Abstract
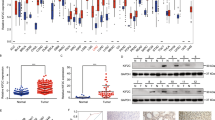
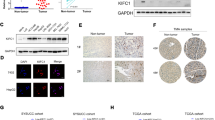
The mechanistic target of rapamycin complex 1 (mTORC1) signaling pathway is frequently reported to be hyperactivated in hepatocellular carcinoma (HCC) and contributes to HCC recurrence. However, the underlying regulatory mechanisms of mTORC1 signaling in HCC are not fully understood. In the present study, we found that the expression of kinesin family member 18B (KIF18B) was positively correlated with mTORC1 signaling in HCC, and the upregulation of KIF18B and p-mTOR was associated with a poor prognosis and HCC recurrence. Utilizing in vitro and in vivo assays, we showed that KIF18B promoted HCC cell proliferation and migration through activating mTORC1 signaling. Mechanistically, we identified Actin gamma 1 (γ-Actin) as a binding partner of KIF18B. KIF18B and γ-Actin synergistically modulated lysosome positioning, promoted mTORC1 translocation to lysosome membrane, and prohibited p70 S6K from entering lysosomes for degradation, which finally led to the enhancement of mTORC1 signaling transduction. Moreover, we found that KIF18B was a direct target of Forkhead box M1, which explains the potential mechanism of KIF18B overexpression in HCC. Our study highlights the potential of KIF18B as a therapeutic target for the treatment of HCC.
Similar content being viewed by others
Introduction
Primary liver cancer is the fourth leading cause of cancer-related death worldwide, with estimated 906,000 new cases and 830,000 deaths in 2020 [1]. Hepatocellular carcinoma (HCC) accounts for the majority of primary liver cancer, integrated studies have demonstrated that HCC is highly heterogeneous, with alternations in genetics, molecules, and cell signaling pathways among patients and even among different compartments within one tumor [2, 3]. Although considerable progress has been achieved in previous studies [48]. mTOR inhibitors have been proved to decrease HCC recurrence rate after liver transplantation [49, 50]. Our research suggests that the combination of mTOR inhibitors and KIF18B inhibitors may synergistically enhance their preventing recurrence and anti-tumor effect.
In summary, the results of this study demonstrate that KIF18B is a direct target of FOXM1 and promotes HCC recurrence through activating mTORC1 signaling. Mechanistically, KIF18B interacts with γ-Actin. They synergistically modulate lysosome positioning, subsequently promote mTORC1 translocation to lysosome membrane, and prevent p70 S6K from entering lysosomes for degradation (Fig. 6F). Our study highlights the potential of KIF18B as a therapeutic target for the treatment of HCC.
Materials and methods
Cell lines and clinical samples
Hep3B, Huh1, Huh7, LO2 and HEK293T were obtained from American Type Culture Collection. HCCLM3, BEL-7402 and SMMC-7721 were purchased from Cell Bank of Chinese Academy of Sciences. The cell lines HCCLM3, BEL-7402, Hep3B, Huh1, Huh7 and 293 T were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) (Biological Industries, 04-001-1), 100 U/ml penicillin and 0.1 mg/ml streptomycin. LO2 and SMMC-7721 cell lines were maintained in Roswell Park Memorial Institute-1640 (RPMI-1640) medium supplemented with 10% FBS, 100 U/ml penicillin and 0.1 mg/ml streptomycin. All of these cells were incubated at 37 °C in an atmosphere of 5% CO2. A total of 60 HCC tissues were obtained from the affiliated hospital of Hangzhou Normal University. Written informed consent was obtained from each patient, and the study was approved by the Biomedical Ethics Committee of the affiliated hospital of Hangzhou Normal University. This study was performed in accordance with the Declaration of Helsinki.
Lentivirus infection and in vivo models
HCCLM3 or BEL-7402 cells were infected with lentiviruses in presence of polybrene and selected with 2 μg/mL and 1 μg/mL puromycin, respectively, for 7 days. For the subcutaneous xenograft model, five-week-old male BALB/c nude mice were used. All animal care and handling procedures were performed in accordance with the National Institutes of Health guide for the care and use of Laboratory animals and approved by the Animal Care and Use Committee of Hangzhou Normal University. The control and experimental HCCLM3 cells (5 × 106) were suspended in 100 μL PBS and then injected subcutaneously into the flanks of the nude mice. Tumor growth was examined over the course of 4 weeks and tumor volume was measured with calipers and calculated using the formula: Volume (mm3) = length × width2/2 as previously reported [51]. After 4 weeks, the mice were sacrificed and the tumors were weighed, fixed, and paraffin-embedded for further analysis.
Statistical analysis
Spearman test was used to analyze the correlation between KIF18B and p-mTOR expression in HCC tissues. The Kaplan-Meier method was used to analyze patient survival. The Student, t test was used to for the comparisons between two groups. Statistical analysis and graphs were generated using the SPSS 20.0 software and GraphPad Prism 7 software. P < 0.05 was considered as significant.
Data availability
The authors declare that all data in the article are available.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Llovet JM, Montal R, Sia D, Finn RS. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat Rev Clin Oncol. 2018;15:599–616.
Li LY, Yang JF, Rong F, Luo ZP, Hu S, Fang H, et al. ZEB1 serves an oncogenic role in the tumourigenesis of HCC by promoting cell proliferation, migration, and inhibiting apoptosis via Wnt/beta-catenin signaling pathway. Acta Pharmacol Sin. 2021;42:1676–89.
Chen J, Rajasekaran M, **a H, Zhang X, Kong SN, Sekar K, et al. The microtubule-associated protein PRC1 promotes early recurrence of hepatocellular carcinoma in association with the Wnt/beta-catenin signalling pathway. Gut. 2016;65:1522–34.
Chen J, **a H, Zhang X, Karthik S, Pratap SV, Ooi LL, et al. ECT2 regulates the Rho/ERK signalling axis to promote early recurrence in human hepatocellular carcinoma. J Hepatol. 2015;62:1287–95.
Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380:1450–62.
Njei B, Rotman Y, Ditah I, Lim JK. Emerging trends in hepatocellular carcinoma incidence and mortality. Hepatology. 2015;61:191–9.
Saxton RA, Sabatini DM. mTOR signaling in growth, metabolism, and disease. Cell. 2017;168:960–76.
Sancak Y, Peterson TR, Shaul YD, Lindquist RA, Thoreen CC, Bar-Peled L, et al. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science. 2008;320:1496–501.
Hara K, Yonezawa K, Kozlowski MT, Sugimoto T, Andrabi K, Weng QP, et al. Regulation of eIF-4E BP1 phosphorylation by mTOR. J Biol Chem. 1997;272:26457–63.
Burnett PE, Barrow RK, Cohen NA, Snyder SH, Sabatini DM. RAFT1 phosphorylation of the translational regulators p70 S6 kinase and 4E-BP1. Proc Natl Acad Sci USA. 1998;95:1432–7.
Ferrín G, Guerrero M, Amado V, Rodríguez-Perálvarez M, De la Mata M. Activation of mTOR signaling pathway in hepatocellular carcinoma. Int J Mol Sci. 2020;21:1266.
Matter MS, Decaens T, Andersen JB, Thorgeirsson SS. Targeting the mTOR pathway in hepatocellular carcinoma: current state and future trends. J Hepatol. 2014;60:855–65.
Miki H, Okada Y, Hirokawa N. Analysis of the kinesin superfamily: insights into structure and function. Trends Cell Biol. 2005;15:467–76.
Tanenbaum ME, Macurek L, van der Vaart B, Galli M, Akhmanova A, Medema RH. A complex of Kif18b and MCAK promotes microtubule depolymerization and is negatively regulated by Aurora kinases. Curr Biol. 2011;21:1356–65.
Stout JR, Yount AL, Powers JA, Leblanc C, Ems-McClung SC, Walczak CE. Kif18B interacts with EB1 and controls astral microtubule length during mitosis. Mol Biol Cell. 2011;22:3070–80.
Walczak CE, Zong H, Jain S, Stout JR. Spatial regulation of astral microtubule dynamics by Kif18B in PtK cells. Mol Biol Cell. 2016;27:3021–30.
van Heesbeen R, Raaijmakers JA, Tanenbaum ME, Halim VA, Lelieveld D, Lieftink C, et al. Aurora A, MCAK, and Kif18b promote Eg5-independent spindle formation. Chromosoma. 2017;126:473–86.
McHugh T, Gluszek AA, Welburn JPI. Microtubule end tethering of a processive kinesin-8 motor Kif18b is required for spindle positioning. J Cell Biol. 2018;217:2403–16.
Ji Z, Pan X, Shang Y, Ni DT, Wu FL. KIF18B as a regulator in microtubule movement accelerates tumor progression and triggers poor outcome in lung adenocarcinoma. Tissue Cell. 2019;61:44–50.
Wu Y, Wang A, Zhu B, Huang J, Lu E, Xu H, et al. KIF18B promotes tumor progression through activating the Wnt/beta-catenin pathway in cervical cancer. Onco Targets Ther. 2018;11:1707–20.
Yang B, Wang S, **e H, Wang C, Gao X, Rong Y, et al. KIF18B promotes hepatocellular carcinoma progression through activating Wnt/beta-catenin-signaling pathway. J Cell Physiol. 2020;235:6507–14.
Li B, Liu B, Zhang X, Liu H, He L. KIF18B promotes the proliferation of pancreatic ductal adenocarcinoma via activating the expression of CDCA8. J Cell Physiol. 2020;235:4227–38.
Zhao F, Feng Y, Zhang X, Liu X, Li A. Kinesin superfamily member 18B (KIF18B) promotes cell proliferation in colon adenocarcinoma. Cancer Manag Res. 2020;12:12769–78.
Gao T, Yu L, Fang Z, Liu J, Bai C, Li S, et al. KIF18B promotes tumor progression in osteosarcoma by activating beta-catenin. Cancer Biol Med. 2020;17:371–86.
Qin L, Wang Z, Tao L, Wang Y. ER stress negatively regulates AKT/TSC/mTOR pathway to enhance autophagy. Autophagy. 2010;6:239–47.
Gao B, Li S, Tan Z, Ma L, Liu J. ACTG1 and TLR3 are biomarkers for alcohol-associated hepatocellular carcinoma. Oncol Lett. 2019;17:1714–22.
Yan Y, Xu H, Zhang L, Zhou X, Qian X, Zhou J, et al. RRAD suppresses the Warburg effect by downregulating ACTG1 in hepatocellular carcinoma. Onco Targets Ther. 2019;12:1691–703.
Cabukusta B, Neefjes J. Mechanisms of lysosomal positioning and movement. Traffic. 2018;19:761–9.
Ballabio A, Bonifacino JS. Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat Rev Mol Cell Biol. 2020;21:101–18.
Saftig P, Klumperman J. Lysosome biogenesis and lysosomal membrane proteins: trafficking meets function. Nat Rev Mol Cell Biol. 2009;10:623–35.
Liu GY, Sabatini DM. mTOR at the nexus of nutrition, growth, ageing and disease. Nat Rev Mol Cell Biol. 2020;21:183–203.
Korolchuk VI, Saiki S, Lichtenberg M, Siddiqi FH, Roberts EA, Imarisio S, et al. Lysosomal positioning coordinates cellular nutrient responses. Nat Cell Biol. 2011;13:453–60.
Shum MS, Pasquier E, Po’uha ST, O’Neill GM, Chaponnier C, Gunning PW, et al. gamma-actin regulates cell migration and modulates the ROCK signaling pathway. FASEB J. 2011;25:4423–33.
Santos-Ledo A, Garcia-Macia M, Campbell PD, Gronska M, Marlow FL. Kinesin-1 promotes chondrocyte maintenance during skeletal morphogenesis. PLoS Genet. 2017;13:e1006918.
Blott EJ, Griffiths GM. Secretory lysosomes. Nat Rev Mol Cell Biol. 2002;3:122–31.
Lucanus AJ, Yip GW. Kinesin superfamily: roles in breast cancer, patient prognosis and therapeutics. Oncogene. 2018;37:833–8.
Qiao Y, Pei Y, Luo M, Rajasekaran M, Hui KM, Chen J. Cytokinesis regulators as potential diagnostic and therapeutic biomarkers for human hepatocellular carcinoma. Exp Biol Med. 2021;246:1343–54.
Li X, Huang W, Huang W, Wei T, Zhu W, Chen G, et al. Kinesin family members KIF2C/4A/10/11/14/18B/20A/23 predict poor prognosis and promote cell proliferation in hepatocellular carcinoma. Am J Transl Res. 2020;12:1614–39.
Li Q, Qiu J, Yang H, Sun G, Hu Y, Zhu D, et al. Kinesin family member 15 promotes cancer stem cell phenotype and malignancy via reactive oxygen species imbalance in hepatocellular carcinoma. Cancer Lett. 2020;482:112–25.
Wu YP, Ke ZB, Zheng WC, Chen YH, Zhu JM, Lin F, et al. Kinesin family member 18B regulates the proliferation and invasion of human prostate cancer cells. Cell Death Dis. 2021;12:302.
Qiu MJ, Zhang L, Chen YB, Zhu LS, Zhang B, Li QT, et al. KIF18B as a regulator in tumor microenvironment accelerates tumor progression and triggers poor outcome in hepatocellular carcinoma. Int J Biochem Cell Biol. 2021;137:106037.
Kim LC, Cook RS, Chen J. mTORC1 and mTORC2 in cancer and the tumor microenvironment. Oncogene. 2017;36:2191–201.
Petherick KJ, Williams AC, Lane JD, Ordonez-Moran P, Huelsken J, Collard TJ, et al. Autolysosomal beta-catenin degradation regulates Wnt-autophagy-p62 crosstalk. EMBO J. 2013;32:1903–16.
Liu X, Gong H, Huang K. Oncogenic role of kinesin proteins and targeting kinesin therapy. Cancer Sci. 2013;104:651–6.
Shah JJ, Kaufman JL, Zonder JA, Cohen AD, Bensinger WI, Hilder BW, et al. A Phase 1 and 2 study of Filanesib alone and in combination with low-dose dexamethasone in relapsed/refractory multiple myeloma. Cancer. 2017;123:4617–30.
Garcia-Saez I, Skoufias DA. Eg5 targeting agents: from new anti-mitotic based inhibitor discovery to cancer therapy and resistance. Biochem Pharmacol. 2021;184:114364.
Yu X-N, Chen H, Liu T-T, Wu J, Zhu J-M, Shen X-Z. Targeting the mTOR regulatory network in hepatocellular carcinoma: are we making headway? Biochim Biophys Acta Rev Cancer. 2019;1871:379–91.
Lu X, Paliogiannis P, Calvisi DF, Chen X. Role of the mammalian target of rapamycin pathway in liver cancer: from molecular genetics to targeted therapies. Hepatology. 2021;73:49–61.
Schnitzbauer AA, Filmann N, Adam R, Bachellier P, Bechstein WO, Becker T, et al. mTOR inhibition is most beneficial after liver transplantation for hepatocellular carcinoma in patients with active tumors. Ann Surg. 2020;272:855–62.
Chang C, Rajasekaran M, Qiao Y, Dong H, Wang Y, **a H, et al. The aberrant upregulation of exon 10-inclusive SREK1 through SRSF10 acts as an oncogenic driver in human hepatocellular carcinoma. Nat Commun. 2022;13:1363.
Acknowledgements
This research was supported by Zhejiang Provincial Natural Science Foundation of China (LR21H160001, LY21H160044, and LY21H160043); “Pioneer” and “Leading Goose” R&D Program of Zhejiang Province (2022C03004); National Natural Science Foundation of China (81902507, 82372664, and 82072646); Research Project of **an Microecological Biomedicine Shandong Laboratory (JNL-2022029C); Fundamental Research Funds for the Central Universities (226-2023-00107); National Administration of Traditional Chinese Medicine-Zhejiang Province co-construction Project (GZY- ZJ-KJ-24045).
Author information
Authors and Affiliations
Contributions
QL, MQS, YM, MQF, CX, and MLW contributed to acquiring and analyzing the data; QL drafted the manuscript; HD, JL, CJC, and FTB discussed results and edited the manuscript; JXC, YTQ, and QL supervised the research and revised manuscript. The authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Q., Sun, M., Meng, Y. et al. Kinesin family member 18B activates mTORC1 signaling via actin gamma 1 to promote the recurrence of human hepatocellular carcinoma. Oncogenesis 12, 54 (2023). https://doi.org/10.1038/s41389-023-00499-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41389-023-00499-7
- Springer Nature Limited