Abstract
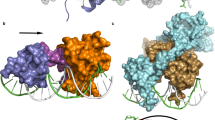
Bacteriophage λ has for many years been a model system for understanding mechanisms of gene regulation1. A ‘genetic switch’ enables the phage to transition from lysogenic growth to lytic development when triggered by specific environmental conditions. The key component of the switch is the cI repressor, which binds to two sets of three operator sites on the λ chromosome that are separated by about 2,400 base pairs (bp)2,3. A hallmark of the λ system is the pairwise cooperativity of repressor binding4. In the absence of detailed structural information, it has been difficult to understand fully how repressor molecules establish the cooperativity complex. Here we present the X-ray crystal structure of the intact λ cI repressor dimer bound to a DNA operator site. The structure of the repressor, determined by multiple isomorphous replacement methods, reveals an unusual overall architecture that allows it to adopt a conformation that appears to facilitate pairwise cooperative binding to adjacent operator sites.




Similar content being viewed by others
References
Ptashne, M. A Genetic Switch (Cold Spring Harbor Press, Cold Spring Harbor, New York, 2004)
Maniatis, T. & Ptashne, M. Multiple repressor binding at the operators in bacteriophage lambda. Proc. Natl Acad. Sci. USA 70, 1531–1535 (1973)
Maniatis, T. et al. Recognition sequences of repressor and polymerase in the operators of bacteriophage lambda. Cell 5, 109–113 (1975)
Johnson, A. D., Meyer, B. J. & Ptashne, M. Interactions between DNA-bound repressors govern regulation by the lambda phage repressor. Proc. Natl Acad. Sci. USA 76, 5061–5065 (1979)
Dodd, I. B., Perkins, A. J., Tsemitsidis, D. & Egan, J. B. Octamerization of lambda CI repressor is needed for effective repression of P(RM) and efficient switching from lysogeny. Genes Dev. 15, 3013–3022 (2001)
Pabo, C. O. & Lewis, M. The operator-binding domain of λ repressor: structure and DNA recognition. Nature 298, 443–447 (1982)
Bell, C. E., Frescura, P., Hochschild, A. & Lewis, M. Crystal structure of the lambda repressor C-terminal domain provides a model for cooperative operator binding. Cell 101, 801–811 (2000)
Whipple, F. W., Kuldell, N. H., Cheatham, L. A. & Hochschild, A. Specificity determinants for the interaction of lambda repressor and P22 repressor dimers. Genes Dev. 8, 1212–1223 (1994)
Pabo, C. O., Sauer, R. T., Sturtevant, J. M. & Ptashne, M. The lambda repressor contains two domains. Proc. Natl Acad. Sci. USA 76, 1608–1612 (1979)
Jordan, S. R. & Pabo, C. O. Structure of the lambda complex at 2.5 Å resolution: details of the repressor–operator interactions. Science 242, 893–899 (1988)
Sauer, R. T., Ross, M. J. & Ptashne, M. Cleavage of the lambda and P22 repressors by recA protein. J. Biol. Chem. 257, 4458–4462 (1982)
Little, J. W. Autodigestion of lexA and phage lambda repressors. Proc. Natl Acad. Sci. USA 81, 1375–1379 (1984)
Slilaty, S. N. & Little, J. W. Lysine-156 and serine-119 are required for LexA repressor cleavage: a possible mechanism. Proc. Natl Acad. Sci. USA 84, 3987–3991 (1987)
Roland, K. L., Smith, M. H., Rupley, J. A. & Little, J. W. In vitro analysis of mutant LexA proteins with an increased rate of specific cleavage. J. Mol. Biol. 228, 395–408 (1992)
Luo, Y. et al. Crystal structure of LexA: a conformational switch for regulation of self-cleavage. Cell 106, 585–594 (2001)
Bell, C. E. & Lewis, M. Crystal structure of the lambda repressor C-terminal domain octamer. J. Mol. Biol. 314, 1127–1136 (2001)
Revet, B., von Wilcken-Bergmann, B., Bessert, H., Barker, A. & Muller-Hill, B. Four dimers of lambda repressor bound to two suitably spaced pairs of lambda operators form octamers and DNA loops over large distances. Curr. Biol. 9, 151–154 (1999)
Terwiliger, T. C. & Berendzen, J. Automated structure solution for MIR and NAD. Acta Crystallogr. D 55, 849–861 (1999)
Winn, M. D. An overview of the CCP4 project in protein crystallography: an example of a collaborative project. J. Synchrotron Radiat. 10, 23–25 (2003)
Chang, G. & Lewis, M. Molecular replacement using genetic algorithms. Acta Crystallogr. D 53, 279–289 (1997)
Brunger, A. T. et al. Crystallography & NMR System: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Otwinowski, Z. & Minor, W. Processing of x-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Emsley, P. & Cowtan, K. COOT: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Acknowledgements
We thank S. Garrity for reading this manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary information
The file contains Supplementary Table 1 and Supplementary Figures 1-2 with Legends. (PDF 3412 kb)
Rights and permissions
About this article
Cite this article
Stayrook, S., Jaru-Ampornpan, P., Ni, J. et al. Crystal structure of the λ repressor and a model for pairwise cooperative operator binding. Nature 452, 1022–1025 (2008). https://doi.org/10.1038/nature06831
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06831
- Springer Nature Limited
This article is cited by
-
Whole genome sequencing identifies an allele responsible for clear vs. turbid plaque morphology in a Mycobacteriophage
BMC Microbiology (2020)
-
Biophysical ambiguities prevent accurate genetic prediction
Nature Communications (2020)
-
The engineering principles of combining a transcriptional incoherent feedforward loop with negative feedback
Journal of Biological Engineering (2019)
-
Intracellular directed evolution of proteins from combinatorial libraries based on conditional phage replication
Nature Protocols (2017)
-
Prophages mediate defense against phage infection through diverse mechanisms
The ISME Journal (2016)