Abstract
While disgust originates in the hard-wired mammalian distaste response, the conscious experience of disgust in humans strongly depends on subjective appraisal and may even extend to socio-moral contexts. Here, in a series of studies, we combined functional magnetic resonance imaging with machine-learning-based predictive modelling to establish a comprehensive neurobiological model of subjective disgust. The developed neurofunctional signature accurately predicted momentary self-reported subjective disgust across discovery (n = 78) and pre-registered validation (n = 30) cohorts and generalized across core disgust (n = 34 and n = 26), gustatory distaste (n = 30) and socio-moral (unfair offers; n = 43) contexts. Disgust experience was encoded in distributed cortical and subcortical systems, and exhibited distinct and shared neural representations with subjective fear or negative affect in interoceptive-emotional awareness and conscious appraisal systems, while the signatures most accurately predicted the respective target experience. We provide an accurate functional magnetic resonance imaging signature for disgust with a high potential to resolve ongoing evolutionary debates.








Similar content being viewed by others
Data availability
fMRI data used to train and validate the signature are available via figshare at https://figshare.com/articles/dataset/Discovery_dataset_disgust/22827974 (ref. 123) (study 1) and https://figshare.com/articles/dataset/validation_dataset_disgust/22841117 (ref. 124) (study 2). fMRI data of the modified disgust induction task are available via figshare at https://figshare.com/articles/dataset/Dataset_of_the_modified_disgust_induction_experiment/25284895 (ref. 125) (study 3). The data of study 4 were provided by the authors of a previous study54. The data of study 5 are from a previous study39 and are available via NeuroVault at https://neurovault.org/collections/1964 (ref. 126). The data of study 6 are from a previous study18 and are available via figshare at https://figshare.com/articles/dataset/Subjective_fear_dataset/13271102?file=25556276 (ref. 127). The data from the gustatory experiment (study 7) as well as the ultimatum game experiment (study 8) are from two independent ongoing projects of our research team and are available from the corresponding author upon request. The data of pain empathy task (study 9) from our previous study49 are available via figshare at https://figshare.com/articles/dataset/Vicarious_pain_dataset/11994498 (ref. 128). Data from the negative/neutral experiment (study 10) are from an ongoing project from our team57 and available upon request. The data of study 11 were provided by the authors of a previous study58 (note that we applied for a subset of randomly selected data from 150 participants (with gender ratio balanced) from the authors). The VIDS and the thresholded statistical maps are available via figshare at https://figshare.com/articles/dataset/Brain_models_and_maps_zip/22827950 (ref. 129). Data from the functional characterizations of the Brainnetome Atlas based on the BrainMap database are available at https://atlas.brainnetome.org/bnatlas.html (ref. 108).
Code availability
Data were analysed using CANLab neuroimaging analysis tools available via GitHub at https://github.com/canlab (ref. 130) and https://github.com/ganxianyang/fMRI-studies/tree/main/Subjective_disgust_experience_signature (ref. 131). Glass brain was drawn using the open-source Python package Nilearn132.
References
Rozin, P., Haidt, J. & Fincher, K. From oral to moral. Science 323, 1179–1180 (2009).
Rozin, P. & Fallon, A. E. A perspective on disgust. Psychol. Rev. 94, 23–41 (1987).
Chapman, H. A., Kim, D. A., Susskind, J. M. & Anderson, A. K. In bad taste: evidence for the oral origins of moral disgust. Science 323, 1222–1226 (2009).
Rozin, P. & Haidt, J. The domains of disgust and their origins: contrasting biological and cultural evolutionary accounts. Trends Cogn. Sci. 17, 367–368 (2013).
**e, Z. et al. The gut-to-brain axis for toxin-induced defensive responses. Cell 185, 4298–4316.e4221 (2022).
Dolensek, N., Gehrlach, D. A., Klein, A. S. & Gogolla, N. Facial expressions of emotion states and their neuronal correlates in mice. Science 368, 89–94 (2020).
Vicario, C. M., Rafal, R. D., Martino, D. & Avenanti, A. Core, social and moral disgust are bounded: a review on behavioral and neural bases of repugnance in clinical disorders. Neurosci. Biobehav. Rev. 80, 185–200 (2017).
Gan, X. et al. Common and distinct neurofunctional representations of core and social disgust in the brain: coordinate-based and network meta-analyses. Neurosci. Biobehav. Rev. 135, 104553 (2022).
Amoroso, C. R. et al. Disgust theory through the lens of psychiatric medicine. Clin. Psychol. Sci. 8, 3–24 (2019).
Jones, D. The depths of disgust. Nature 447, 768–771 (2007).
Tybur, J. M., Lieberman, D., Kurzban, R. & DeScioli, P. Disgust: evolved function and structure. Psychol. Rev. 120, 65–84 (2013).
Tybur, J. M., Lieberman, D. & Griskevicius, V. Microbes, mating, and morality: individual differences in three functional domains of disgust. J. Pers. Soc. Psychol. 97, 103–122 (2009).
Haidt, J., McCauley, C. & Rozin, P. Individual differences in sensitivity to disgust: a scale sampling seven domains of disgust elicitors. Pers. Individ. Differ. 16, 701–713 (1994).
Chapman, H. A. & Anderson, A. K. Things rank and gross in nature: a review and synthesis of moral disgust. Psychol. Bull. 139, 300–327 (2013).
Zhang, C., Vincelette, L. K., Reimann, F. & Liberles, S. D. A brainstem circuit for nausea suppression. Cell Rep. 39, 110953 (2022).
Zhang, C. et al. Area postrema cell types that mediate nausea-associated behaviors. Neuron 109, 461–472.e465 (2021).
Becker, B. et al. Fear processing and social networking in the absence of a functional amygdala. Biol. Psychiatry 72, 70–77 (2012).
Zhou, F. et al. A distributed fMRI-based signature for the subjective experience of fear. Nat. Commun. 12, 6643 (2021).
Zhou, F. et al. Human extinction learning is accelerated by an angiotensin antagonist via ventromedial prefrontal cortex and its connections with basolateral amygdala. Biol. Psychiatry 86, 910–920 (2019).
Taschereau-Dumouchel, V., Kawato, M. & Lau, H. Multivoxel pattern analysis reveals dissociations between subjective fear and its physiological correlates. Mol. Psychiatry 25, 2342–2354 (2020).
Reddan, M. C., Wager, T. D. & Schiller, D. Attenuating neural threat expression with imagination. Neuron 100, 994–1005.e1004 (2018).
Chapman, H. A. & Anderson, A. K. Understanding disgust. Ann. N. Y. Acad. Sci. 1251, 62–76 (2012).
Wicker, B. et al. Both of us disgusted in my insula: the common neural basis of seeing and feeling disgust. Neuron 40, 655–664 (2003).
Stark, R. et al. Erotic and disgust-inducing pictures—differences in the hemodynamic responses of the brain. Biol. Psychol. 70, 19–29 (2005).
Schienle, A., Übel, S., Schöngaßner, F., Ille, R. & Scharmüller, W. Disgust regulation via placebo: an fMRI study. Soc. Cogn. Affect. Neurosci. 9, 985–990 (2014).
Baumann, O. & Mattingley, J. B. Functional topography of primary emotion processing in the human cerebellum. NeuroImage 61, 805–811 (2012).
Schienle, A., Höfler, C., Keck, T. & Wabnegger, A. Neural underpinnings of perception and experience of disgust in individuals with a reduced sense of smell: an fMRI study. Neuropsychologia 141, 107411 (2020).
Viol, K. et al. Erroneously disgusted: fMRI study supports disgust-related neural reuse in obsessive-compulsive disorder (OCD). Front. Behav. Neurosci. 13, 81 (2019).
Li, J. et al. Common and dissociable contributions of alexithymia and autism to domain-specific interoceptive dysregulations: a dimensional neuroimaging approach. Psychother. Psychosom. 88, 187–189 (2019).
Yao, S. et al. Oxytocin modulates attention switching between interoceptive signals and external social cues. Neuropsychopharmacology 43, 294–301 (2018).
Ferraro, S. et al. The central autonomic system revisited—convergent evidence for a regulatory role of the insular and midcingulate cortex from neuroimaging meta-analyses. Neurosci. Biobehav. Rev. 142, 104915 (2022).
Levine, S. M., Wackerle, A., Rupprecht, R. & Schwarzbach, J. V. The neural representation of an individualized relational affective space. Neuropsychologia 120, 35–42 (2018).
Levine, S. M. & Schwarzbach, J. V. Individualizing representational similarity analysis. Front. Psychiatry 12, 729457 (2021).
Bhikram, T., Abi-Jaoude, E. & Sandor, P. OCD: obsessive–compulsive … disgust? The role of disgust in obsessive–compulsive disorder. J. Psychiatry Neurosci. 42, 300 (2017).
Shapira, N. A. et al. Brain activation by disgust-inducing pictures in obsessive-compulsive disorder. Biol. Psychiatry 54, 751–756 (2003).
Schienle, A., Schäfer, A., Hermann, A. & Vaitl, D. Binge-eating disorder: reward sensitivity and brain activation to images of food. Biol. Psychiatry 65, 654–661 (2009).
Benuzzi, F., Lui, F., Duzzi, D., Nichelli, P. & Porro, C. Does it look painful or disgusting? Ask your parietal and cingulate cortex. J. Neurosci. 28, 923–931 (2008).
Stark, R. et al. Hemodynamic brain correlates of disgust and fear ratings. Neuroimage 37, 663–673 (2007).
Chang, L. J., Gianaros, P. J., Manuck, S. B., Krishnan, A. & Wager, T. D. A sensitive and specific neural signature for picture-induced negative affect. PLoS Biol. 13, e1002180 (2015).
Kragel, P. A., Koban, L., Barrett, L. F. & Wager, T. D. Representation, pattern information, and brain signatures: from neurons to neuroimaging. Neuron 99, 257–273 (2018).
Haxby, J. V. Multivariate pattern analysis of fMRI: the early beginnings. NeuroImage 62, 852–855 (2012).
Kragel, P. A. & LaBar, K. S. Multivariate neural biomarkers of emotional states are categorically distinct. Soc. Cogn. Affect. Neurosci. 10, 1437–1448 (2015).
Čeko, M., Kragel, P. A., Woo, C.-W., López-Solà, M. & Wager, T. D. Common and stimulus-type-specific brain representations of negative affect. Nat. Neurosci. 25, 760–770 (2022).
Wager, T. D., Krishnan, A. & Hitchcock, E. in The Nature of Emotion. Fundamental Questions (eds Fox, A. S. et al.) 112–118 (Oxford Univ. Press, 2018).
Weaverdyck, M., Lieberman, M. & Parkinson, C. Multivoxel pattern analysis in fMRI: a practical introduction for social and affective neuroscientists. Soc. Cogn. Affect. Neurosci. 15, 487–509 (2020).
Kragel, P. A. & LaBar, K. S. Decoding the nature of emotion in the brain. Trends Cogn. Sci. 20, 444–455 (2016).
Haynes, J.-D. A primer on pattern-based approaches to fMRI: principles, pitfalls, and perspectives. Neuron 87, 257–270 (2015).
Woo, C.-W., Chang, L. J., Lindquist, M. A. & Wager, T. D. Building better biomarkers: brain models in translational neuroimaging. Nat. Neurosci. 20, 365–377 (2017).
Zhou, F. et al. Empathic pain evoked by sensory and emotional-communicative cues share common and process-specific neural representations. eLife 9, e56929 (2020).
LeDoux, J. E. & Brown, R. A higher-order theory of emotional consciousness. Proc. Natl Acad. Sci. USA 114, E2016–E2025 (2017).
Barrett, L. F. The theory of constructed emotion: an active inference account of interoception and categorization. Soc. Cogn. Affect. Neurosci. 12, 1–23 (2017).
Saarimäki, H. et al. Discrete neural signatures of basic emotions. Cereb. Cortex 26, 2563–2573 (2015).
Corradi-Dell’Acqua, C., Tusche, A., Vuilleumier, P. & Singer, T. Cross-modal representations of first-hand and vicarious pain, disgust and fairness in insular and cingulate cortex. Nat. Commun. 7, 10904 (2016).
Chen, S. et al. Functional decoupling of emotion co** network subsides automatic emotion regulation by implementation intention. Neural Plast. 2021, 6639739 (2021).
Haufe, S. et al. On the interpretation of weight vectors of linear models in multivariate neuroimaging. NeuroImage 87, 96–110 (2014).
Peelen, M. V. & Downing, P. E. Testing cognitive theories with multivariate pattern analysis of neuroimaging data. Nat. Hum. Behav. 7, 1430–1441 (2023).
Xu, T. et al. Angiotensin antagonist inhibits preferential negative memory encoding via decreasing hippocampus activation and its coupling with the amygdala. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 7, 970–978 (2022).
Fastenrath, M. et al. Human cerebellum and corticocerebellar connections involved in emotional memory enhancement. Proc. Natl Acad. Sci. USA 119, e2204900119 (2022).
Haberkamp, A., Glombiewski, J. A., Schmidt, F. & Barke, A. The DIsgust-RelaTed-Images (DIRTI) database: validation of a novel standardized set of disgust pictures. Behav. Res. Ther. 89, 86–94 (2017).
Wager, T. D. et al. An fMRI-based neurologic signature of physical pain. N. Engl. J. Med. 368, 1388–1397 (2013).
Kragel, P. A., Reddan, M. C., LaBar, K. S. & Wager, T. D. Emotion schemas are embedded in the human visual system. Sci. Adv. 5, eaaw4358 (2019).
Kohoutová, L. et al. Toward a unified framework for interpreting machine-learning models in neuroimaging. Nat. Protoc. 15, 1399–1435 (2020).
LeDoux, J. E. & Pine, D. S. Using neuroscience to help understand fear and anxiety: a two-system framework. Am. J. Psychiatry 173, 1083–1093 (2016).
Fan, L. et al. The Human Brainnetome Atlas: a new brain atlas based on connectional architecture. Cereb. Cortex 26, 3508–3526 (2016).
Lee, J.-J. et al. A neuroimaging biomarker for sustained experimental and clinical pain. Nat. Med. 27, 174–182 (2021).
Damasio, A. & Carvalho, G. B. The nature of feelings: evolutionary and neurobiological origins. Nat. Rev. Neurosci. 14, 143–152 (2013).
Kirby, L. A. J. & Robinson, J. L. Affective map**: an activation likelihood estimation (ALE) meta-analysis. Brain Cogn. 118, 137–148 (2017).
Vytal, K. & Hamann, S. Neuroimaging support for discrete neural correlates of basic emotions: a voxel-based meta-analysis. J. Cogn. Neurosci. 22, 2864–2885 (2010).
Costafreda, S. G., Brammer, M. J., David, A. S. & Fu, C. H. Y. Predictors of amygdala activation during the processing of emotional stimuli: a meta-analysis of 385 PET and fMRI studies. Brain Res. Rev. 58, 57–70 (2008).
Satpute, A. B. & Lindquist, K. A. The default mode network’s role in discrete emotion. Trends Cogn. Sci. 23, 851–864 (2019).
Touroutoglou, A., Lindquist, K. A., Dickerson, B. C. & Barrett, L. F. Intrinsic connectivity in the human brain does not reveal networks for ‘basic’ emotions. Soc. Cogn. Affect. Neurosci. 10, 1257–1265 (2015).
Sanfey, A. G., Rilling, J. K., Aronson, J. A., Nystrom, L. E. & Cohen, J. D. The neural basis of economic decision-making in the ultimatum game. Science 300, 1755–1758 (2003).
Han, X. et al. Effect sizes and test–retest reliability of the fMRI-based neurologic pain signature. NeuroImage 247, 118844 (2022).
Poldrack, R. A. et al. Scanning the horizon: towards transparent and reproducible neuroimaging research. Nat. Rev. Neurosci. 18, 115–126 (2017).
Reddan, M. C., Lindquist, M. A. & Wager, T. D. Effect size estimation in neuroimaging. JAMA Psychiatry 74, 207–208 (2017).
Taschereau-Dumouchel, V., Michel, M., Lau, H., Hofmann, S. G. & LeDoux, J. E. Putting the “mental” back in “mental disorders”: a perspective from research on fear and anxiety. Mol. Psychiatry 27, 1322–1330 (2022).
Kyzar, E. J. & Denfield, G. H. Taking subjectivity seriously: towards a unification of phenomenology, psychiatry, and neuroscience. Mol. Psychiatry 28, 10–16 (2023).
Pujol, J. et al. Map** the sequence of brain events in response to disgusting food. Hum. Brain Mapp. 39, 369–380 (2018).
Qi, S. et al. How cognitive and reactive fear circuits optimize escape decisions in humans. Proc. Natl Acad. Sci. USA 115, 3186–3191 (2018).
Lindquist, K. A., Wager, T. D., Kober, H., Bliss-Moreau, E. & Barrett, L. F. The brain basis of emotion: a meta-analytic review. Behav. Brain Sci. 35, 121–143 (2012).
Clore, G. L. & Ortony, A. in Handbook of Emotions 3rd ed. (eds Lewis, M., Haviland-Jones, J. M. & Barrett, L. F.) 628–642 (The Guilford Press, 2008).
Wager, T. D. et al. A Bayesian model of category-specific emotional brain responses. PLoS Comput. Biol. 11, e1004066 (2015).
Insel, T. et al. Research Domain Criteria (RDoC): toward a new classification framework for research on mental disorders. Am. J. Psychiatry 167, 748–751 (2010).
Steiner, J. E. The gustofacial response: observation on normal and anencephalic newborn infants. in Fourth Symposium on Oral Sensation and Perception (ed. Bosma, J. F.) 254–278 (US Department of Health, Education and Welfare, 1973).
Sharvit, G., Lin, E., Vuilleumier, P. & Corradi-Dell’Acqua, C. Does inappropriate behavior hurt or stink? The interplay between neural representations of somatic experiences and moral decisions. Sci. Adv. 6, eaat4390 (2020).
Zhou, F. et al. Capturing dynamic fear experiences in naturalistic contexts: an ecologically valid fMRI signature integrating brain activation and connectivity. Preprint at bioRxiv https://doi.org/10.1101/2023.08.18.553808 (2023).
Lamm, C., Decety, J. & Singer, T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. NeuroImage 54, 2492–2502 (2011).
Russell, P. S. & Giner-Sorolla, R. Bodily moral disgust: what it is, how it is different from anger, and why it is an unreasoned emotion. Psychol. Bull. 139, 328–351 (2013).
Henrich, J. et al. Foundations of Human Sociality: Economic Experiments and Ethnographic Evidence from Fifteen Small-Scale Societies (Oxford Univ. Press, 2004).
Sheskin, M. in Interdisciplinary Perspectives on Fairness, Equity, and Justice (eds Li, M. & Tracer, D. P.) 33–49 (Springer, 2017).
Finn, E. S., Poldrack, R. A. & Shine, J. M. Functional neuroimaging as a catalyst for integrated neuroscience. Nature 623, 263–273 (2023).
Zhang, R. et al. Angiotensin II regulates the neural expression of subjective fear in humans: a precision pharmaco-neuroimaging approach. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 8, 262–270 (2023).
Marchewka, A., Żurawski, Ł., Jednoróg, K. & Grabowska, A. The Nencki Affective Picture System (NAPS): introduction to a novel, standardized, wide-range, high-quality, realistic picture database. Behav. Res. Methods 46, 596–610 (2014).
Lang, P., Bradley, M. & Cuthbert, B. International Affective Picture System (IAPS): Affective Ratings of Pictures and Instruction Manual. Technical Report A-8 (University of Florida, 2008).
SPM12. SPM https://www.fil.ion.ucl.ac.uk/spm/software/spm12/ (2020).
Woo, C.-W. et al. Separate neural representations for physical pain and social rejection. Nat. Commun. 5, 5380 (2014).
Losin, E. A. R. et al. Neural and sociocultural mediators of ethnic differences in pain. Nat. Hum. Behav. 4, 517–530 (2020).
Op de Beeck, H. P. Against hyperacuity in brain reading: spatial smoothing does not hurt multivariate fMRI analyses? NeuroImage 49, 1943–1948 (2010).
Shmuel, A., Chaimow, D., Raddatz, G., Ugurbil, K. & Yacoub, E. Mechanisms underlying decoding at 7 T: ocular dominance columns, broad structures, and macroscopic blood vessels in V1 convey information on the stimulated eye. NeuroImage 49, 1957–1964 (2010).
CanlabCore. GitHub https://github.com/canlab/CanlabCore (2014).
MPI Biol. Cybernetics. The Spider. http://people.kyb.tuebingen.mpg.de/spider (2006).
Poldrack, R. A., Huckins, G. & Varoquaux, G. Establishment of best practices for evidence for prediction: a review. JAMA Psychiatry 77, 534–540 (2020).
Zhou, F. et al. Shifted balance of dorsal versus ventral striatal communication with frontal reward and regulatory regions in cannabis-dependent males. Hum. Brain Mapp. 39, 5062–5073 (2018).
Atlas, L. Y., Lindquist, M. A., Bolger, N. & Wager, T. D. Brain mediators of the effects of noxious heat on pain. Pain 155, 1632–1648 (2014).
Büchel, C. et al. Dissociable neural responses related to pain intensity, stimulus intensity, and stimulus awareness within the anterior cingulate cortex: a parametric single-trial laser functional magnetic resonance imaging study. J. Neurosci. 22, 970–976 (2002).
Courville, T. & Thompson, B. Use of structure coefficients in published multiple regression articles: β is not enough. Educ. Psychol. Meas. 61, 229–248 (2001).
Thompson, B. & Borrello, G. M. The importance of structure coefficients in regression research. Educ. Psychol. Meas. 45, 203–209 (1985).
Brainnetome Atlas. https://atlas.brainnetome.org/bnatlas.html (2023).
BrainMap Taxonomy. brainmap.org http://www.brainmap.org/taxonomy/ (2022).
Yarkoni, T., Poldrack, R. A., Nichols, T. E., Van Essen, D. C. & Wager, T. D. Large-scale automated synthesis of human functional neuroimaging data. Nat. Methods 8, 665–670 (2011).
Salo, T. et al. NiMARE: neuroimaging meta-analysis research environment. Aperture Neuro 3, 1–32 (2023).
Schurz, M. et al. Toward a hierarchical model of social cognition: a neuroimaging meta-analysis and integrative review of empathy and theory of mind. Psychol. Bull. 147, 293–327 (2021).
Maliske, L. Z., Schurz, M. & Kanske, P. Interactions within the social brain: co-activation and connectivity among networks enabling empathy and theory of mind. Neurosci. Biobehav. Rev. 147, 105080 (2023).
Pankey, B. S. et al. Extended functional connectivity of convergent structural alterations among individuals with PTSD: a neuroimaging meta-analysis. Behav. Brain Funct. 18, 9 (2022).
BrainStat. GitHub https://github.com/MICA-MNI/BrainStat (2022).
Larivière, S. et al. BrainStat: a toolbox for brain-wide statistics and multimodal feature associations. NeuroImage 266, 119807 (2023).
Yeo, B. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
MediationToolbox. GitHub https://github.com/canlab/MediationToolbox (2014).
Woo, C.-W. et al. Quantifying cerebral contributions to pain beyond nociception. Nat. Commun. 8, 14211 (2017).
Eskine, K. J., Kacinik, N. A. & Prinz, J. J. A bad taste in the mouth: gustatory disgust influences moral judgment. Psychol. Sci. 22, 295–299 (2011).
Jabbi, M., Swart, M. & Keysers, C. Empathy for positive and negative emotions in the gustatory cortex. NeuroImage 34, 1744–1753 (2007).
Jabbi, M., Bastiaansen, J. & Keysers, C. A common anterior insula representation of disgust observation, experience and imagination shows divergent functional connectivity pathways. PLoS ONE 3, e2939 (2008).
Disgust discovery cohort dataset. figshare https://figshare.com/articles/dataset/Discovery_dataset_disgust/22827974 (2023).
Disgust validation cohort dataset. figshare https://figshare.com/articles/dataset/validation_dataset_disgust/22841117 (2023).
Dataset of the modified disgust induction experiment. figshare https://figshare.com/articles/dataset/Dataset_of_the_modified_disgust_induction_experiment/25284895 (2024).
PINES holdout test dataset. NeuroVault https://neurovault.org/collections/1964 (2015).
VIFS discovery cohort dataset. figshare https://figshare.com/articles/dataset/Subjective_fear_dataset/13271102?file=25556276 (2021).
Pain empathy task dataset. figshare https://figshare.com/articles/dataset/Vicarious_pain_dataset/11994498 (2020).
The VIDS and thresholded stastistical maps. figshare https://figshare.com/articles/dataset/Brain_models_and_maps_zip/22827950 (2023).
Cognitive and Affective Neuroscience Laboratory, CANLab. GitHub https://github.com/canlab (2014).
Subjective_disgust_experience_signature. GitHub https://github.com/ganxianyang/fMRI-studies/tree/main/Subjective_disgust_experience_signature (2023).
Abraham, A. et al. Machine learning for neuroimaging with scikit-learn. Front. Neuroinform. 8, 14 (2014).
Acknowledgements
We thank D. Coynel and D. J.-F. de Quervain as well as S. Chen for sharing their data. We also thank the CANLab for providing the PINES signature and the PINES holdout dataset. In addition, we thank X. Tian and Q. **e (both majored in pharmacy) who provided us with the essential background knowledge on how to calculate the concentration of different gustatory liquids and the appropriate medical-grade equipment for the gustatory experiment. Any opinions, findings, conclusions or recommendations expressed in this publication do not reflect the views of the Government of the Hong Kong Special Administrative Region or the Innovation and Technology Commission. This work was partly supported by the China MOST2030 Brain Project (grant no. 2022ZD0208500) to D.Y., the National Natural Science Foundation of China (grant nos. 32250610208 and 82271583 to B.B., and 32300862 to F.Z.), National Key Research and Development Program of China (grant no. 2018YFA0701400) to B.B., the Fundamental Research Funds for the Central Universities (SWU2309733) to F.Z. and a start-up grant from the University of Hong Kong to B.B. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
X.G., F.Z. and B.B. conceived and designed the experiment. X.G., F.Z. and B.B. analysed the data and were responsible for interpretation of data. T.X., X.L., R.Z., Z.Z., X.Y., X.Z., F.Y., J.L. and R.C. provided important suggestions during formal analysis. X.G., R.Z., T.X. and L.W. conducted the experiment. X.G. and R.C. were responsible for visualization. X.G. and B.B. drafted the paper; F.Z., J.Y. and D.Y. provided feedback and revised the paper. B.B. supervised the project and acquired the funding. All authors meet the four ICMJE authorship criteria and were responsible for revising the paper, for approving the final version for publication and for accuracy and integrity of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interevsts.
Peer review
Peer review information
Nature Human Behaviour thanks Corrado Corradi-Dell’Acqua, Peter de Jong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1
The distribution of subjective disgust ratings for each category (that is, animal, human and scene), respectively.
Extended Data Fig. 2 The spatial topography of the unthresholded patterns in some anatomical regions of interest (ROIs).
This panel illustrates the VIDS pattern thresholded using a 10,000-sample bootstrap procedure at q < 0.05, FDR corrected. Inserts show the spatial topography of the unthresholded patterns in some anatomical ROIs. ACC=anterior cingulate cortex, Amy=amygdala, Ins=insula, PAG=periaqueductal gray, Put=putamen, SFG=superior frontal gyrus, Thal=thalamus.
Extended Data Fig. 3 VIDS pattern response without occipital lobe.
a, The predicted disgust ratings compared to the true ratings for the cross-validated discovery cohort (n = 78). b, The predicted disgust ratings compared to the true ratings for the independent validation cohort (n = 30). Accuracies reflect forced-choice comparisons. P values were based on binomial tests, two-sided (uncorrected). r indicates the Pearson correlation coefficient between predicted and true ratings. Error bars reflect the s.e.m.
Extended Data Fig. 4 The VIDS tracks disgust experience independent of the motor responses.
a, The modified disgust induction paradigm included a jittered period between the stimulus and the rating, and the rating numbers were provided in a randomized order. This allowed to better uncouple the motor and emotional response. Of note, schematic figures were used for display purpose only to avoid copyright issues and were not included in the original stimulus set. b, Predicted disgust experience (subjective ratings; mean ± s.e.m.) compared to actual disgust ratings using data from the modified disgust induction task (acquired in n = 34 individuals). Accuracy provided for forced-choice comparisons. P values were based on two-sided independent binomial tests (uncorrected). r indicates Pearson correlation coefficient between predicted and true ratings. c, Averaged peristimulus plot (mean ± s.e.m.) of the VIDS response using data from the modified disgust induction task at every repetition time (TR; 2 s) for each disgust intensity rating separately. pic, picture. Error bars and shaded regions indicate the s.e.m.
Extended Data Fig. 5 Subjective experience of disgust is associated with and predicted by distributed brain regions.
a, The univariate parametric effects of disgust ratings. b, Multivariate patterns trained on individual subjects and depicts brain regions consistently predictive of subjective disgust across participants. c, Thresholded transformed ‘activation patterns’ from within-subject disgust-predictive patterns. d, Overlap** (that is, from a conjunction analysis) brain regions between (b and c). Hot color indicates positive associations (a and c) or weights (b) whereas cold color indicates negative associations (a and c) or weights (b).
Extended Data Fig. 6 Neurosynth functional decoding of the unthresholded VIDS.
Here, the 100 most strongly correlated terms were displayed, with a larger font size indicating a larger Pearson correlation coefficient.
Extended Data Fig. 7 Predictions of models trained on discovery cohort on validation cohort.
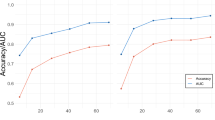
a,b, Brain regions that can significantly predict subjective disgust revealed by searchlight- (Panel a) and parcellation-based (Panel b) analyses, respectively. Statistical significance was evaluated by prediction−outcome correlation (Pearson; two-sided; P < 0.001, uncorrected). Histograms: Predictions (correlations) from searchlights (Panel a) and parcellations (Panel b), respectively. The orange line indicates the prediction-outcome correlation from VIDS. c,d, Predictions (mean ± s.e.m.) from insula- (Panel c) and amygdala-based (Panel d) prediction analyses, respectively. Error bar indicates the s.e.m.; r indicates overall (between- and within-subjects; that is, n = 149 pairs) prediction-outcome Pearson correlation coefficient. e, The information about subjective experience of disgust is distributed across multiple systems. Model performance was evaluated as increasing numbers of voxels/features (x-axis) were used to predict subjective disgust in different regions of interest including the entire brain (black), consciousness network, subcortical regions or large-scale cerebral networks. The y-axis denotes the prediction-outcome correlation. Colored dots indicate the mean correlation coefficients, solid lines indicate the mean parametric fit and shaded regions indicate the s.d.
Extended Data Fig. 8 Physical pain empathy decoder predicts disgust experience.
a, The physical pain empathy decoder could predict high versus low (shown as forced-choice classification accuracy, P value and Cohen’s d) and high versus moderate disgust, nonetheless, it fails to discriminate moderate versus low disgust in the discovery cohort. b, The classification results of the physical pain empathy decoder in the validation cohort, which replicates the findings as shown in (a). P values were based on binomial tests, two-sided (uncorrected).
Extended Data Fig. 9 Comparing VIDS, PINES and VIFS.
a, River plots displaying spatial similarity (calculated as cosine similarity) between the stable decoding maps and the subcortical as well as consciousness network. Ribbons are normalized by the max cosine similarity across networks. Stable decoding models were thresholded at FDR q < 0.05 and positive voxels were retained only for similarity calculation and interpretation. Ribbon locations in relation to the boxes are arbitrary. Pie charts show relative contributions of each model to each network (that is, percentage of voxels with highest cosine similarity for each map). b, The multilevel mediation analytic results showing that VIDS response partially mediates the association between VIFS response and subjective disgust rating in both discovery and validation cohorts. c, The VIFS response plays a partial mediation role in the effect of VIDS response on the disgust rating. In b and c, the mediation analysis examines whether the observed covariance between the independent variable (X) and the dependent variable (Y) can be explained by the third variable (M, also mediator), details see Methods section. Two-sided P values are based on bootstrap tests with 10,000 samples, uncorrected.
Extended Data Fig. 10 VIDS predicts negative/positive versus neutral emotion.
The VIDS reacted somehow to negative/positive versus neutral emotion (shown as forced-choice classification accuracy, P value and Cohen’s d). P values were based on two-sided independent binomial tests (uncorrected).
Supplementary information
Supplementary Information
Supplementary Methods, Results, Tables 1–5 and References.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gan, X., Zhou, F., Xu, T. et al. A neurofunctional signature of subjective disgust generalizes to oral distaste and socio-moral contexts. Nat Hum Behav (2024). https://doi.org/10.1038/s41562-024-01868-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41562-024-01868-x
- Springer Nature Limited