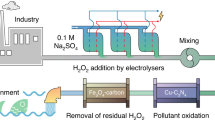
Abstract
Industrial hypersaline wastewaters contain diverse pollutants that harm the environment. Recovering clean water, alkali and acid from these wastewaters can promote circular economy and environmental protection. However, current electrochemical and advanced oxidation processes, which rely on hydroxyl radicals to degrade organic compounds, are inefficient and energy intensive. Here we report a flow-through redox-neutral electrochemical reactor (FRER) that effectively removes organic contaminants from hypersaline wastewaters via the chlorination–dehalogenation–hydroxylation route involving radical–radical cross-coupling. Bench-scale experiments demonstrate that the FRER achieves over 75% removal of total organic carbon across various compounds, and it maintains decontamination performance for over 360 h and continuously treats real hypersaline wastewaters for two months without corrosion. Integrating the FRER with electrodialysis reduces operating costs by 63.3% and CO2 emissions by 82.6% when compared with traditional multi-effect evaporation-crystallization techniques, placing our system at technology readiness levels of 7–8. The desalinated water, high-purity NaOH (>95%) and acid produced offset industrial production activities and thus support global sustainable development objectives.






Similar content being viewed by others
Data availability
All data generated for this study are available in the Article and Supplementary Information. Source experimental data are available from figshare repository at https://doi.org/10.6084/m9.figshare.25390759 (ref. 51).
References
Chung, M. G., Frank, K. A., Pokhrel, Y., Dietz, T. & Liu, J. Natural infrastructure in sustaining global urban freshwater ecosystem services. Nat. Sustain. 4, 1068–1075 (2021).
Zhang, X. et al. Managing nitrogen for sustainable development. Nature 528, 51–59 (2015).
Panagopoulos, A. & Giannika, V. Comparative techno-economic and environmental analysis of minimal liquid discharge (MLD) and zero liquid discharge (ZLD) desalination systems for seawater brine treatment and valorization. Sustain. Energy Technol. Assess. 53, 102477 (2022).
Menon, A. K., Haechler, I., Kaur, S., Lubner, S. & Prasher, R. S. Enhanced solar evaporation using a photo-thermal umbrella for wastewater management. Nat. Sustain. 3, 144–151 (2020).
Kumar, A., Phillips, K. R., Thiel, G. P., Schröder, U. & Lienhard, J. H. Direct electrosynthesis of sodium hydroxide and hydrochloric acid from brine streams. Nat. Catal. 2, 106–113 (2019).
Shehzad, M. A. et al. Shielded goethite catalyst that enables fast water dissociation in bipolar membranes. Nat. Commun. 12, 9 (2021).
Zhang, W. et al. Impact of chloride ions on UV/H2O2 and UV/persulfate advanced oxidation processes. Environ. Sci. Technol. 52, 7380–7389 (2018).
Chen, Y. et al. Tip-intensified interfacial microenvironment reconstruction promotes an electrocatalytic chlorine evolution reaction. ACS Catal. 12, 14376–14386 (2022).
Liu, J., Zhang, X. & Li, Y. Photoconversion of chlorinated saline wastewater DBPs in receiving seawater is overall a detoxification process. Environ. Sci. Technol. 51, 58–67 (2017).
Yang, T. et al. Efficient degradation of organoarsenic by UV/chlorine treatment: kinetics, mechanism, enhanced arsenic removal, and cytotoxicity. Environ. Sci. Technol. 55, 2037–2047 (2021).
Rao, U. et al. Structural dependence of reductive defluorination of linear PFAS compounds in a UV/electrochemical system. Environ. Sci. Technol. 54, 10668–10677 (2020).
Guo, K., Wu, Z., Chen, C. & Fang, J. UV/chlorine process: an efficient advanced oxidation process with multiple radicals and functions in water treatment. Acc. Chem. Res. 55, 286–297 (2022).
Choi, C. et al. Efficient electrocatalytic valorization of chlorinated organic water pollutant to ethylene. Nat. Nanotechnol. 18, 160–167 (2023).
Fang, J., Fu, Y. & Shang, C. The roles of reactive species in micropollutant degradation in the UV/free chlorine system. Environ. Sci. Technol. 48, 1859–1868 (2014).
Zhang, J., Zhang, G., Lan, H., Qu, J. & Liu, H. Synergetic hydroxyl radical oxidation with atomic hydrogen reduction lowers the organochlorine conversion barrier and potentiates effective contaminant mineralization. Environ. Sci. Technol. 55, 3296–3304 (2021).
Llorente, M. J., Nguyen, B. H., Kubiak, C. P. & Moeller, K. D. Paired electrolysis in the simultaneous production of synthetic intermediates and substrates. J. Am. Chem. Soc. 138, 15110–15113 (2016).
Min, Y. et al. Mimicking reductive dehalogenases for efficient electrocatalytic water dechlorination. Nat. Commun. 14, 5134 (2023).
Dwinandha, D., Zhang, B. & Fujii, M. Prediction of reaction mechanism for OH radical-mediated phenol oxidation using quantum chemical calculation. Chemosphere 291, 132763 (2022).
Atobe, M., Tateno, H. & Matsumura, Y. Applications of flow microreactors in electrosynthetic processes. Chem. Rev. 118, 4541–4572 (2018).
Finke, C. E. et al. Enhancing the activity of oxygen-evolution and chlorine-evolution electro-catalysts by atomic layer deposition of TiO2. Energy Environ. Sci. 12, 358–365 (2019).
Karlsson, R. K. B. & Cornell, A. Selectivity between oxygen and chlorine evolution in the chlor-alkali and chlorate processes. Chem. Rev. 116, 2982–3028 (2016).
Näslund, L.-Å. et al. The role of TiO2 do** on RuO2-coated electrodes for the water oxidation reaction. J. Phys. Chem. C 117, 6126–6135 (2013).
Jiao, Y., Jiang, H. & Chen, F. RuO2/TiO2/Pt ternary photocatalysts with epitaxial heterojunction and their application in CO oxidation. ACS Catal. 4, 2249–2257 (2014).
Morales-Guio, C. G. et al. Solar hydrogen production by amorphous silicon photocathodes coated with a magnetron sputter deposited Mo2C catalyst. J. Am. Chem. Soc. 137, 7035–7038 (2015).
Dong, C. et al. Fully exposed palladium cluster catalysts enable hydrogen production from nitrogen heterocycles. Nat. Catal. 5, 485–493 (2022).
**ong, H. et al. Engineering catalyst supports to stabilize PdOx two-dimensional rafts for water-tolerant methane oxidation. Nat. Catal. 4, 830–839 (2021).
Ghassemzadeh, L., Peckham, T. J., Weissbach, T., Luo, X. & Holdcroft, S. Selective formation of hydrogen and hydroxyl radicals by electron beam irradiation and their reactivity with perfluorosulfonated acid ionomer. J. Am. Chem. Soc. 135, 15923–15932 (2013).
Xu, B., Chen, Z., Zhang, G. & Wang, Y. On-demand atomic hydrogen provision by exposing electron-rich cobalt sites in an open-framework structure toward superior electrocatalytic nitrate conversion to dinitrogen. Environ. Sci. Technol. 56, 614–623 (2022).
Wang, J. et al. Ambient ammonia synthesis via palladium-catalyzed electrohydrogenation of dinitrogen at low overpotential. Nat. Commun. 9, 1795 (2018).
Kakade, B. A., Tamaki, T., Ohashi, H. & Yamaguchi, T. Highly active bimetallic PdPt and CoPt nanocrystals for methanol electro-oxidation. J. Phys. Chem. C 116, 7464–7470 (2012).
Zuo, K. et al. Electrified water treatment: fundamentals and roles of electrode materials. Nat. Rev. Mater. 8, 472–490 (2023).
Wang, H.-X., Toh, W. L., Tang, B. Y. & Surendranath, Y. Metal surfaces catalyse polarization-dependent hydride transfer from H2. Nat. Catal. 6, 351–362 (2023).
Sakamoto, H. et al. Photocatalytic dehalogenation of aromatic halides on Ta2O5-supported Pt–Pd bimetallic alloy nanoparticles activated by visible light. ACS Catal. 7, 5194–5201 (2017).
Walter, T. H. et al. Spin trap** in heterogeneous electron transfer processes. Can. J. Chem. 60, 1621–1636 (1982).
Barroso-Martínez, J. S. et al. Real-time detection of hydroxyl radical generated at operating electrodes via redox-active adduct formation using scanning electrochemical microscopy. J. Am. Chem. Soc. 144, 18896–18907 (2022).
Hong, J. et al. Metastable hexagonal close-packed palladium hydride in liquid cell TEM. Nature 603, 631–636 (2022).
Van Buren, J., Prasse, C., Marron, E. L., Skeel, B. & Sedlak, D. L. Ring-cleavage products produced during the initial phase of oxidative treatment of alkyl-substituted aromatic compounds. Environ. Sci. Technol. 54, 8352–8361 (2020).
Wang, Y., Wei, Y., Song, W., Chen, C. & Zhao, J. Photocatalytic hydrodehalogenation for the removal of halogenated aromatic contaminants. ChemCatChem 11, 258–268 (2019).
Yang, J. et al. CO2-mediated organocatalytic chlorine evolution under industrial conditions. Nature 617, 519–523 (2023).
Over, H. Surface chemistry of ruthenium dioxide in heterogeneous catalysis and electrocatalysis: from fundamental to applied research. Chem. Rev. 112, 3356–3426 (2012).
Lin, J. et al. Shielding effect enables fast ion transfer through nanoporous membrane for highly energy-efficient electrodialysis. Nat. Water 1, 725–735 (2023).
Butler, J. D., Parkerton, T. F., Redman, A. D., Letinski, D. J. & Cooper, K. R. Assessing aromatic-hydrocarbon toxicity to fish early life stages using passive-dosing methods and target-lipid and chemical-activity models. Environ. Sci. Technol. 50, 8305–8315 (2016).
Stinn, C. & Allanore, A. Selective sulfidation of metal compounds. Nature 602, 78–83 (2022).
Zabaniotou, A. Redesigning a bioenergy sector in EU in the transition to circular waste-based bioeconomy—a multidisciplinary review. J. Clean. Prod. 177, 197–206 (2018).
Andrew, R. M. Global CO2 emissions from cement production. Earth Syst. Sci. Data 10, 195–217 (2018).
Zhao, C., Dao, R., Wang, Y., Yao, J. & Li, H. A DFT investigation exploring the influence of lone electron pair on hyperfine structures of N-centered radicals. Chem. Phys. 517, 13–23 (2019).
Houriez, C., Ferre, N., Siri, D., Tordo, P. & Masella, M. Assessing the accuracy of a QM/MM//MD combined protocol to compute spectromagnetic properties of polyfunctional nitroxides in solution. Theor. Chem. Acc. 131, 1240 (2012).
Yamaguchi, M. DFT calculation of isotropic hyperfine coupling constants of spin adducts of 5,5-dimethyl-1-pyrroline-N-oxide in benzene and water. Comput. Theor. Chem. 1104, 24–31 (2017).
Cohen, A. J., Mori-Sánchez, P. & Yang, W. Challenges for density functional theory. Chem. Rev. 112, 289–320 (2012).
Gaussian 16 rev. C.01 (Gaussian, Inc., 2016).
Zhang, G. & Liu, H. Source data for “Redox-neutral electrochemical decontamination of hypersaline wastewater with high technology readiness level”. figshare https://doi.org/10.6084/m9.figshare.25390759 (2024).
Acknowledgements
We acknowledge the National Natural Science Foundation of China (grants 52221004 to H.L., 22022606 to G. Zhang), X. Li and J. Lin from Bei**g Capital Eco-Environment Protection Group Co., Ltd. for supporting this project and M.Z. Li from Huaibei GO-ON Power Co., Ltd. for supporting our field experiment. We thank Q. Liu from the Department of Chemistry, Tsinghua University, for discussions on the mechanisms of catalysis and electrochemical measurements. We also thank S. Qu from Bei**g Institute of Technology for supporting LCA analysis. We thank A.L. Chun of Science Storylab for critically reading and editing the paper.
Author information
Authors and Affiliations
Contributions
H.L. directed the project. G. Zhang designed the experiments. G. Zhang and Y.L. synthesized the materials and performed the bench-scale degradation and electrochemical experiments. G. Zhang and C.Z. analysed results of in situ EPR experiments. J.G., Y.L. and Q.C. performed the on-site field experiments. G. Zhou carried out the DFT calculations. W.-J.F. and Y.L. collected and analysed the HRMS data. Q.Z. and Y.L. conducted the LCA analysis. G. Zhang, Y.S., Y.L. and H.L. co-wrote the Article. H.L., Q.J., F.X. and J.Q. revised the Article. All authors discussed the results and commented on the Article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Yang Yang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–39, Discussion and Tables 1–8.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, G., Li, Y., Zhao, C. et al. Redox-neutral electrochemical decontamination of hypersaline wastewater with high technology readiness level. Nat. Nanotechnol. (2024). https://doi.org/10.1038/s41565-024-01669-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41565-024-01669-3
- Springer Nature Limited