Springer Nature Limited
This article is cited by
-
Axo-glial interactions between midbrain dopamine neurons and oligodendrocyte lineage cells in the anterior corpus callosum
Brain Structure and Function (2023)
-
White matter integrity, duration of untreated psychosis, and antipsychotic treatment response in medication-naïve first-episode psychosis patients
Molecular Psychiatry (2021)
-
Oligodendrocyte lineage cells and depression
Molecular Psychiatry (2021)
-
Integrative systems and functional analyses reveal a role of dopaminergic signaling in myelin pathogenesis
Journal of Translational Medicine (2020)
-
Dopamine receptor density and white mater integrity: 18F-fallypride positron emission tomography and diffusion tensor imaging study in healthy and schizophrenia subjects
Brain Imaging and Behavior (2020)