Abstract
A new soluble conjugated polymer, poly[{9-(4-methoxyphenyl)-9H-carbazole}(9,9-dioctylfluorene)(2,5-diphenyl-1,3,4-oxadiazole)](PCFO), was synthesized through the Suzuki coupling reaction. The absolute fluorescence quantum yield of PCFO was measured using the integrating sphere of a photoluminescence spectrofluorometer, and changed from 49.1% for a dilute tetrahydrofuran solution to 16.2% for a thin film due to the existence of a strong fluorescence quenching effect in the solid state. The HOMO–LUMO bandgap (3.07 eV) calculated from the electrochemical measurement is nearly identical to the optical bandgap (3.06 eV) estimated from the ultraviolet/visible absorption onset data. This film was attached to aluminum and indium–tin oxide contacts to fabricate a memory device with typical bistable electrical switching, nonvolatile write-once read-many-times memory performance, a turn-on voltage of ∼−2.3 V and an ON/OFF ratio of ∼105. Degradation of the current density was observed for neither the ON nor OFF states after one hundred million continuous read cycles, which indicates that both states were insensitive to read cycles.
Similar content being viewed by others
Introduction
Polymer memories are proposed to revolutionize electrical applications by providing extremely inexpensive, lightweight and transparent modules that can be fabricated onto plastic, glass or the top layer of CMOS hybrid integration circuits,1, 2 and have been identified as an emerging memory technology by the International Technology Roadmap for Semiconductors since year 2005.3 Rather than encoding ‘0’and ‘1’ as a charge stored in the cell of a silicon device, polymer memory stores data in an entirely different form, such as in a high- and low-conductivity response to an applied voltage.4 The molecular structure of polymers can be tailored by functionalizing them with electron donors (D) and acceptors (A) of different strengths, spacer moieties of different steric effects for the electroactive pendant groups, or nanostructured electroactive materials, to induce different memory behaviors in simple metal/polymer/metal devices.5, 6 Very recently, Liu and Chen7 highlighted the recent developments in D–A polymers for resistive switching memory device applications including conjugated polymers, functional polyimides, nonconjugated pendent polymers and polymer composites. D/A polymer materials (usually carbazole or fluorene donors) with moderate (or incomplete) charge transfer (CT) are likely to exhibit switching behaviors by external voltage bias. The strong dipole moment of molecule helps to sustain the conductive CT state and thus presents a nonvolatile behavior. The optimization of donor/acceptor ratios and the depth of trap** barrier define the volatile/nonvolatile nature.7
In our previous work, we designed a series of D–A-type functional polymers.8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 The device based on conjugated poly[9,9–bis(4-diphenylaminophenyl)-2,7-fluorene] donors covalently bridged with Disperse Red 1 acceptors (DR1-PDPAF-DR1) exhibited an accessible rewritable memory characteristic.10, 17 The excitation of donor promoted CT to the conjugated channel, finally the electrons were at LUMO1 or further at LUMO2 and gave rise to a conductive charge separation state. Polyfluorene-based copolymers containing the side-chain electron-rich TPA and electron-poor cyano substituents exhibited a typical write-once read-many-times (WORM) memory characteristic as the positive charges on the TPA or fluorene moieties were rapidly consumed by the cyano groups as a result of switching operation.14 TPA-based conjugated polyazomethine covalently grafted with graphene oxide (TPAPAM-GO) was directly used to fabricate nonvolatile rewritable memory devices.11 Electrons transmitted from the HOMO of the hole-transporting polymer TPAPAM into LUMO of graphene layer by intramolecular CT interaction established charge transport pathways and switched the device from the OFF to ON state. The effective electron delocalization in graphene nanosheets might stabilize the CT state of TPAPAM-GO, leading to a nonvolatile nature. The electrical bistable switching behavior of the pendent D–A polymer devices was affected by both the electronic structure and surface morphology. The electron-withdrawing DR1-functionalized PVK copolymers (PVDR) were fabricated for memory device as the active layer sandwiched between ITO and Al electrode.13 The PVDR film displayed helical columnar stacks with large grain sizes, whereas a non-aggregated PVDR film exhibited an amorphous morphology with a smaller grain size. Both the PVDR devices showed WORM performance with an ON/OFF current ratio of 105. The asymmetric distribution of the HOMO and LUMO level could stabilize the conductive CT state of the PVDR copolymer leading to the nonvolatile and non-erasable behavior. Besides, the stability of the nano-aggregated PVDR device was much better than that of the non-aggregated PVDR device. The surface defects and large numbers of the grain boundaries in the amorphous PVDR sample acted as charge-trap** centers. An effective charge transport channel and better polymer/metal contacts of the self-assembled smooth PVDR film contributed to the more stable ON state current in the corresponding device. Similar WORM devices were constructed from two PVK derivatives with the pendent azobenzene chromopheres and terminal electron acceptor moieties (–NO2 or –CN).15 Electric field-formed high dipole moments originally from increased charge separation upon CT led to the nonvolatile behavior.
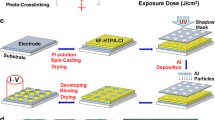
For polymer memory materials, it is very important and profitable to achieve a more balanced charge transport by integrating both electron acceptors and donors into the polymer. The electron-deficient 2,5-diphenyl-1,3,4-oxadiazole (OXD) and its derivatives have been recognized as excellent electron acceptors because of their good chemical, thermal and optical stabilities, as well as the high fluorescence quantum yields.20 Usually, the OXD units are covalently bonded to emissive polymers or small molecules to enhance the electron injection and transport in the emission layer.21, 22, 23, 24, A new conjugated polymer, PCFO, was synthesized through a Suzuki coupling reaction using tetrakis-(triphenylphosphine)palladium(0) as the catalyst. This copolymer exhibited good solubilities in several common organic solvents and good thermal stability. The absolute fluorescence quantum yield of PCFO changed from 49.1% for a dilute THF solution to 16.2% for a thin film due to the existence of a strong fluorescence quenching effect in the solid state. The HOMO, LUMO, IP and EA values experimentally estimated from the onset of the redox potentials were −5.84, −2.77, 6.10 and 3.03 eV, respectively. The HOMO–LUMO bandgap (3.07 eV) calculated from the electrochemical measurement is nearly identical to the optical bandgap (3.06 eV) estimated from the UV/Vis absorption onset data. A WORM memory device was fabricated by spin coating a PCFO solution as the active layer sandwiched between aluminum electrode and an indium–tin oxide electrodes. This device showed very good memory performance, with an ON/OFF current ratio of ∼105, which is high enough to provide a low misreading rate through the precise control of the ON and OFF states. Intramolecular CT might be responsible for the electrical conductivity switching and memory effects of this polymer. Synthesis of PCFO.Conclusion

References
Kuang, Y., Huang, R., Tang, Y., Ding, W., Zhang, L. & Wang, Y. Flexible single-component-polymer resistive memory for ultrafast and highly compatible nonvolatile memory applications. IEEE. Electr. Device Lett. 31, 758–760 (2010).
Song, Y., Ling, Q. D., Zhu, C., Kang, E. T., Chan, D. S. H., Wang, Y. H. D. & Kwong, L. Memory performance of a thin-film device based on a conjugated copolymer containing fluorene and chelated europium complex. IEEE Electr. Device Lett. 27, 154–156 (2006).
Emerging research devices. In: International Technology Roadmap for Semiconductors (ITRS) 2005 edn 1–70 (Semiconductor Industry Association, International Sematech, Austin, TX, 2005).
Ling, Q. D., Zhu, C. X., Chan, D. S. H., Kang, E. T. & Neoh, K. G. in Encyclopedia of Nanoscience and Nanotechnology 2nd edn (ed. Nalwa, H.S.) (American Scientific Publishers, Stevenson Ranch, CA, USA, 2007).
Ling, Q. D., Liaw, D. J., Teo, E. Y. H., Zhu, C. X., Chan, D. S. H., Kang, E. T. & Neoh, K. G. Polymer memories: bistable electrical switching and device performance. Polymer 48, 5182–5201 (2007).
Ling, Q. D., Liaw, D. J., Zhu, C., Chan, D. S. H., Kang, E. T. & Neoh, K. G. Polymer electronic memories: materials, devices and mechanisms. Prog. Polym. Sci. 33, 917–978 (2008).
Liu, C. L. & Chen, W. C. Donor–acceptor polymers for advanced memory device applications. Polym. Chem. 2, 2169–2174 (2011).
Lin, Y., EI-Khouly, M. E., Chen, Y., Supur, M., Gu, L., Li, Y. & Fukuzumi, S. A New cyanofluorene-triphenylamine copolymer: synthesis and photoinduced intramolecular electron transfer processes. Chem. Eur. J. 15, 10818–10824 (2009).
Chen, Y., El-Khouly, M. E., Zhuang, X. D., He, N., Araki, Y., Lin, Y. & Ito, O. Synthesis and photoinduced electron-transfer process of a novel triphenylamine-substituted polyfluorene–C60 triad. Chem. Eur. J. 13, 1709–1714 (2007).
El-Khouly, M. E., Chen, Y., Zhuang, X. D. & Fukuzumi, S. Long-lived charge-separated configuration of a push-pull archetype of disperse red 1 end-capped poly[9,9-bis(4-diphenylaminophenyl)fluorene]. J. Am. Chem. Soc. 131, 6370–6371 (2009).
Zhuang, X. D., Chen, Y., Liu, G., Li, P. P., Zhu, C. X., Kang, E. T., Noeh, K. G., Zhang, B., Zhu, J. H. & Li, Y X. Conjugated-polymer-functionalized graphene oxide: synthesis and nonvolatile rewritable memory effect. Adv. Mater. 22, 1731–1735 (2010).
Liu, G., Zhuang, X. D., Chen, Y., Zhang, B., Zhu, J. H., Zhu, C. X., Neoh, K. G. & Kang, E. T. Bistable electrical switching and electronic memory effect in a solution-processable graphene oxide-donor polymer complex. Appl. Phys. Lett. 95, 253301/1–253301/3 (2009).
Zhuang, X. D., Chen, Y., Liu, G., Zhang, B., Neoh, K. G., Kang, E. T., Zhu, C. X. & Li, Y. X. Preparation and memory performance of a nanoaggregated dispersed red 1-functionalized poly( N-vinylcarbazole) film via solution-phase self-assembly. Adv. Funct. Mater. 20, 2916–2922 (2010).
Zhuang, X. D., Chen, Y., Li, B. X., Ma, D. G., Zhang, B., Li, Y. X. & He, N. Polyfluorene-based push-pull type functional materials for write- once- read- many -times memory devices. Chem. Mater. 22, 4455–4461 (2010).
Liu, G., Zhang, B., Chen, Y., Zhu, C. X., Zeng, L. J., Chan, D. S. H., Neoh, K. G. & Kang, E. T. Electrical conductivity switching and memory effects in poly(N-vinylcarbazole) derivatives with pendant azobenzene chromophores and terminal electron acceptor moieties. J. Mater. Chem. 21, 6027–6033 (2011).
Zhang, B., Liu, Y. L., Chen, Y., Neoh, K. G., Li, Y. X., Zhu, C. X., Tok, E. S. & Kang, E. T. Nonvolatile rewritable memory effects in graphene oxide functionalized by conjugated polymer containing fluorene and carbazole units. Chem. Eur. J. 17, 10304–10311 (2011).
Ling, Q. D., Kang, E. T., Neoh, K. G., Chen, Y., Zhuang, X. D., Zhu, C. & Chan, D. S. H. Thermally stable polymer memory devices based on a ∂-conjugated triad. Appl. Phys. Lett. 92, 143302/1–143302/3 (2008).
Zhang, B., Chen, Y., Zhuang, X. D., Liu, G., Yu, B., Kang, E. T. & Li, Y X. Poly(N-vinylcarbazole) chemically modified graphene oxide. J. Polym. Sci. A: Polym. Chem. 48, 2642–2649 (2010).
Zhang, B., Chen, Y., Xu, L. Q., Zeng, L.J., He, Y., Kang, E.T. & Zhang, J.J. Growing poly(N-vinylcarbazole) from the surface of graphene oxide via RAFT polymerization. J. Polym. Sci. A: Polym. Chem. 49, 2043–2050 (2011).
Schultz, B., Bruma, M. & Brehmer, L. Aromatic poly(1,3,4-oxadiazoe)s as advanced materials. Adv. Mater. 9, 601–613 (1997).
Zhan, X. W., Liu, Y. Q., Wu, X., Wang, S. & Zhu, D. B. New series of blue-emitting and electron-transporting copolymers based on fluorene. Macromolecules 35, 2529–2537 (2002).
Ding, J. F., Day, M., Robertson, G. & Roovers, J. Synthesis and characterization of alternating copolymers of fluorene and oxadiazole. Macromolecules 35, 3474–3483 (2002).
Oyston, S., Wang, C. S., Hughes, G., Batsanov, A. S., Perepichka, I. F., Bryce, M. R., Ahn, J. H., Pearson, C. & Petty, M. C. New 2,5-diaryl-1,3,4-oxadiazole-fluorene hybrids as electron transporting materials for blended-layer organic light emitting diodes. J. Mater. Chem. 15, 194–203 (2005).
Kamtekar, K. T., Wang, C. S., Bettington, S., Batsanov, A. S., Perepichka, I. F., Bryce, M. R., Ahn, J. H., Rabinal, M. & Petty, M. C. New electroluminescent bipolar compounds for balanced charge-transport and tuneable colour in organic light emitting diodes: triphenylamine oxadiazole-fluorene triad molecules. J. Mater. Chem. 16, 3823–3835 (2006).
Guo, X., Cheng, Y. X., **e, Z. Y., Geng, Y. H., Wang, L. X., **g, X. B. & Wang, F. S. Fluorene-based copolymers containing dinaphtho-s-indacene as new building blocks for high-efficiency and color-stable blue LEDs. Macromol. Rapid. Commun. 30, 816–825 (2009).
Wu, F. I., Reddy, D. S., Shu, C. F., Liu, M. S. & Jen, A. K. Y. Novel oxadiazole-containing polyfluorene with efficient blue electroluminescence. Chem. Mater. 15, 269–274 (2003).
Shu, C. F., Dodda, R., Wu, F. I., Liu, M. S. & Jen, A. K. Y. Highly efficient blue-light-emitting diodes from polyfluorene containing bipolar pendant groups. Macromolecules 36, 6698–6703 (2003).
Fang, Y. K., Liu, C. L. & Chen, W. C. New random copolymers with pendant carbazole donor and 1,3,4-oxadiazole acceptor for high performance memory device applications. J. Mater.Chem. 21, 4778–4786 (2011).
Ling, Q. D., Wang, W., Song, Y., Zhu, C. X., Chan, D. S. H., Kang, E. T. & Neoh, K. G. Bistable electrical switching and memory effects in a thin film of copolymer containing electron donor-acceptor moieties and europium complexes. J. Phys. Chem. B. 110, 23995–24001 (2006).
Choi, S., Hong, S. H., Cho, S. H., Park, S., Park, S. M., Kim, O. & Ree, M. High-performance programmable memory devices based on hyperbranched copper phthalocyanine polymer thin films. Adv. Mater. 20, 1766–1771 (2008).
Möller, S., Perlov, C., Jackson, W., Taussig, C. & Forrest, S. R. A polymer/semiconductor write-once read-many-times memory. Nature 426, 166–169 (2003).
Mukherjee, B. & Pal, A. J. Write-once-read-many-times (WORM) memory applications in a monolayer of donor/acceptor supramolecule. Chem. Mater. 19, 1382–1387 (2007).
Liu, G., Ling, Q. D., Teo, E. Y. H., Zhu, C. X., Chan, D. S. H., Neoh, K. G. & Kang, E. T. Electrical conductance tuning and bistable switching in poly(N-vinylcarbazole)-carbon nanotube composite films. Acs Nano 3, 1929–1937 (2009).
Kim, D. M., Park, S., Lee, T. J., Hahm, S. G., Kim, K., Kim, J. C., Kwon, W. & Ree, M. Programmable permanent data storage characteristics of nanoscale thin films of a thermally stable aromatic polyimide. Langmuir 25, 11713–11719 (2009).
Möller, S., Forrest, S. R., Perlov, C., Jackson, W. & Taussig, C. Electrochromic conductive polymer fuses for hybrid organic/inorganic semiconductor memories. J. Appl. Phys. 94, 7811–7819 (2003).
Smith, S. & Forrest, S. R. A low switching voltage organic-on-inorganic heterojunction memory element utilizing a conductive polymer fuse on a doped silicon substrate. Appl. Phys. Lett. 84, 5019–5021 (2004).
Xu, X., Register, R. A. & Forrest, S. R. Mechanisms for current-induced conductivity changes in a conducting polymer. Appl. Phys. Lett. 89, 1421091 (2006).
Li, L., Ling, Q. D., Zhu, C. X., Chan, D. S. H., Kang, E. T. & Neoh, K. G. Bilayer memory device based on a conjugated copolymer and a carbon nanotube/polyaniline composite. J. Electrochem. Soc. 155, H205–H209 (2008).
Wang, B. C., Liao, H. R., Yeh, H. C., Wu, W. C. & Chen, C. T. Theoretical investigation of stokes shift of 3,4-diaryl-substituted maleimide fluorophores. J. Lumin. 113, 321–328 (2005).
Mann, C. K. & Barnes, K. K. Electrochemical Reactions in Nonaqueous Systems (Marcel Dekker, New York, 1990).
Pei, J., Ni, J., Zhou, X., Cao, X. & Lai, Y. Regioregular head-to-tail oligothiophene-functionalized 9,9’-spirobifluorene derivatives. 2. NMR characterization, thermal behaviors, and electrochemical properties. J. Org. Chem. 67, 8104–8113 (2002).
Kumaresan, D., Thummel, R. P., Bura, T., Ulrich, G. & Ziessel, R. Color tuning in new metal-free organic sensitizers (bodipys) for dye-sensitized solar cells. Chem. Eur. J. 15, 6335–6339 (2009).
Chen, Z. K., Huang, W., Wang, L. H., Kang, E. T., Chen, B. J., Lee, C. S. & Lee, S. T. A family of electroluminescent silyl-substituted poly(p-phenylenevinylene)s: synthesis, characterization, and structure-property relationships. Macromolecules 33, 9015–9025 (2000).
Yang, C. & Jenekhe, A. S. Conjugated aromatic polyimines. 2. synthesis, structure, and properties of new aromatic polyazomethines. Macromolecules 28, 1180–1196 (1995).
Iosip, M. D., Destri, S., Pasini, M., Porzioc, W., Pernstich, K. P. & Batlogg, B. New dithieno[3,2-b:2’,3’-d]thiophene oligomers as promising materials for organic field-effect transistor applications. Synth. Met. 146, 251–257 (2004).
Frommer, J. E. & Chance, R. R. in Encylopedia of Polymer Science and Engineering (eds Grayson, M. & Kroschwitz, J.) 2nd edn, Vol. 5, 462–507 (John Wiley & Sons, Inc., New York, USA, 1986).
Sun, Y. M., Liu, Y. Q. & Zhu, D. B. Advances in organic field-effect transistors. J. Mater. Chem. 15, 53–65 (2005).
Facchetti, A. Semiconductors for organic transistors. Mater. Today. 10, 28–37 (2007).
Acknowledgements
We are grateful for the financial support of the National Natural Science Foundation of China (21074034), the Ministry of Education of China (309013), the Fundamental Research Funds for the Central Universities, the State Key Laboratory of ASIC & System of Fudan University (11KF007), the Shanghai Municipal Educational Commission for the Shuguang fellowship (08GG10) and the Shanghai Eastern Scholarship.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zeng, LJ., Liu, G., Zhang, B. et al. Synthesis and memory performance of a conjugated polymer with an integrated fluorene, carbazole and oxadiazole backbone. Polym J 44, 257–263 (2012). https://doi.org/10.1038/pj.2011.114
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pj.2011.114
- Springer Nature Limited