Abstract
Background
Neonatal rats can manifest post-stroke mood disorders (PSMD) following middle cerebral artery occlusion (MCAO). We investigated whether cannabidiol (CBD) neuroprotection, previously demonstrated in neonatal rats after MCAO, includes prevention of PSMD development.
Methods
Seven-day-old Wistar rats (P7) underwent MCAO and received either vehicle or 5 mg/kg CBD treatment. Brain damage was quantified by MRI, and neurobehavioral and histological (TUNEL) studies were performed at P14 and P37. PSMD were assessed using the tail suspension test, forced swimming test, and open field tests. The dopaminergic system was evaluated by quantifying dopaminergic neurons (TH+) in the Ventral Tegmental Area (VTA), measuring brain dopamine (DA) concentration and DA transporter expression, and assessing the expression and function D2 receptors (D2R) through [35S]GTPγS binding. Animals without MCAO served as controls.
Results
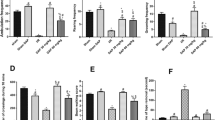
CBD reduced MCAO-induced brain damage and improved motor performance. At P14, MCAO induced depressive-like behavior, characterized by reduced TH+ cell population and DA levels, which CBD did not prevent. However, CBD ameliorated hyperactivity observed at P37, preventing increased DA concentration by restoring D2R function.
Conclusions
These findings confirm the development of PSMD following MCAO in neonatal rats and highlight CBD as a neuroprotective agent capable of long-term functional normalization of the dopaminergic system post-MCAO.
Impact
-
MCAO in neonatal rats led to post-stroke mood disorders consisting in a depression-like picture in the medium term evolving towards long-term hyperactivity, associated with an alteration of the dopaminergic system.
-
The administration of CBD after MCAO did not prevent the development of depressive-like behavior, but reduced long-term hyperactivity, normalizing dopamine receptor function.
-
These data point to the importance of considering the development of depression-like symptoms after neonatal stroke, a well-known complication after stroke in adults.
-
Our work confirms the interest of CBD as a possible treatment for neonatal stroke.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed in this study are available from the corresponding author on reasonable request.
References
Wang, Z. et al. Diversiform etiologies for poststroke depression. Front. Psychiatry 10, 761 (2019).
Villa, R. F., Ferrari, F. & Moretti, A. Post-stroke depression: Mechanisms and pharmacological treatment. Pharmacol. Ther. 184, 131–144 (2018).
Lehman, L. L. & Rivkin, M. J. Perinatal arterial ischemic stroke: Presentation, risk factors, evaluation, and outcome. Pediatr. Neurol. 51, 760–768 (2014).
Kirton, A. et al. Symptomatic neonatal arterial ischemic stroke: The International Pediatric Stroke Study. Pediatrics 128, e1402–e1410 (2011).
Álvarez-García, M., Cuellar-Flores, I., Sierra-García, P. & Martínez-Orgado, J. Mood disorders in children following neonatal hypoxic-ischemic encephalopathy. PLoS One 17, e0263055 (2022).
Villa, M. et al. The role of the Dopamine system in post-stroke mood disorders in newborn rats. Int J. Mol. Sci. 24, 3229 (2023).
Martínez-Orgado, J., Villa, M. & Del Pozo, A. Cannabidiol for the treatment of neonatal hypoxic-ischemic brain injury. Front Pharm. 11, 584533 (2021).
Ceprián, M. et al. Cannabidiol reduces brain damage and improves functional recovery in a neonatal rat model of arterial ischemic stroke. Neuropharmacology 116, 151–159 (2017).
Grace, A. A. Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat. Rev. Neurosci. 17, 524–532 (2016).
Giannopoulou, I., Pagida, M. A., Briana, D. D. & Panayotacopoulou, M. T. Perinatal hypoxia as a risk factor for psychopathology later in life: the role of dopamine and neurotrophins. Hormones 17, 25–32 (2018).
Semple, B. D., Blomgren, K., Gimlin, K., Ferriero, D. M. & Noble-Haeusslein, L. J. Brain development in rodents and humans: Identifying benchmarks of maturation and vulnerability to injury across species. Prog. Neurobiol. 106–107, 1–16 (2013).
Derugin, N., Ferriero, D. M. & Vexler, Z. S. Neonatal reversible focal cerebral ischemia: A new model. Neurosci. Res. 32, 349–353 (1998).
Zaichenko, M. I., Vanetsian, G. L. & Merzhanova, G. K. Differences in the behavior of impulsive and self-controlled rats in the open field and dark-light box tests. Neurosci. Behav. Physiol. 42, 1046–1054 (2012).
Pazos, M. R. et al. Cannabidiol administration after hypoxia-ischemia to newborn rats reduces long-term brain injury and restores neurobehavioral function. Neuropharmacology 63, 776–783 (2012).
Paxinos G., Watson C. The rat brain in stereotaxic coordinates. Academic Press, San Diego 1997.
Ashok, A. H. et al. The dopamine hypothesis of bipolar affective disorder: The state of the art and implications for treatment. Mol. Psychiatry 22, 666–679 (2017).
Bosenbark, D. D., Krivitzky, L., Ichord, R., Jastrzab, L. & Billinghurst, L. Attention and executive functioning profiles in children following perinatal arterial ischemic stroke. Child Neuropsychol. 24, 106–123 (2018).
Mcpherson R. J., & Juul S. E. Recent Trends in Erythropoietin-mediated Neuroprotection. Int J Dev Neurosci 26, 103–111 (2009).
Egusquiza, I. et al. Characterization of dopamine D2 receptor coupling to G proteins in postmortem brain of subjects with schizophrenia. Pharm. Rep. 73, 1136–1146 (2021).
Demers, E. J., McPherson, R. J. & Juul, S. E. Erythropoietin protects dopaminergic neurons and improves neurobehavioral outcomes in juvenile rats after neonatal hypoxia-ischemia. Pediatr. Res 58, 297–301 (2005).
Ceprián, M. et al. Cannabidiol administration prevents hypoxia-ischemia-induced hypomyelination in newborn rats. Front. Pharm. 10, 1131 (2019).
Abrantes De Lacerda Almeida, T. et al. Intraperitoneal cannabidiol attenuates neonatal germinal matrix hemorrhage-induced neuroinflamation and perilesional apoptosis. Neurol. Res. 41, 980–990 (2019).
Ruiz Brunner, M., Cuestas, E., Heinen, F. & Schroeder, A. S. Growth in infants, children and adolescents with unilateral and bilateral cerebral palsy. Sci. Rep. 12, 1–9 (2022).
Rice, F. et al. Adolescent and adult differences in major depression symptom profiles. J. Affect. Disord. 243, 175–181 (2019).
Northoff, G., Hirjak, D., Wolf, R. C., Magioncalda, P. & Martino, M. All roads lead to the motor cortex: psychomotor mechanisms and their biochemical modulation in psychiatric disorders. Mol. Psychiatry 26, 92–102 (2021).
Loubinoux, I. et al. Post-stroke depression: Mechanisms, translation and therapy. J. Cell Mol. Med. 16, 1961–1969 (2012).
Acknowledgements
We wish to thank Carlos Vargas and Alysha Hernández for excellent assistance and Jason Willis-Lee for scientific writing assistance This research was funded by the PI19/00927, PID2019-106404RB-I00 and RD21/0012/0023 projects, integrated in the Plan Nacional de R + D + I, AES 2017-2020 and 2021-2023, funded by the “Instituto de Salud Carlos III” (ISCIII) and co-funded by the European Regional Development Fund (ERDF) “A way to make Europe” and Next Generation EU funds (supporting actions from Resilience and Recovery Mechanisms [MRR]), and the Basque Government (IT1512/22).
Author information
Authors and Affiliations
Contributions
Conceptualization, M.V. and J.M-O; methodology, M.V., L.F.C, M.J.C and J.M-O.; experimental model and neurobehavioral studies: M.V., M.M-V, A.H.; histologic and biochemical studies: M.dH-R, A.R., L.S.; HPLC A.G-S., M.J.C; [35S]GTPγS binding assays: I.M-A, C.M., L.F.C; data curation: M.V., M.M-V, A.H, I.M-A, A.G-S., C.M, M.dH-R, A.R., L.S; data analysis: M.V., L.F.C, M.J.C and J.M-O; writing—original draft preparation, J.M-O; writing—review and editing, M.V., L.F.C, M.J.C and J.M-O.; funding acquisition, L.F.C, M.J.C and J.M-O. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Villa, M., Martínez-Vega, M., Silva, L. et al. Effects of cannabidiol in post-stroke mood disorders in neonatal rats. Pediatr Res 95, 1783–1790 (2024). https://doi.org/10.1038/s41390-024-03077-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-024-03077-8
- Springer Nature America, Inc.