Abstract
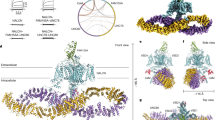
NALCN regulates the resting membrane potential by mediating the Na+ leak current in neurons, and it functions as a channelosome in complex with FAM155A, UNC79, and UNC80. Dysfunction of the NALCN channelosome causes a broad range of neurological and developmental diseases called NALCN channelopathies in humans. How the auxiliary subunits, especially the two large components UNC79 and UNC80, assemble with NALCN and regulate its function remains unclear. Here we report an overall architecture of the human NALCN channelosome. UNC79 and UNC80 each adopt an S-shape super-helical structure consisting of HEAT and armadillo repeats, forming a super-coiled heterodimeric assembly in the cytoplasmic side, which may provide a scaffold for the binding of other potential modulators of the channelosome. The UNC79–UNC80 assembly specifically associates with the NALCN–FAM155A subcomplex through the intracellular II–III linker of NALCN. Disruptions of the interaction interfaces between UNC79 and UNC80, and between the II–III linker of NALCN and the UNC79–UNC80 assembly, significantly reduce the NALCN-mediated currents in HEK293T system, suggesting the importance of the UNC79–UNC80 assembly in regulating channelosome function. Cross-linking mass spectrometry analysis identified an additional calmodulin (CaM) bound in the carboxyl-terminal domain of NALCN. Our study thus provides a structural basis for understanding the unique assembly mechanism and functional regulation of the NALCN channelosome, and also provides an opportunity for the interpretation of many disease-related mutations in UNC80.
Similar content being viewed by others
Introduction
The sodium leak channel NALCN plays an important role in regulations of neuronal excitability, motor function, pain sensitivity, and circadian rhythm1,2,3,4. NALCN forms a unique complex termed NALCN channelosome with the auxiliary subunits FAM155A, UNC79, and UNC805,6,7. The formation of the NALCN channelosome is critical to a fully functional Na+ conductance carried by NALCN8. Knockout of NALCN, UNC79, or UNC80 is lethal in mice1,9,10. In humans, variants of NALCN and the auxiliary subunits, especially UNC80, are linked to a variety of diseases known as NALCN channelopathies11,12, which could cause severe intellectual disabilities, such as infantile hypotonia with psychomotor retardation and characteristic facies (IHPRF)11,13 and congenital contractures of limbs and face, hypotonia and developmental delay (CLIFAHDD)12,14,15.
The ion-conducting subunit NALCN is topologically similar to Nav and Cav channels, which contain four homologous repeats (I–IV) connected by intracellular linkers and belong to the 4 × 6 transmembrane helices (TM) channels within the superfamily of voltage-gated ion channels16. However, the auxiliary subunits of the NALCN channelosome are unique and share no sequence homology to the auxiliary subunits of other 4 × 6 TM channels. Among the auxiliary subunits, FAM155A may facilitate the folding and membrane translocation of NALCN through forming a stable subcomplex with NALCN5). Our previous study has revealed that the mutations in NALCN mainly focus on the pore regions that may directly affect the ion permeation properties a A list of previously reported disease-related mutations in UNC809,11,37,38,39,40,41,42,43. The missense mutations, nonsense mutations, and frameshift mutations are shaded in yellow, cyan, and magenta, respectively. ARSIE, autosomal-recessive severe infantile encephalopathy; ID, intellectual disability; IHPRF2, Infantile hypotonia with psychomotor retardation and characteristic facies-2; PHGRSI, persistent hypotonia, growth retardation and self-injury; PHEGRSID, persistent hypotonia, encephalopathy, growth retardation, and severe intellectual disability; HSIDDD, hypotonia, severe intellectual disability, dyskinesia and dysmorphism; SPGDDPWG, spastic paraplegia, global developmental delay and poor weight gain; IHEH, infantile hypotonia, epilepsy and hyperactivity; HDFF, hypotonia and dysmorphic facial features. b Structural map** of disease-related mutations onto the NALCN channelosome. The diseased-related mutations in NALCN (shown in green sticks) have been discussed in our previous study17. Disease-related mutation sites in UNC80 are shown as spheres and colored by mutation type. Close-up views of the boxed region are shown in c. c Enlarged views of three missense mutation sites in UNC80. Hydrogen bonds are indicated by dashed lines. The mutations are likely to break local hydrogen bonds to affect UNC80 folding and local stability.
Discussion
Our study reports an overall architecture of the human NALCN channelosome, providing an important framework to understand its assembly, regulation, and function. The structure has revealed the interfaces between UNC79 and UNC80, and between the II–III linker of NALCN and the UNC79–UNC80 assembly, which are critical for the formation of intact channelosome and its function. Apart from the structural evidence, results from our patch clamp approach show that the presence of UNC79–UNC80 assembly in the channelosome is essential for the electrophysiological function, which are consistent with other previous studies9,18,20 (Fig. 1c).
The unique intracellular super-helical scaffold of the channelosome provides a molecular basis for the potential coupling between its conformational change and the gating of NALCN, as reported in RyR124. We speculate that the intracellular scaffold not only plays an essential role in directly mediating the channelosome function, but also establishes a platform for the binding of a variety of other regulators, as observed for the bound CaM. Some other proteins, including Src21 and M3R25, have been reported to regulate the channelosome activity through physical interactions. It is likely that these potential regulators modulate the channelosome property through interacting with the channelosome intracellular scaffold. However, structural evidence for the interaction of Src, M3R, and other potential regulators with the channelsome needs further studies.
During the preparation of this manuscript, a similar work was reported elsewhere26, and the major structural observations and conclusions are mostly consistent with our work. There are differences in the protein sample preparation strategies, electrophysiological designs, and some structural interpretations, which make the two works complement each other. The C-interface, considered to be dispensable in that work, turns out to be important for channelosome function in our study. This discrepancy may be due to different electrophysiological systems used in the two works. An extra interface between the I–II linker of NALCN and UNC79 C-half identified in that work was not observed in our study, probably due to different sample conditions (Supplementary Fig. S7). It seems that I–II linker is not as essential as the II–III linker in association between NALCN and the UNC79–UNC80 subcomplex, as alterations of the I–II linker did not obviously impact the channelosome function26. The absence of the I–II linker interacting interface in our study suggests that it is highly dynamic, which in turn leads to a more pronounced local conformational flexibility (Supplementary Video S1). Combined with the other study26, our structure suggests an asynchronous assembly process between NALCN–FAM155A subcomplex and the UNC79–UNC80 assembly, in which the formation of the I–II linker interface may be a subsequent step after the formation of II–III linker interface to further stabilize the channelosome.
Materials and methods
Expression and purification of the human NALCN channelosome
The codon optimized DNAs of human FAM155A, NALCN, UNC80 and UNC79 were individually cloned into a pCAG vector behind a CMV promoter. FAM155A and UNC80 were untagged. The carboxyl termini of NALCN and UNC79 were fused with a green fluorescent protein (GFP) and a 2× FLAG tag, respectively. The four plasmids were co-transfected at a molar ratio of 2:3:3:10 into HEK293F cells (Invitrogen) with polyethylenimine (PEI) when the cell density reached 2 × 106 cells/mL. Transfected cells were collected after 48 h culturing in SMM 293-TII medium (Sino Biological Inc.) under 5% CO2 at 37 °C. Collected cells were resuspended in buffer containing 25 mM MOPS (pH 7.4), 300 mM NaCl, and protease inhibitor cocktail including 2 mM phenylmethylsulfonyl fluoride (PMSF), 1.3 μg/mL aprotinin, 0.7 μg/mL pepstatin, and 5 μg/mL leupeptin, and subsequently lysed by sonication. Subsequently, 1% (w/v) lauryl maltose neopentyl glycol (LMNG, Anatrace), 0.12% (w/v) cholesteryl hemisuccinate Tris salt (CHS, Anatrace) and 0.6 mg/mL GFP-nanobodies were added and incubated at 4 °C for 2 h. The insoluble fraction was precipitated by ultracentrifugation at 255,700× g for 40 min, and the supernatant was applied to glutathione sepharose 4B resin (GS4B, GE Healthcare). The protein-bound resin was washed with 25 mM MOPS (pH 7.4), 400 mM NaCl, and 0.015% GDN (Anatrace). The target protein was eluted with buffer containing 15 mM Tris–HCl (pH 8.0), 15 mM Tris–HCl (pH 8.8), 150 mM NaCl, 0.015% GDN, and 13 mM reduced glutathione. The eluent was then applied to anti-FLAG G1 affinity resin (GenScript) and incubated with the resin for 1 h at 4 °C. The resin was washed with buffer containing 25 mM MOPS (pH 7.4), 150 mM NaCl, and 0.015% GDN. The protein was then eluted with buffer containing 25 mM MOPS (pH 7.4), 150 mM NaCl, 0.015% GDN and 250 μg/mL FLAG peptide. The eluent was concentrated and cross-linked using 2 mM bis(sulfosuccinimidyl) suberate (BS3) at 4 °C for 1.5 h and then quenched by 25 mM Tris–HCl (pH 8.0). The sample was then applied to size-exclusion chromatography (Superose 6, 10/30, GE Healthcare) in buffer containing 25 mM MOPS (pH 7.4), 150 mM NaCl and 0.015% GDN. The fractions containing the NALCN-FAM155A–UNC79–UNC80 complex were pooled and concentrated to about 0.4 mg/mL for electron microscopy and mass spectrometric analysis.
Cryo-EM sample preparation and data collection
For cryo-EM sample preparation, aliquots (4 μL) of the protein sample were loaded onto glow-discharged grids (Quantifoil R 1.2/1.3 Cu 300 mesh) coated with 2 nm carbon film. Under 100% humidity at 8 °C, the grids were blotted for 3 s with Vitrobot (Mark IV, Thermo Fisher Scientific) after waiting for 60 s, and immersed in liquid ethane cooled by liquid nitrogen. The imaging system comprises of a Titan Krios operating at 300 kV, a Gatan K3 Summit detector, and a GIF Quantum energy filter with a 20-eV slit width. Movie stacks were automatically collected via EPU (Thermo Fisher Scientific) in super-resolution mode (×81,000 magnification), with a defocus range from −1.4 to −2.0 μm. Each image stack was exposed for 2.56 s with 0.08 s per frame, resulting in 32 frames and ~50 e−/Å2 of total dose.
Cryo-EM data processing
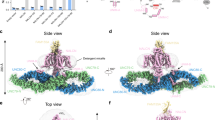
A simplified data processing flowchart can be found in Supplementary Fig. S2f. 33,028 movie stacks (1.087 Å/pixel) were collected and motion-corrected using MotionCor227. The following data processing procedures were performed using CryoSPARC v328. After patch-CTF estimation, around six million particles were automatically picked with picking templates generated from template-free 2D classification. Particles were extracted with a box size of 512 pixels. After two rounds of 2D classification, a total of 590,796 particles were selected from good 2D classes. An initial low-resolution map was generated using these particles as the reference for the subsequent non-uniform refinement job29, yielding a reconstruction with identifiable secondary structure features in the large cytosolic region. The conformational dynamics of the particles were analyzed by the 3D Variability Analysis job30 in CryoSPARC.
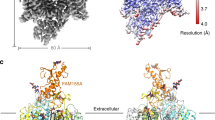
To improve the overall map quality, a heterogeneous refinement (K = 5) was performed and resulted in a class with better secondary structure feature in both the cytosolic and transmembrane regions, consisting of 174,294 selected particles. Further non-uniform refinement of these selected particles yielded a reconstruction of the whole complex at a resolution of 4.5 Å (Map1). To further improve the resolution of local regions, several different masks were applied for local refinements of corresponding re-centered particles. Basically, 383,924 good particles selected from an extra round of 2D classification were first recentered focusing on a masked region. The re-extracted particles were then subtracted using the given mask and applied for local refinement. We have tried masks on NALCN–FAM155A, UNC79–UNC80, UNC79 N-half + UNC80 C-half, and UNC79 C-half + UNC80 N-half. One of such attempts significantly improved the local resolution to 3.3 Å, when the mask surrounding UNC79 N-half + UNC80 C-half was applied (Map2). However, other attempts did not generate maps with significantly improved quality compared to Map1 and therefore were not used. Map resolutions were determined by gold-standard Fourier shell correlation (FSC) at 0.143.
Model building and refinement
A composite map combining Map1 and Map2 was generated for model building. The structure of NALCN–FAM155A (PDB: 7CM3), CaM (PDB: 6MUD) and the predicted structures of UNC79 and UNC80 segments by AlphaFold231 were used as the initial templates. The template models were first docked into the cryo-EM map in Chimera32 and then manually adjusted in Coot33. Large flexible linkers of the UNC79 and UNC80 that were invisible in the density map were removed. The modeling of N-interface and C-interface between UNC79 and UNC80 were facilitated by predicated complex structures by AlphaFold-Multimer34. The modeling of CaM was guided by the cross-linking mass spectrometry data. The map quality of Map2 is relatively high that allows for side chain assignment for some local regions, including the L1, H1, and H2 segments of the NALCN II–III linker. The final model was refined against the composite map by PHENIX35 in real space (phenix.real_space_refine) using the rigid-body parameter. The final overall model was validated using phenix.validation_cryoem. The statistics of the 3D reconstructions and model refinement, and a summary of the overall model can be found in Supplementary Tables S1 and S2, respectively.
Western blotting assay
HEK293F cells were transfected with same amounts of plasmids of C-terminal GFP tagged NALCN (WT/mutant)–FAM155A–UNC79–UNC80 in the same ratio for protein expression. Cells were lysed 48 h post transfection in RIPA buffer (Solarbio) with 1.3 μg/mL aprotinin, 0.7 μg/mL pepstatin, 5 μg/mL leupeptin, and 2 mM phenylmethylsulfonyl fluoride (PMSF), followed by sonication. Lysates normalized by the expression level of GAPDH were loaded onto 14% sodium dodecyl sulfate–polyacrylamide gels for electrophoresis (SDS–PAGE). The separated proteins were then transferred to poly-vinylidene fluoride (PVDF) membranes (Merck Millipore). After blocking with 3% BSA for 1 h at 37 °C in Tris-buffered saline with Tween-20 (TBS-T), the membranes were incubated with the primary antibodies for 1 h at 37 °C and then with the horseradish peroxidase (HRP) labeled secondary antibody (1:5000 dilution, CWBIO) for 1 h. WB bands were visualized with the eECL western blot kit (CWBIO). The primary antibodies used were anti-GFP (1:3000 dilution, Abmart) and anti-GAPDH (1:5000 dilution, Proteintech).
HEK-293T cell culture and transfection
HEK293T cells authenticated by short tandem repeat DNA profiling (gifted from Dr. Chunqing Song) were grown in SMM 293-TII medium (Sino Biological Inc.) supplemented with 10% fetal bovine serum (Biochannel) and 1% penicillin–streptomycin (10,000 U/mL; Cytiva) at 37 °C in a 5% CO2 humidified growth incubator. Cells between passages 10 and 20 were used for electrophysiological experiments. Here, 20–24 h before transfection, HEK293 cells were detached by treatment with 0.25% trypsin for 5 min at room temperature and seeded into new 12-well culture plates at 50% confluency. For patch-clamp experiments, cells reaching 60%–70% confluency were transiently transfected with 3.5 μg total expression plasmids using PEI. All plasmids used for transfection were constructed into the same vector for protein expression. For the WT NALCN channelosome, NALCNGFP, FAM155A, UNC79FLAG, and UNC80 plasmids were mixed at the molar ratio of 3:2:10:3, same to the ratio for protein expression. The different mutation or deletion constructs of NALCN, UNC79, UNC80 contain the same tag to the WT. For each mutant group, the corresponding WT plasmid was replaced by the mutant and other three plasmids are unchanged with the exact same ratio. The transfection mix was removed after 6–8 h, and cells were washed with PBS and cultured in supplemented DMEM. For mock-transfected cells, the vector expressing GFP was used. For NALCN alone-transfected cells, 2.2 μg total plasmids of NALCNGFP was used.
Immunostaining and imaging
About 8–10 h before staining, the transfected HEK293T were seeded on poly-d-lysine-coated glass coverslips. Samples were rinsed with 1× phosphate-buffered saline (PBS) once, and then fixed in 4% paraformaldehyde (pH 7.4) for 10 min at room temperature, followed by permeabilization with 0.25% Triton-X100 (Sigma) in PBS for 10 min at room temperature. Samples were blocked for 1 h in 5% bovine serum albumin (BSA) in PBS and incubated with rabbit anti-GFP antibody (1:500; Abcam) in 5% BSA overnight at 4 °C. After rinsed with PBS, samples were incubated with Alexa 488 secondary antibody (1:1000, Life Technologies) in 5% BSA for 1 h at room temperature and stained with 4′,6-diamidino-2-phenylindole DAPI (Lablead) for 5 min at room temperature. Images were captured by the FV3000-IX83 confocal system (Olympus).
Whole-cell patch clamp electrophysiology
Electrophysiological experiments were conducted 72–96 h after transfection and the transfected HEK-293T cells were seeded on new poly-d-lysine-coated glass coverslips at least 1 h before recording. Patch clamp recordings were performed with a HEKA EPC10 amplifier with PatchMaster software (HEKA) in whole-cell configuration at room temperature (23 ± 2 °C). Micropipettes were pulled with a P-1000 flaming/Brown Micropipette Puller System (Sutter Instrument) and fire-polished with Micro Forge MF2 (Narishige) from the 1.5/1.2 mm (outer diameter (OD)/inner diameter (ID)), thin-walled glass (Sutter Instrument). The series resistance of micropipettes was typically 3–5 MΩ. The recording micropipettes were filled with internal solution containing: 136 mM NaCl, 5 mM EGTA, 10 mM HEPES, and 2 mM Na2ATP (adenosine 5′-triphosphate) (pH 7.2) with NaOH. The recipe of external solution was: 150 mM NaCl, 10 mM HEPES, and 30 mM d-(+)-glucose (pH 7.4) with NaOH. Solution osmolarity was ~290–310 mOsm/L adjusted with glucose, and ~5 mOsm/L lower in the internal solutions than the external solutions of the same experiment.
Traces were acquired at a repetition interval of 4 s. Currents signals were sampled at 25 kHz and filtered at 1 kHz. The holding potential was 0 mV. I–V curves were generated from a group of step potentials (−100 to +100 mV with a 20 mV increment) and at the steady-state during the last 20 ms of voltage steps was averaged. Data from patch clamp recordings were analyzed in Igor Pro (WaveMatrix) and Graphpad Prism. Statistically significant differences (P < 0.05) between means of two groups were determined by a two-tailed t-test. Data are presented as means ± SEM or mean ± SD.
Cross-linking mass spectrometry (XL-MS) analysis
The peak fractions after gel filtration purification containing the BS3 cross-linked protein sample were applied to SDS–PAGE gel and stained with Coomassie Blue G-250. The cross-linked band containing the NALCN channelosome was cut into pieces and put in 50 mM ammonium bicarbonate with trypsin at 37 °C overnight for prior reduction and alkylation. The digested products were extracted twice with 1% formic acid in 50% acetonitrile aqueous solution and dried to reduce volume by speedvac.
For LC–MS/MS analysis, the peptides were separated by a 65 min gradient elution at a flow rate 0.300 µl/min with the Thermo EASY-nLC1200 integrated nano-HPLC system, which is directly interfaced with the Thermo Q Exactive HF-X mass spectrometer. The analytical column was a home-made fused silica capillary column (75 µm ID, 150 mm length; Upchurch, Oak Harbor, WA) packed with C-18 resin (300 A, 3 µm, Varian, Lexington, MA). Mobile phase A consisted of 0.1% formic acid, and mobile phase B consisted of 80% acetonitrile and 0.1% formic acid. The mass spectrometer was operated in the data-dependent acquisition mode using the Xcalibur 4.1 software and there is a single full-scan mass spectrum in the Orbitrap (350–1800m/z, 60,000 resolution) followed by 20 data-dependent MS/MS scans at 30% normalized collision energy. Each mass spectrum was analyzed using the Proteome Discoverer 2.4 and pLink 2.3.936 for the database searching and cross-linking analysis.