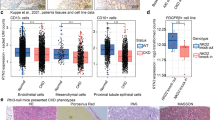
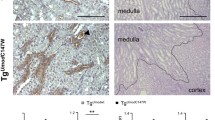
Abstract
Reticulon 3 (RTN3) is an endoplasmic reticulum protein that has previously been shown to play roles in neurodegenerative diseases, but little is known about its function in the kidneys. The aim of the present study was to clarify the roles of RTN3 in chronic kidney disease (CKD) and kidney fibrosis. In this study, RTN3 levels were measured in kidney tissues from healthy controls and CKD or kidney fibrosis patients. An RTN3-null mouse model was generated to explore the pathophysiological roles of RTN3 in the kidneys. The underlying mechanisms were studied in primary proximal tubular epithelial cells and HEK293 cells in vitro. The results showed that (1) a reduction in RTN3 in mice induces CKD and kidney fibrosis; (2) decreased RTN3 expression is found in patients with CKD; (3) RTN3 plays critical roles in regulating collagen biosynthesis and mitochondrial function; and (4) mechanistically, RTN3 regulates these phenotypes by interacting with GC-Rich Promoter Binding Protein 1 (GPBP1), which activates the IGF2-JAK2-STAT3 pathway. Our study indicates that RTN3 might play crucial roles in CKD and kidney fibrosis and that a reduction in RTN3 in the kidneys might be a risk factor for CKD and kidney fibrosis.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) is a type of kidney disease in which there is gradual loss of kidney function over a period of months or years1. It affects 10–15% of the population worldwide and is now recognized as the most rapidly increasing contributor to the global burden of disease. The costs related to CKD and end-stage renal disease (the terminal manifestation of CKD) are an enormous burden for all healthcare systems around the world2. Usually, CKD does not cause symptoms until it reaches an advanced stage. At the middle and late stages of CKD, edema, fatigue, vomiting, loss of appetite, or confusion may develop3. In the clinic, the recommended diagnosis and testing procedures for CKD include blood pressure measurement, urine testing, and serum creatinine measurement2. Although many early-onset CKD cases were not previously considered to be of genetic origin, studies in recent years have discovered that approximately 20% of CKD cases may be associated with genetic factors4. Thus far, more than 200 candidate genes, including collagen-related genes, mitochondria-related genes, and ion channel-related genes, have been identified for 70% of CKD cases associated with genetic factors4.
The reticulon (RTN) protein family consists of RTN1 through RTN4 in mammals, and its members have a signature C-terminal RTN homolog domain (RHD)5. Biochemically, RTNs can shape the structure of the tubular endoplasmic reticulum (ER) due to the ω- (wedge-shaped) membrane topology in the N- and C-terminal domains6. Functionally, RTNs have been found to regulate neurite outgrowth and negatively modulate the activity of Alzheimer’s β-secretase, and they are pathologically linked to axonopathy in hereditary spastic paraplegias7,8,9. Interestingly, the functions of RTNs in human peripheral organs are still not clear. Previously, we found that increased RTN3 can lead to obesity and hypertriglyceridemia via interaction of RTN3 with heat shock protein family A (Hsp70) member 531, an important risk factor linked to diabetic kidney disease (DKD). In addition, upregulation of IGF2 has been proven to lead to Perlman syndrome, a disease related to kidney injury, in mouse nephron progenitor cells15. In proximal tubule cells, increased IGF levels promote fibronectin expression through a pathway involving Nox-dependent ROS generation and Akt signaling32. In our study, the expression of IGF2 was dramatically increased in RTN3-null mice with CKD, which indicated that kidney injury might have occurred via the IGF2 pathway.
JAK2-STAT3 activation has been detected in several types of kidney disease, such as acute kidney injury, CKD and DKD33,34,35. Podocyte-specific JAK2 overexpression has been found to accelerate disease progression in a DKD mouse model36, and a STAT3 inhibitor (S3I-201) can attenuate fibrosis and inflammation in UUO kidneys37. In a CKD mouse model, activation of the JAK2-STAT3 pathway has been found to induce oxidative stress and aggravate CKD38. In acute kidney injury and DKD, activation of the JAK2-STAT3 pathway also promotes the immune inflammatory response and induces apoptosis33. Furthermore, administration of JAK2 inhibitors, including baricitinib and Huang Gan formula, effectively improves kidney function in CKD and DKD, which indicates that JAK2 inhibitors might be useful as new therapies for CKD and DKD38,39.
Oxidative stress is harmful to cells due to excessive generation of ROS, which has been proven to lead to CKD40. Disruption of mitochondrial structure and function may promote the oxidative stress response41,42. In our study, we detected disruption of mitochondria in RTN3-null mouse kidneys. Previous studies have revealed that activation of the JAK2-STAT3 pathway may induce mitochondrial dysfunction and oxidative stress by disrupting the balance between BCL2 and BAX43,44. A specific JAK2 inhibitor can attenuate TNF-α-induced oxidative stress in renal tubular epithelial cells45. Hence, the mitochondrial dysfunction in RTN3-null mouse kidneys described in our current study may also have occurred via the JAK2-STAT3 pathway.
In summary, our study suggests that the RTN3-null mouse model might be an ideal model for research on CKD and kidney fibrosis because the model recapitulates features of CKD. Reduced RTN3 expression is a potential risk factor for glomerulosclerosis and tubulointerstitium in the kidney because it promotes collagen synthesis and aggregation and impairs mitochondrial structure and function, partly through alteration of the localization of GPBP1 and activation of the IGF2-JAK2-STAT3 pathway. Hence, our findings shed light on the importance of the relationship between ER proteins and CKD/kidney fibrosis in humans and animals. Collectively, our data suggest that RTN3 is a key molecule in the kidneys.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Webster, A. C., Nagler, E. V., Morton, R. L. & Masson, P. Chronic kidney disease. Lancet 389, 1238–1252 (2017).
Glassock, R. J., Warnock, D. G. & Delanaye, P. The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat. Rev. Nephrol. 13, 104–114 (2017).
Provenzano, M. et al. Epidemiology of cardiovascular risk in chronic kidney disease patients: the real silent killer. Rev. Cardiovasc. Med. 20, 209–220 (2019).
Vivante, A. & Hildebrandt, F. Exploring the genetic basis of early-onset chronic kidney disease. Nat. Rev. Nephrol. 12, 133–146 (2016).
Yan, R., Shi, Q., Hu, X. & Zhou, X. Reticulon proteins: emerging players in neurodegenerative diseases. Cell. Mol. Life. Sci. 63, 877–889 (2006).
Voeltz, G. K., Prinz, W. A., Shibata, Y., Rist, J. M. & Rapoport, T. A. A class of membrane proteins sha** the tubular endoplasmic reticulum. Cell 124, 573–586 (2006).
Chiurchiu, V., Maccarrone, M. & Orlacchio, A. The role of reticulons in neurodegenerative diseases. Neuromol. Med. 16, 3–15 (2014).
Sharoar, M. G. & Yan, R. Effects of altered RTN3 expression on BACE1 activity and Alzheimer’s neuritic plaques. Rev. Neurosci. 28, 145–154 (2017).
Montenegro, G. et al. Mutations in the ER-sha** protein reticulon 2 cause the axon-degenerative disorder hereditary spastic paraplegia type 12. J. Clin. Investig. 122, 538–544 (2012).
**ang, R. et al. Increased reticulon 3 (RTN3) leads to obesity and hypertriglyceridemia by interacting with heat shock protein family A (Hsp70) member 5 (HSPA5). Circulation 138, 1828–1838 (2018).
Shi, Q. et al. Impact of RTN3 deficiency on expression of BACE1 and amyloid deposition. J. Neurosci. 34, 13954–13962 (2014).
De Rasmo, D. et al. Mitochondrial dynamics of proximal tubular epithelial cells in nephropathic cystinosis. Int. J. Mol. Sci. 21, 192 (2019).
Serasinghe, M. N. & Chipuk, J. E. Mitochondrial fission in human diseases. Handb. Exp. Pharm. 240, 159–188 (2017).
Bach, L. A. & Hale, L. J. Insulin-like growth factors and kidney disease. Am. J. Kidney Dis. 65, 327–336 (2015).
Hunter, R. W. et al. Loss of Dis3l2 partially phenocopies Perlman syndrome in mice and results in up-regulation of Igf2 in nephron progenitor cells. Genes. Dev. 32, 903–908 (2018).
Wang, K., Wang, C., **ao, F., Wang, H. & Wu, Z. JAK2/STAT2/STAT3 are required for myogenic differentiation. J. Biol. Chem. 283, 34029–34036 (2008).
Mahmoud, A. M. et al. Mesoporous silica nanoparticles trigger liver and kidney injury and fibrosis via altering TLR4/NF-kappaB, JAK2/STAT3 and Nrf2/HO-1 signaling in rats. Biomolecules 9, 528 (2019).
Patera, F., Cudzich-Madry, A., Huang, Z. & Fragiadaki, M. Renal expression of JAK2 is high in polycystic kidney disease and its inhibition reduces cystogenesis. Sci. Rep. 9, 4491 (2019).
Zhao, X. et al. Edaravone alleviates cell apoptosis and mitochondrial injury in ischemia-reperfusion-induced kidney injury via the JAK/STAT pathway. Biol. Res. 53, 28 (2020).
Feng, J. et al. SIRT6 suppresses glioma cell growth via induction of apoptosis, inhibition of oxidative stress and suppression of JAK2/STAT3 signaling pathway activation. Oncol. Rep. 35, 1395–1402 (2016).
Revert-Ros, F. et al. Goodpasture antigen-binding protein (GPBP) directs myofibril formation: identification of intracellular downstream effector 130-kDa GPBP-interacting protein (GIP130). J. Biol. Chem. 286, 35030–35043 (2011).
Deng, M. et al. Increased expression of reticulon 3 in neurons leads to reduced axonal transport of beta site amyloid precursor protein-cleaving enzyme 1. J. Biol. Chem. 288, 30236–30245 (2013).
**ang, R. & Zhao, S. RTN3 inducing apoptosis is modulated by an adhesion protein CRELD1. Mol. Cell. Biochem. 331, 225–230 (2009).
Grumati, P. et al. Full length RTN3 regulates turnover of tubular endoplasmic reticulum via selective autophagy. Elife 6, e25555 (2017).
Fan, Y. et al. RTN1 mediates progression of kidney disease by inducing ER stress. Nat. Commun. 6, 7841 (2015).
**ao, W. et al. Knockdown of RTN1A attenuates ER stress and kidney injury in albumin overload-induced nephropathy. Am. J. Physiol. Ren. Physiol. 310, F409–F415 (2016).
Fan, Y. et al. Inhibition of reticulon-1A-mediated endoplasmic reticulum stress in early AKI attenuates renal fibrosis development. J. Am. Soc. Nephrol. 28, 2007–2021 (2017).
Pu, Y. et al. Association of genetic variations in RTN4 3’-UTR with risk for clear cell renal cell carcinoma. Fam. Cancer 17, 129–134 (2018).
Werner, H. Insulin-like growth factors in development, cancers and aging. Cells 9, 2309 (2020).
Youssef, A., Aboalola, D. & Han, V. K. The roles of insulin-like growth factors in mesenchymal stem cell niche. Stem. Cells Int. 2017, 9453108 (2017).
Coto, E. et al. Genetic variation in the H19-IGF2 cluster might confer risk of develo** impaired renal function. DNA Cell. Biol. 37, 617–625 (2018).
New, D. D., Block, K., Bhandhari, B., Gorin, Y. & Abboud, H. E. IGF-I increases the expression of fibronectin by Nox4-dependent Akt phosphorylation in renal tubular epithelial cells. Am. J. Physiol. Cell. Physiol. 302, C122–C130 (2012).
Zhu, H. et al. Curcumin attenuates inflammation and cell apoptosis through regulating NF-kappaB and JAK2/STAT3 signaling pathway against acute kidney injury. Cell. Cycle 19, 1941–1951 (2020).
Liu, Y. et al. C-X-C motif chemokine receptor 4 aggravates renal fibrosis through activating JAK/STAT/GSK3beta/beta-catenin pathway. J. Cell. Mol. Med. 24, 3837–3855 (2020).
Hu, T. Y., Li, L. M. & Pan, Y. Z. CTRP3 inhibits high glucose-induced human glomerular mesangial cell dysfunction. J. Cell. Biochem. 120, 5729–5736 (2019).
Zhang, H. et al. Podocyte-specific JAK2 overexpression worsens diabetic kidney disease in mice. Kidney Int. 92, 909–921 (2017).
Pang, M. et al. A novel STAT3 inhibitor, S3I-201, attenuates renal interstitial fibroblast activation and interstitial fibrosis in obstructive nephropathy. Kidney Int. 78, 257–268 (2010).
Deng, Q. et al. Huang Gan formula eliminates the oxidative stress effects of advanced oxidation protein products on the divergent regulation of the expression of AGEs receptors via the JAK2/STAT3 pathway. Evid. Based Complement. Altern. Med. 2017, 4520916 (2017).
Tuttle, K. R. et al. JAK1/JAK2 inhibition by baricitinib in diabetic kidney disease: results from a Phase 2 randomized controlled clinical trial. Nephrol. Dial. Transpl. 33, 1950–1959 (2018).
Daenen, K. et al. Oxidative stress in chronic kidney disease. Pediatr. Nephrol. 34, 975–991 (2019).
Wei, P. Z. & Szeto, C. C. Mitochondrial dysfunction in diabetic kidney disease. Clin. Chim. Acta 496, 108–116 (2019).
Han, Y. et al. Reactive oxygen species promote tubular injury in diabetic nephropathy: the role of the mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol. 16, 32–46 (2018).
Sun, Y. et al. JAK2/STAT3 involves oxidative stress-induced cell injury in N2a cells and a rat MCAO model. Int. J. Neurosci. 130, 1142–1150 (2020).
Zhang, M., Wang, C., Cai, H. L., Wen, J. & Fang, P. F. Licorice extracts attenuate nephrotoxicity induced by brucine through suppression of mitochondria apoptotic pathway and STAT3 activation. Curr. Med. Sci. 39, 890–898 (2019).
Du, C. et al. SOCS-1 is involved in TNF-alpha-induced mitochondrial dysfunction and apoptosis in renal tubular epithelial cells. Tissue Cell. 49, 537–544 (2017).
Acknowledgements
The authors thank the patients and their families for participating in this study. We also thank BerryGenomics (Bei **g, China) for providing mass spectrometric and RNA-seq-related technical assistance. This study was supported by the National Natural Science Foundation of China (82170598, 82070738, 81970403 and 82000427), Natural Science Foundation of Hunan Province (2020JJ5785 and 2021JJ31015) and Research Project of the Hunan Provincial Health Commission (202103012102, 202103050563, and 202104022248).
Author information
Authors and Affiliations
Contributions
L.-L.F., R.D., and J.-S.L. wrote the draft of the manuscript and performed the cell and molecular experiments; J.-S.L. enrolled the patients’ samples; J.-Y.J. and C.-Y.W. performed HE, PAS staining, and IHC; Y.D. performed animal feeding; W-X.H. performed cell culture; and R.-Q.Y. and R.X. designed and supported the project. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fan, LL., Du, R., Liu, JS. et al. Loss of RTN3 phenocopies chronic kidney disease and results in activation of the IGF2-JAK2 pathway in proximal tubular epithelial cells. Exp Mol Med 54, 653–661 (2022). https://doi.org/10.1038/s12276-022-00763-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s12276-022-00763-7
- Springer Nature Limited