Abstract
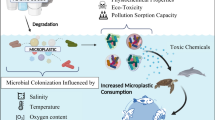
To unveil the interaction of radioactive Cs and Sr with polyethylene-derived microplastics in the marine environment, a mesocosm study was conducted in a stepwise manner by investigating the uptake capability of microplastics at three different stages: pristine, radiation-exposed, and marine-exposed microplastics. The study demonstrates that the physio-chemical properties of microplastics undergo significant alterations upon exposure to the environment, leading to the emergence of biofilm formation upon marine exposure, while radiation exposure induces surface roughness and cracks. Biofilm growth enhances the uptake of radionuclides by microplastics. However, the growth of biofilms increases the density of microplastics through aggregation, leading to a buoyancy transition from positive to negative buoyancy. Various interaction mechanisms, such as electrostatic, ion–dipole, and physical diffusion interactions, were identified as important mechanisms playing key roles in radionuclide binding to polyethylene-derived microplastics. Despite the significantly lower apparent distribution coefficients observed for radio Cs (in the range of 7.3–23.6 L/kg) and Sr (in the range of 4.3–8.06 L/kg) in the marine system, typically 2–3 orders of magnitude lower than those on marine suspended sediment, this study offers compelling evidence that microplastics in marine environments are capable of sequestering radio Cs and Sr. Consequently, microplastics can potentially accumulate these radionuclides, highlighting their role as potential reservoirs as well as vectors of radionuclides in marine environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Microplastics (MPs), particles with a largest dimension of less than 5 mm, have become a global environmental issue, owing to their widespread distribution and escalating accumulation in marine ecosystems. These minute plastic fragments originate from both primary and secondary sources. Primary MPs, intentionally designed to be less than 5 mm, are present in personal care products, plastic pellets used in industrial manufacturing, etc. while secondary MPs result from the breakdown of larger plastic items in the environment [1, 2]. Marine environments are particularly impacted by MPs pollution due to human activities such as littering, run off from urban environment, improper waste disposal, plastic waste from fishing, ship** and offshore activities and inadequate recycling systems. Despite surface-level assessments suggesting scant plastic debris, the staggering annual influx of approximately 4.8 million tonnes into marine environment hints at substantial sub-surface deposition [2]. The fate of MPs is influenced by factors such as size, shape, density, and environmental conditions, including coastal currents, drift, river outflow, wind, and rotational ocean currents. As a result, MPs undergo a dynamic journey, moving from coastal areas to deeper ocean regions. This trajectory is shaped by a complex interplay of physical, chemical, and biological forces, which contribute to processes like weathering and degradation [3, 4]. As a result of weathering in marine systems, MPs release additives and unreacted monomers. This poses an insidious threat to aquatic life through ingestion by a diverse array of marine organisms, ranging from fish and mussels to zooplankton, seabirds, sand hoppers, and worms. Such ingestion leads to alterations in behaviour, physiological functions, oxidative stress, gastrointestinal tract damage, DNA damage, tissue damage, and more [5]. The effect can also be observed at higher tropic levels such as human due to tropic transfer.
MPs also act as a vector due to their ability to sorb wide range of contaminants. In aquatic environments, the surfaces of MPs promote the formation of biofilms (or biofouling), facilitating the rapid colonization by algae, bacteria, fungi, and protozoa and as well as the adherence of dissolved organic carbon (DOC) and inorganic matter to the MPs’ surfaces [6,7,8,9]. This process also influences the ecological dynamics of MPs by altering the density of MP particles, potentially affecting their transport and fate. Biofilm formation enhances the capacity of MPs to uptake contaminants, as a result biofilm developed MPs acts as a vector for a wide range of contaminants [10, 11]. Understanding these interactions is essential for comprehensively assessing the ecological implications of microplastic pollution in marine ecosystems. Plenty of literature is available on MPs, such as: inventory of MPs in marine ecosystems, ecological toxicity, biofilm development on MPs [12,13,14]. A few studies are also available on the MPs as vectors for contaminants, which are mainly focused on non-radiological contaminants such as stable heavy metals and organic contaminants [15]. However, there is a scarcity of published data on the interaction of radionuclides with MPs in both terrestrial and aquatic environments. Among the artificial radioactive isotopes released into the marine environment, Cs-137 and Sr-(89 + 90) are considered particularly significant [16,17,18]. These fission products, notable for their high fission yield, are the primary fission products generated in nuclear reactors. Both Sr and Cs radionuclides present risks to humans and non-human biota since they emit beta and gamma radiation. Sr accumulates in bones, while Cs accumulates in muscles. As reported, nuclear weapon testing alone resulted in the deposition of 950 PBq of Cs-137 in the world’s oceans [19]. The Chernobyl and Fukushima nuclear accidents released approximately 85–200 PBq and 12–15 PBq of Cs-137 into the sea, respectively [20, 21]. Additionally, the Fukushima nuclear accidents resulted in the release of around 900 TBq of Sr-90 into the sea [22]. Johansen et al. [23] reported preliminary findings on the adsorption of Cs-134 and Sr-85 on MP surfaces with biofilm in freshwater and estuarine system [23]. Ioannidis et al. [24] evaluated Kd of U-232 at MPs and water (Deionised water) interface [24]. These studies provide a preliminary information about sorption of radionuclides with MP in freshwater system. However, study in the marine environment is very important as marine water receives radionuclides through routine as well as accidental releases from nuclear facilities such as spent fuel reprocessing facilities, purge water from fuel storage ponds, power plants etc., containing a range of actinides and fission products [17, 25, 26].
The ability of MPs to sorb various pollutants poses a significant ecological threat, with MP inventories increasing in marine environments daily [25, 27,28,29,30,31]. Additionally, the expansion of the nuclear energy industry leads to more radioactive waste entering the marine system annually [32]. Consequently, it is crucial for radiological impact assessments to investigate the interactions between radionuclides and both MPs and biofilm-developed MPs to address radiological implications. Therefore, understanding the interactions between radionuclides and MPs in marine environments is crucial. Thus, the primary objective of this research is to study the Cs-137 and Sr-85 uptake capability of polyethylene-derived MPs in marine environment at different stages of weathering and derive a plausible interaction mechanism by considering the physio-chemical properties of MPs (including those with biofilm) and the speciation of Cs-137 and Sr-85 in the marine system.
2 Materials and methods
2.1 MPs sample preparation
Bags made of high and low-density polyethylene (HDPE and LDPE, respectively) were purchased from the store and cut into film pieces with an area of less than 5 mm2. Then the samples from each type (HDPE and LDPE) were divided into two parts, respectively. The first group of both HDPE and LDPE were considered “pristine (virgin). To mimic the surface characteristics of plastics that have been damaged due to exposure to the environment for several years, the second part was exposed to radiation (1.17 MeV and 1.33 MeV, 60Co source) in a γ-chamber.
2.2 Exposure to marine environment
In marine environments, MPs undergo various forms of weathering, including physical, chemical, and biological processes. These weathering mechanisms lead to modifications in surface morphology and the formation of additional functional groups. Changes in surface morphology brought about by weathering are an important indicator of environmental exposure. Therefore, to simulate the, natural weathering of MPs in marine environment, a mesocosm study was designed. Microplastic (MP) samples (4 g of pristine and irradiated) were sealed in nylon mesh bags (mesh size: 10 µm) and then transferred to high impact resistance polycarbonate (PC) container of 0.5 L capacity (cuboidal form) with openings (with a diameter of about 2.5 mm) on all four sides. A cage made of stainless steel was used to hold the container and the overall design was such that it protected the container from large organisms while allowing the water and suspended materials such as silt and sediment to exchange through the holes. The setup was deployed in the Arabian Sea at a depth of ~ 1 m for a period of 5 months with restricted access. Samples with different codes deployed in the sea are listed in Table 1.
2.3 Pre-treatment of MPs (marine exposed)
After being submerged in the sea for 5 months, the deployed setup carrying MPs (HD3, HD4, LD3, and LD4) was removed from the sea, samples were taken to lab, and kept in same seawater in laminar hood at room temperature till further analysis. Samples were collected from nylon mesh bags and transferred to pre labelled centrifuge tubes and processed as per standard protocol for characterisation [7, 33, 34].
MP samples were washed with sterile seawater (Filtered through a 0.2-micron filter and then autoclaved) 2–3 times to get rid of any adhered particles other than biofilms. Then the samples were processed as per standard procedure for morphological analysis using scanning electron microscope. Washed samples were then air-dried over Whatman AE 98 paper in a petri dish and taken for spectroscopic, hydrophilicity, biofilm and zeta potential analysis.
2.4 Characterisation of MP sample
Spectroscopic characterization: MP compositions and their weathering characteristics were investigated using Fourier-transform infrared spectroscopy. For characterization, cleaned and dried MP samples (HD1 to HD4 and LD1 to LD4) were analysed using Fourier-transform infrared spectroscopy (Alpha, make Bruker, Germany) in ATR mode in the range of 500–4000 cm−1, scan rate 40, and resolution: 4 cm−1. In brief, MP samples were dried using lint-free tissue to remove the moisture. Subsequently, they were placed on the diamond crystal of an attenuated total reflection Fourier-transform infrared (ATR-FTIR) spectrometer to acquire spectra. HD1 and LD1 serve as control samples since they are in their pristine state, free from any alterations. These control samples provide a baseline reference for comparison against the environmental exposed samples. To ensure both reproducibility and result reliability, we recorded spectra of MP samples from each type in triplicates. The acquired FTIR spectra were compared with a comprehensive polymer reference library within specialized software. Additionally, manual inspection was conducted to verify the characteristic peaks of each polymer by cross-referencing with the library and spectra presented in previously published research papers.
Hydrophilicity Analysis: Contact angle is the indicator of the hydrophilicity. Dried MP samples were subjected to contact angle measurement using Data Physics OCA50 (Germany). In brief, the dried sample was placed on the slide surface then the slide containing dried MP sample was mounted in the light path of contact angle meter, to which a water droplet of about ~ 2 μL was dropped with a micro-syringe and remain in contact for 10 s till photographed and ensured that the entire droplet and surrounding surface are visible in the image. Then the static angle of droplet on MP surface was analysed using SCA20 (Ver. 2) software which is typically done by identifying the tangent lines of the droplet and surface interface and calculating the angle between them. Static angle of different types of MPs were carried out in triplicate mode by performing the measurement process for multiple droplets on each sample to ensure accuracy and reproducibility and analysed the collected data to calculate average contact angles and assess the surface properties of the samples. Moreover, to provide comprehensive insights, measurements were carried out for more than three types of MPs, enabling a thorough investigation of their surface characteristics and behaviour.
Surface Morphology: Surface morphological investigation of all the MPs was carried out using scanning electron microscopy (SEM) (Make: Hitachi S-4800 Japan). Since the development of biofilm on MP surfaces is a natural phenomenon, MP samples were processed as per established procedure assuming biofilm developed on MPs [33, 35]. In brief, biofilms were immobilized by submerging the samples in a 2.5% glutaraldehyde solution for 3 h at room temperature. MPs were then thoroughly rinsed with 0.1 M phosphate buffer saline (PBS) and subsequently dehydrated using ethanol to eliminate moisture. Processed samples were fixed on an aluminium stub using a double-sided carbon tape then fixed samples were coated with gold (10 nm). Finally, the gold coated sample was analysed using SEM. To ensure the reproducibility of the experiment, samples were analysed in duplicates for each MP types. This approach involved capturing images of the MP samples at high magnification to visualize their surface morphology and structure.
Estimation of biofilm: Quantitative estimation of biofilm formation on Marine exposed MPs were analysed using UV–Visible spectroscopy (Specord S600 Diode Array Scanning UV/Vis Spectrophotometer, Analytik Jena AG). For absorbance measurement samples were stained with crystal violet (1%) dye as per standard protocol [34, 35]. Amount of biofilm formation is assumed, directly proportionate to absorbance. Briefly, dried MP samples were treated with a 1% crystal violet solution. After incubation for 45 min, excess dye was removed. The samples were then washed three times with 5 mL of sterilized seawater. Following this, they were air-dried for 45 min. Next, the dried samples were placed in centrifuge tubes, and 1 mL of 95% ethanol solution was added to each tube for decolorization. The tubes were incubated for 10 min. The decolorized solutions were then measured for absorbance at 595 nm using UV–visible spectroscopy. The experiment was performed three times to ensure reliability, with an additional ethanol-only treatment serving as a control to account for any background interference.
Measurement of Zeta potential: Zeta potential is an important property, which gives the valuable insight in to the surface charge and stability of the particles (MPs in this study) including optimal aggregation behaviour of the MPs in aquatic environment.
The zeta potential of MPs can impact several processes, including aggregation, adhesion, and interaction with other particles as well as biofilm formation. Zeta potential of MP samples was measured using zeta potential analyser (Stabino Zeta, Germany). MP samples were dispersed into deionised (DI) water and 10 mL of the solution containing MP samples were taken for measurement of zeta potential. Zeta potential measurements were carried out in triplicates for each MP types.
2.5 Physicochemical characterisation of host water
The host seawater was collected from the sample deployment site experience low tides. The collected sample was filtered using 0.45 μm filter. The physicochemical characterization of host water is very important for speciation modelling. Aqua-meter with an Aqua Probe 1000 (UK) was used to measure the pH and Eh of the host water. Major cations such as Na+, K+, Ca2+ and Mg2+ were estimated using Atomic Absorption Spectroscopy (make: GBC, Avanta, France). Major ionic compositions: Cl−, NO3−, SO42− and PO43− were estimated using ion chromatography (ICS 2100) (make Dionex, RFIC, USA). For measurement of carbonate and bicarbonate host water was subjected to titration using Auto titrator (798 MPT Titrino, make: Metrohm, Switzerland).
2.6 Sorption experiment
Standard batch sorption method was adopted to study the interaction of the Cs-137 and Sr-85 on all types of PE MP Samples. Cs-137 level in the host water is very low (~ 0.73 mBq L−1). All the sorption experiments were carried out using unfiltered and filtered (0.45-micron Whatman filter paper) host water (pH of 8.23; seawater) and batch study was carried out separately for Cs-137 and Sr-85. Host water was spiked with Cs-137 and Sr-85 (produced by the Board of Radiation and Isotope Technology, Department of Atomic Energy, Mumbai, India) separately so that the final activities of Cs-137 and Sr-85 in spiked host waters are 2.17 Bq mL−1 and 6.37 Bq mL−1 respectively. Prior to the radioisotope spiking, care was taken to ensure that the spiking would not alter the pH of the host water. To achieve the desired activity levels of 2.17 Bq mL−1 of Cs-137 and 6.37 Bq mL−1 of Sr-85, a minimum volume (0.1 mL) of the higher active solutions (Cs-137: 217 Bq mL−1 and Sr-85: 637 Bq mL−1) was separately added to a conical flask. Host water was then added to make up the volume to 10 mL. This process was implemented to prevent any alteration of the pH of the host water due to dilution. Then, 200 mg of MPs from each MP samples was added separately to the flasks containing the spiked host water. The flasks were agitated in a circular motion (at 60 rpm) for a period of 60 h, assuming that equilibrium would be reached within 22 h (Unpublished). After 60 h, the solution in the flask was separated from the MPs by vacuum filtration using 0.45-micron filter paper. The filtrate was subjected to γ-counting using co-axial HPGe (50% relative efficiency) for 3600 s against an in-house matrix and geometry-matched standards. Sorption on MPs was estimated by taking the difference between the initial activity and the filtrate activity. Each sorption study was carried out in triplicates, and the mean activity was reported. The apparent distribution coefficients (Kd) of Cs-137 and Sr-85 were calculated from the standard equation as mentioned in Eq. 1[36].
where, A0: is the initial added activity (Bq); Ae: is the activity at equilibrium (Bq); V: Volume of host solution (L) and M: mass of MP sample (kg).
2.7 Speciation calculation
To understand the interaction of Cs and Sr with MP samples it is necessary to know the speciation of radionuclides in host water system and identify the dominant species. Hence, speciation of Cs and Sr in seawater is necessary. MINTEQA2 thermodynamic database was used for speciation calculation, taking physicochemical parameters along with ‘Cs and Sr’ concentration as input data.
3 Results and discussions
3.1 Characterisation of host water
Table S1 (Supplementary Material) presents the physicochemical characteristic data of the host seawater. The pH of host water is 8.23, which is close to the oceanic pH (8.3). The redox potential (Eh) ~ + 435 mV shows environment is oxic i.e., continuous air exchange with the atmosphere. Other ionic composition data shows similarity to open ocean compositions.
3.2 MPs characterisation
Figure 1a, b presents the Fourier transform infrared spectroscopy spectra of HDPE and LDPE MPs. The IR spectrum of pristine HDPE (HD1: control group) showed five characteristic peaks. Each corresponding to a different mode of vibration of the methylene group. 2888 cm−1 corresponds to anti-symmetric contraction of –CH2–. 2821 cm−1 corresponds to symmetric contraction of –CH2–. 1457 cm−1 corresponds to shear bending vibration peak of –CH2–. 712 cm−1 corresponds to rocking deformation of -CH2- group in amorphous region and 723 cm−1 corresponds to rocking deformation of –CH2– group in crystalline regions. All five characteristic peaks were also observed in pristine LDPE (LD1), with a minor shift for the anti-symmetric bending vibration peak of –CH2 around 1461 cm−1.
In irradiated HDPE (HD2) samples four additional peaks at 3296 cm−1, 1615 cm−1, 1010 cm−1 and 865 cm−1 corresponding to –OH (hydro peroxide:–O–O–H group may also be possible), –C=O stretching, –C–O stretching and vinylidene group respectively, were recorded. Although the peak intensities of –C=O stretching, –C–O stretching are low, the presence of –C=O and –C–O stretching peaks suggests conclusively that irradiation causes degradation of chain and oxidation which leads to the formation of new aliphatic molecules [33]. In case of irradiated LDPE (LD2), two additional peaks at 1700 cm−1 and 1020 cm−1 were observed compared to LD1, which corresponds to –C=O and –C–O stretching. Hydroxyl and vinylidene group peaks were not recorded. This clearly indicates interaction with radiation alter molecular structure of the plastic materials, which occurs in the natural environment due to background radiation. The observed difference in the IR spectra of HD2 and LD2 could be due to the difference in polymer structure scission [37, 38].
Similar peaks were observed in the IR spectra of marine-exposed HDPE and LDPE samples as are seen in the spectra of irradiated samples, with the exception of an additional peak recorded at 3305 cm−1 for LDPE samples (LD3 and LD4). This corresponds to hydroxy (-OH) and/or hydro-peroxide (–O–O–H) functional group. And small peaks appearing in both HD (3,4) and LD (3, 4) MPs at around 1540–1570, which correspond to amide (II) protein [39]. Figure 1a shows that the peak intensities for the –CH2 stretching vibration were found to decrease from HD1 to HD4, whereas the peak intensities of ‘–C–O,’ ‘–C=O,’ and ‘–O–H’ vary from HD3 > HD4 > HD2. This clearly indicates that formation –C–O group leads to cleavage of the –C–H bond. The simultaneous increase in the peak intensities corresponds to ‘–C=O,’ and ‘–O–H’ stretching vibration also supports that development of ‘-COOH’ group on MP surface. Higher intense peak around 1010 cm−1 in pristine-marine exposed samples may be due to more oxygen accessibility in pristine samples compared to irradiated-marine exposed samples, as irradiation can lead to chain scission, that can induce the crystallinity which in turn reduces the oxygen accessibility [7]. Variation in peak intensities confirms the importance of processes other than background gamma radiation exposure in plastic weathering in aquatic environments, including hydrolysis, mechanical abrasion, photolysis induced by UV radiation, and biodegradation. Secondly, microbial colonization of the MP surface may account for the increase of the ‘–C–O’, ‘–C=O’ and ‘–OH’ stretching peaks in marine-exposed samples [40].
The hydrophilic nature of any material is evaluated by water contact angle (CA). The degree of hydrophilicity decreases with increase in contact angle. Figure 2 presents the image of water droplets on different MPs along with their contact angle captured by contact angle measurement instrument. From the figure, it is clear that, the pristine samples such as HD1 (CA: ~ 930) & LD1 (CA: ~ 970) possess a lower degree of hydrophilicity than irradiated samples HD2 (CA: ~ 860) and LD2 (CA: ~ 890). In addition, the hydrophilicity of all marine-exposed samples: HD3 (CA: ~ 730), HD4 (CA: ~ 630), LD3 (CA: ~ 860) and LD4 (CA: ~ 860) is noticeably higher than that of the pristine and irradiated MPs.
Compared to HDPE, LDPE has a lower hydrophilicity, which is supported by IR data, i.e., an increase in peak intensities of –OH, –CO, and –C=O stretching corresponding to the increase in hydrophilicity. Limited literatures are available on hydrophilicity of MP. Nauendorf et al. [41] reported that hydrophilicity of MP increases with biofilm growth on MP surface [41]. Another investigation found that the film’s hydrophilicity increases and become neutrally buoyant as marine exposure time increased [34]. This increase in hydrophilicity brought on by marine exposure encourages the uptake of both organic and inorganic nutrients, which causes microorganisms to quickly colonize on the surface of MPs [6]. Furthermore, the higher degree of hydrophilicity in irradiated samples (HD2 and LD2) in comparison to pristine samples (HD1 and LD1) suggests that the rise in hydrophilicity of the samples may not be entirely attributable to the development of a biofilm on the MP. The radiation-induced functionalization of polymer chains results in the formation of polar groups on the MP surface, which enhances the hydrophilicity of the samples.
Figure 3 (presents SEM images of MPs) shows that, the surface of pristine samples (HD1 & LD1) is relatively smooth with a few uneven patches. The surface roughness (HD2 & LD2) increased consequent to gamma-irradiation and cracking of MP surface is also observed. This indicates that plastic samples constantly exposed to background radiation caused surface roughness, cracking and fractures. The extent of morphological changes relies on the molecular weight and crosslinking and chain scission of plastics. Rod (3-5 μm) and spherical (Dia: 1–2 μm) shaped particles were observed on the marine exposed samples (HD 3–4 and LD 3–4), which maybe microorganisms. Marine exposure of MPs has been shown to result in microbial colonization followed by biofilm growth on MP surfaces [42]. To confirm the biofilm formation and degree of formation on different MP samples, crystal violet staining data were investigated and quantitatively presented in terms of absorbance (Fig. 4). Investigation shows that, the biofilm growth on different marine exposed samples is in the order of HD4 > LD4 > HD3 > LD3. This shows that, biofilm growth on marine exposed irradiated samples is higher than the un-irradiated samples. Secondly, the growth on HDPE is relatively higher than the LDPE samples. The reason behind the difference in biofilm growth is not clear, but it is likely that irradiation induced change in surface morphology and functionalisation is playing important role.
Aggregation of MPs in aquatic system can alter their transport and fate in the environment, which in turn decide the fate of contaminants. Aggregation behaviour of all the MP samples were investigated in terms of zeta potential and is shown in Fig. 5.
Investigation of Fig. 5 shows that the zeta value for pristine HDPE and LDPE are high and in the range of − 100 mV. However, after irradiation the value drastically falls and is in the range of (− 52 mV) to (− 55 mV). Furthermore, subsequent to marine deployment the zeta potential again falls to − 52 to – 49 mv in case of HDPE and − 47 to − 36 in case of LDPE. This shows that in the marine system tendency of LDPE to aggregate is higher than HDPE. Decrease in zeta potential revealed that, the decrease in the tendency of the plastics to be in suspension in marine environment due to biofilm growth. This supports the fact that marine exposure can cause a transition in MPs' buoyancy states, potentially shifting them from positive buoyancy to neutral or even negative buoyancy [34].
3.3 Sorption behaviour of Cs-137 and Sr-85 on MP
In a real-world scenario, achieving equilibrium in a natural system (at the interface of marine water and MPs) may be a rare occurrence due to temporal variations in radioisotope concentration, temperature, biofilm biomass on MPs, and other factors. As a result, the interaction between radioisotopes and MPs is dynamic in nature. Radioisotopes continuously exchange across the interface between the MPs (including those with biofilm) and seawater. Therefore, we used the term apparent Kd instead of Kd. Table 2 presents the apparent Kd data of Cs-137 and Sr-85 on different MP samples generated using both filtered and unfiltered host sea water, which is also presented graphically in Fig. 6a, b. Data confirmed that the pristine MPs are incapable of sorbing Cs-137 and Sr-85. The sorption capacity of HDPE MPs is relatively higher than the LDPE MPs. In both cases, the apparent Kd value increases from pristine to irradiated marine exposed MPs. This may be attributed to the change in the surface morphology of the MPs due to exposure to environment, i.e., unfiltered host water. The higher apparent Kd for Cs-137 in comparison to the Sr-85 can be correlated to the aqueous complexation as shown in speciation diagram (Figure S1 & S2). The diagram indicates that, Cs-137 remains as Cs+-137 in seawater whereas, Sr-85 form 85SrSO4 species in addition to Sr2+-85. The speciation analysis confirms that, although Cs-137 and Sr-85 possess little tendency to form aqueous complex with chloride, sulphate, nitrate, and carbonates, Sr-85 has relatively higher tendency than Cs-137 for complexation with SO42−, which lowers the Kd of Sr-85. Cs and Sr sorption onto all MPs (except pristine) were measurable in all experimental cases in this study. As MPs, including those with biofilm, have the capacity to uptake Cs-137 and Sr-85, they can serve as vectors for these radioisotopes similar to suspended sediments. Consequently, we conducted a comparison between the apparent distribution coefficient (Kd) data generated in our current study and reported data. Our findings indicate that the apparent Kd values for Cs-137 and Sr-85 are approximately 2–3 orders of magnitude lower than the reported Kd values for sediments, as documented by the International Atomic Energy Agency [36].
Compared to the laboratory data, the reference field data typically have greater apparent Kd due to the influence of processes such as dilution in the water column via tidal exchange etc., that dilute the water component of the Kd equation, which is not considered in laboratory study [43]. Kd for biofilm developed MPs in marine environment reported in this study is relatively lower than Kd value generated for fresh water [23]. Lower value of Kd is anticipated in the marine environment because competition among cations for active sites to sorb is greater in seawater due to the high ionic strength (IS). However, the Kd, value is low, positive adsorption results shows that like sediments MPs and biofilm developed MPs act as a sink for radio Cs and Sr. Since MPs are mobile and buoyant, they may be more important in the open ocean, where they can interact with atmospherically deposited radioisotopes of Cs and Sr without competition from bottom sediments [23].
Multi-correlation analysis among the apparent Kd data and influencing parameters such as contact angle, zeta potential and biofilm biomass revealed that, MPs with higher biofilm growth uptake higher Cs-137 & Sr-85, whereas decrease in contact angle (hydrophilicity) leads to increase in the apparent Kd of both Cs and Sr (See Table S2).
Hydrophobicity exhibits strong negative correlation with biofilm growth (r = − 0.88), Kd (r = ~ − 0.9) but moderately correlated with zeta potential (r = 0.6). Biofilm growth exhibits strong positive correlation with Kd and strong negative correlation (r = ~ 85) with hydrophobicity and moderate negative correlation with Zeta potential (-0.59). Zeta potential exhibits negative correlation with Kd, biofilm growth but moderately correlated with hydrophobicity. This qualitatively indicates that, though biofilm growth leads to increase in the uptake of Cs and Sr it promotes aggregation among the MPs, which may lead to transition of positive buoyancy to negative buoyancy via neutral buoyancy.
3.4 Mechanism of interaction
The binding of contaminants to the MPs and biofilm developed MPs could be attributed to the several mechanisms such as physical diffusion (pore filling), ion-induced dipole interaction, ion–dipole interactions, dipole–dipole interactions, electrostatic interactions etc. [44]. The interaction of metals with the sorbent is generally modelled by statistically fitting sorption kinetics, isotherm, and thermodynamic data to established models. Investigating the interaction of metal ion with MPs and biofilm developed MPs through model fitting is associated with several limitation [45], which raise question on its environmental applicability. Interaction of metals with biofilm developed MPs is very different, as metals can be uptake by microorganism as nutrient through transmembrane transport and metals can be hold up by the extracellular structure of bacteria surface [46]. Even though few studies are available they are mainly controlled laboratory studies and mostly dealt with interaction of metal with MPs without biofilms. Considering the limitations and lack of environmental realism of model derived mechanism, here we have tried to derive plausible mechanism of interaction based on physical and chemical properties of MPs (and biofilm developed MPs) and radio Cs and Sr with supporting information from available literature. The interaction mechanism is pictorially elucidated in Fig. 7.
Interaction with pristine MP(HD1&LD1): Polyethylene (PE) surfaces lack distinct functional groups, thereby suggesting that the uptake of contaminants on pristine MPs (MPs) can only be influenced by ion-induced dipole forces and interactions related to pore filling [47,48,49,50,51,52], which is a physical diffusion process. Random-coil model suggest that, polymer with semicrystalline region possess pore space where the contaminants can be trapped but it again depends on the speciation of the contaminants [45]. From IR spectrum (Fig. 1a, b) it is confirmed that the MPs possesses semi crystalline region i.e., peak at 712 and 723 cm−1 corresponds to the -CH2- rocking deformation peak for amorphous and crystalline region but sorption data clearly indicates negligible uptake of radio Cs and Sr by pristine (HD1 and LD1) MPs. This is attributed to a) low effective charge of Cs+ and Sr2+ ions, resulting from their larger hydrated radii, weakens the ion-induced dipole force, thereby reducing the likelihood of binding to the pristine MP surface, and b) larger hydrated radii of Cs+ and Sr2+ and presence of other species such as SrSO4 (Figure S2, supplementary material) disfavour the uptake due to pore filling. Furthermore, the smooth surface of the pristine MPs, confirmed by SEM analysis also does not favour the physical diffusion of radio Cs and Sr to the cracks on MPs, which often occurs in the weathered MPs.
Interaction with Irradiated MPs (HD2 & LD2): Irradiation of MPs leads to the induction of surface functionalization due to the formation of –OH, –C–O, and –C=O functional groups, as supported by FT-IR data. This increase in the density of oxygen-containing functional groups favours ion–dipole interactions, leading to the uptake of radio Cs+ and Sr2+ by irradiated MPs. This process involves the interaction between the positive charge of Cs+ and Sr2+ ions and the partial negative charge on the oxygen atoms of oxygen-bearing functional groups (–OH, –C–O, and –C=O) present on the MPs [ The study findings indicate that the uptake of Cs-137 and Sr-85 on MPs increases with exposure to marine environments, primarily due to the development of biofilm on the MP surfaces. Multiple interaction mechanisms, such as electrostatic interactions, ion–dipole interactions, and physical diffusion of Cs+-137, Sr2+-85, and 85SrSO4 species into cracks on the surface of the MPs, play an important role in the sequestration of these isotopes by marine-exposed MPs. Sequestration is insignificant in the case of pristine MPs and is noticeable in the case of radiation-weathered MPs via ion-induced dipole and physical diffusion interactions. The apparent Kd data generated in this study can be valuable for predicting the distribution and transport behaviour of Cs-137 and Sr-85 in the marine environment in the presence of MPs, especially for screening radiological impact assessments. The results of this investigation align with the notion that plastic surfaces undergo modifications upon environmental exposure, resulting in a similar sorption behaviour of radio Cs and Sr radionuclides as observed for other metals. Furthermore, the study highlights that though MPs in the marine environment act as a potential reservoir and vector for Cs-137 and Sr-85, the mobility of MPs diminishes in marine systems as a consequence of particle aggregation induced by environmental exposure, and this effect is more prominent in LDPE compared to HDPE.4 Conclusion
Data availability
Sequence data that support the findings of this study are provided within the manuscript and supplementary material file.
References
Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle D, Russell AE. Lost at sea: where is all the plastic? Science. 2004;304:838. https://doi.org/10.1126/science.1094559.
Hoseini M, Bond T. Predicting the global environmental distribution of plastic polymers. Environ Pollut. 2022;300:118966. https://doi.org/10.1016/j.envpol.2022.118966.
Yang H, Chen G, Wang J. Microplastics in the marine environment: sources, fates, impacts and microbial degradation. Toxics. 2021;9(2):41.
Microplastics in the marine environment: A review of their sources, distribution processes, uptake and exchange in ecosystems. Case Studies in Chemical and Environmental Engineering. 2020. https://www.sciencedirect.com/science/article/pii/S2666016420300086.
Hamed M, Soliman HAM, Osman AGM, Sayed AE-DH. Antioxidants and molecular damage in Nile Tilapia (Oreochromis niloticus) after exposure to MP. Environ Sci Pollut Res. 2020;27:14581–8. https://doi.org/10.1007/s11356-020-07898-y.
Oberbeckmann S, Löder MGJ, Labrenz M. Marine microplastic-associated biofilms–a review. Environ Chem. 2015;12(5):551–62.
Rout S, Yadav S, Joshi V, Karpe R, Pulhani V, Kumar AV. Microplastics as vectors of radioiodine in the marine environment: a study on sorption and interaction mechanism. Environ Pollut. 2022;307:119432. https://doi.org/10.1016/j.envpol.2022.119432.
Wetzel RG. Microcommunities and microgradients: linking nutrient regeneration, microbial mutualism, and high sustained aquatic primary production. Netherl J Aquat Ecol. 1993;27:3–9.
Zettler ER, Mincer TJ, Amaral-Zettler LA. Life in the “plastisphere”: microbial communities on plastic marine debris. Environ Sci Technol. 2013;47(13):7137–46. https://doi.org/10.1021/es401288x.
Qiongjie W, Yong Z, Yangyang Z, Zhouqi L, **xiaoxue W, Huijuan C. Effects of biofilm on metal adsorption behavior and microbial community of microplastics. J Hazard Mater. 2022;424:127340. https://doi.org/10.1016/j.jhazmat.2021.127340.
He S, Tong J, **ong W, **ang Y, Peng H, Wang W, Yang Y, Ye Y, Hu M, Yang Z, Zeng G. Microplastics influence the fate of antibiotics in freshwater environments: biofilm formation and its effect on adsorption behavior. J Hazard Mater. 2023;442:130078. https://doi.org/10.1016/j.jhazmat.2022.130078.
Park H, Park B. Review of microplastic distribution, toxicity, analysis methods, and removal technologies. Water. 2021;13(19):2736.
Akdogan Z, Guven B. Microplastics in the environment: a critical review of current understanding and identification of future research needs. Environ Pollut. 2019;254(Pt A):113011. https://doi.org/10.1016/j.envpol.2019.113011.
Sangkham S, Faikhaw O, Munkong N, Sakunkoo P, Arunlertaree C, Chavali M, Mousazadeh M, Tiwari A. A review on microplastics and nanoplastics in the environment: their occurrence, exposure routes, toxic studies, and potential effects on human health. Mar Pollut Bull. 2022;181:113832. https://doi.org/10.1016/j.marpolbul.2022.113832.
Cao Y, Zhao M, **anying M, Song Y, Zuo S, Li H, Deng W. A critical review on the interactions of microplastics with heavy metals: mechanism and their combined effect on organisms and humans. Sci Total Environ. 2021;788:147620. https://doi.org/10.1016/j.scitotenv.2021.147620.
Povinec PP, Hirose K, Aoyama M. Radiostrontium in the western North Pacific: characteristics, behavior, and the Fukushima impact. Environ Sci Technol. 2012;46(18):10356–63. https://doi.org/10.1021/es301997c.
Ray D, Leary P, Livens F, Gray N, Morris K, Law KA, Fuller AJ, Abrahamsen-Mills L, Howe J, Tierney K, Muir G, Law GTW. Controls on anthropogenic radionuclide distribution in the sellafield-impacted eastern irish sea. Sci Total Environ. 2020;743:140765. https://doi.org/10.1016/j.scitotenv.2020.140765.
Ens CC, Resources N, Iv C, Ashraf M, Science E. 1.3. Nuclear Energy. 1–17.
Prăvălie R. Nuclear weapons tests and environmental consequences: a global perspective. Ambio. 2014;43(6):729–44. https://doi.org/10.1007/s13280-014-0491-1.
Kotilainen AT, Kotilainen MM, Vartti V-P, Hutri K-L, Virtasalo JJ. Chernobyl still with us: 137caesium activity contents in seabed sediments from the Gulf of Bothnia, Northern Baltic Sea. Mar Pollut Bull. 2021;172:112924. https://doi.org/10.1016/j.marpolbul.2021.112924.
Aoyama M, Ka**o M, Tanaka T, Sekiyama T, Tsumune D, Tsubono T, Hamajima Y, Inomata Y, Gamo T. 134Cs and 137Cs in the North Pacific Ocean derived from the march 2011 TEPCO Fukushima Dai-Ichi nuclear power plant accident, Japan. Part two: estimation of 134Cs and 137Cs inventories in the North Pacific Ocean. J Oceanogr. 2015. https://doi.org/10.1007/s10872-015-0332-2.
Casacuberta N, Masqué P, Garcia-Orellana J, Garcia-Tenorio R, Buesseler KO. ^90Sr and ^89Sr in seawater off Japan as a consequence of the Fukushima Dai-Ichi nuclear accident. Biogeosciences. 2013;10(6):3649–59. https://doi.org/10.5194/bg-10-3649-2013.
Johansen MP, Prentice E, Cresswell T, Howell N. Initial data on adsorption of Cs and Sr to the surfaces of microplastics with biofilm. J Environ Radioact. 2018;190:130–3.
Ioannidis I, Anastopoulos I, Pashalidis I. Microplastics as radionuclide (U-232) carriers. J Mol Liq. 2022;351:118641. https://doi.org/10.1016/j.molliq.2022.118641.
O’Grady J, Currivan L, McEnri C, O’Colmain M, Colgan PA, Cunningham JD. Radioactivity Monitoring of the Irish Marine Environment 1988–1990; 1991.
Kershaw PJ, Pentreath RJ, Woodhead DS, Hunt GJ. A Review of Radioactivity in the Irish Sea. Aquat. Environ. Monit. Rep. 1992, 32.
Andrady AL. Microplastics in the marine environment. Mar Pollut Bull. 2011;62(8):1596–605. https://doi.org/10.1016/j.marpolbul.2011.05.030.
Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, Narayan R, Law KL. Marine pollution. Plastic waste inputs from land into the ocean. Science. 2015;347(6223):768–71. https://doi.org/10.1126/science.1260352.
Rochman C, Tahir A, Williams S, Baxa D, Lam R, Miller J, Teh F-C, Werorilangi S, Swee Teh J. Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci Rep. 2015. https://doi.org/10.1038/srep14340.
Samadi A, Kim Y, Lee S-A, Kim YJ, Esterhuizen M. Review on the ecotoxicological impacts of plastic pollution on the freshwater invertebrate daphnia. Environ Toxicol. 2022;37(11):2615–38. https://doi.org/10.1002/tox.23623.
Yao X, Luo X-S, Fan J, Zhang T, Li H, Wei Y. Ecological and human health risks of atmospheric microplastics (MPs): a review. Environ Sci Atmos. 2022;2(5):921–42. https://doi.org/10.1039/D2EA00041E.
Vivesi Batlle J. Radioactivityradioactivityin the marine environment. In: Meyers RA, editor. BT encyclopedia of sustainability science and technology. New York: Springer; 2012. p. 8387–425.
Tu C, Chen T, Zhou Q, Liu Y, Wei J, Waniek JJ, Luo Y. Biofilm formation and its influences on the properties of microplastics as affected by exposure time and depth in the seawater. Sci Total Environ. 2020;734:139237.
Lobelle D, Cunliffe M. Early microbial biofilm formation on marine plastic debris. Mar Pollut Bull. 2011;62(1):197–200.
Yadav S, Rout S, Tiwari M, Mhatre S, Karpe R, Pulhani V, Kumar AV. Simulated experimental investigation of microplastic weathering in marine environment. J Environ Sci Heal - Part A. 2022. https://doi.org/10.1080/10934529.2022.2085984.
IAEA. Sediment distribution coefficients and concentration factors for biota in the marine environment. Vienna: IAEA: International Atomic Energy Agency (IAEA); 2004.
Gaylord NG, Adler G. Radiation chemistry of polymeric systems high polymers, A. Chapiro, Ed., Interscience, New York, 1962, Xvi + 712 Pp. $21.00. J Polym Sci Part A Gen Pap. 1963;1(6):2237. https://doi.org/10.1002/pol.1963.100010646.
Henglein AJWT, Spinks RJ. Woods: an introduction to radiation chemistry, third edition, John-Wiley and Sons, Inc., New York, Toronto 1990. ISBN 0-471-61403-3. 574 Seiten, Preis: DM 91, 45. Berichte der Bunsengesellschaft für Phys Chemie. 1991;95(3):451. https://doi.org/10.1002/bbpc.19910950346.
Talari ACS, Martinez MAG, Movasaghi Z, Rehman S, Rehman IU. Advances in ourier transform infrared (FTIR) spectroscopy of biological tissues. Appl Spectrosc Rev. 2017;52(5):456–506.
Wang J, Guo X, Xue J. Biofilm-developed microplastics as vectors of pollutants in aquatic environments. Environ Sci Technol. 2021;55(19):12780–90.
Nauendorf A, Krause S, Bigalke NK, Gorb EV, Gorb SN, Haeckel M, Wahl M, Treude T. Microbial colonization and degradation of polyethylene and biodegradable plastic bags in temperate fine-grained organic-rich marine sediments. Mar Pollut Bull. 2016;103(1–2):168–78.
Hartmann R, Singh PK, Pearce P, Mok R, Song B, Díaz-Pascual F, Dunkel J, Drescher K. Emergence of three-dimensional order and structure in growing biofilms. Nat Phys. 2019;15:251–6.
Periáñez R, Brovchenko I, Jung KT, Kim KO, Maderich V. The marine kd and water/sediment interaction problem. J Environ Radioact. 2018;192:635–47.
Atugoda T, Vithanage M, Wijesekara H, Bolan N, Sarmah AK, Bank MS, You S, Ok YS. Interactions between microplastics, pharmaceuticals and personal care products: implications for vector transport. Environ Int. 2021;149:106367. https://doi.org/10.1016/j.envint.2020.106367.
Binda G, Spanu D, Monticelli D, Pozzi A, Bellasi A, Bettinetti R, Carnati S, Nizzetto L. Unfolding the interaction between microplastics and (trace) elements in water: a critical review. Water Res. 2021;204:117637. https://doi.org/10.1016/j.watres.2021.117637.
Liu S, Shi J, Wang J, Dai Y, Li H, Li J, Liu X, Chen X, Wang Z, Zhang P. Interactions between microplastics and heavy metals in aquatic environments: a review. Front Microbiol. 2021. https://doi.org/10.3389/fmicb.2021.652520.
Guo X, Pang J, Chen S, Jia H. Sorption properties of tylosin on four different microplastics. Chemosphere. 2018;209:240–5.
Hüffer T, Hofmann T. Sorption of non-polar organic compounds by micro-sized plastic particles in aqueous solution. Environ Pollut. 2016;214:194–201.
Xu B, Liu F, Brookes PC, Xu J. The sorption kinetics and isotherms of sulfamethoxazole with polyethylene microplastics. Mar Pollut Bull. 2018;131(Pt A):191–6.
Ma J, Zhao J, Zhu Z, Li L, Yu F. Effect of microplastic size on the adsorption behavior and mechanism of triclosan on polyvinyl chloride. Environ Pollut. 2019;254:113104.
Uber TH, Hüffer T, Planitz S, Schmidt TC. Characterization of sorption properties of high-density polyethylene using the poly-parameter linearfree-energy relationships. Environ Pollut. 2019;248:312–9.
Velez JFM, Shashoua Y, Syberg K, Khan FR. Considerations on the use of equilibrium models for the characterisation of HOC-microplastic interactions in vector studies. Chemosphere. 2018;210:359–65.
Holmes LA, Turner A, Thompson RC. Adsorption of trace metals to plastic resin pellets in the marine environment. Environ Pollut. 2012;160:42–8. https://doi.org/10.1016/j.envpol.2011.08.052.
Gao X, Hassan I, Peng Y, Huo S, Ling L. Behaviors and influencing factors of the heavy metals adsorption onto microplastics: a review. J Clean Prod. 2021;319:128777. https://doi.org/10.1016/j.jclepro.2021.128777.
Späth R, Flemming H-C, Wuertz S. Sorption properties of biofilms. Water Sci Technol. 1998;37(4–5):207–10.
Lu Y, Zhang Y, Deng Y, Jiang W, Zhao Y, Geng J, et al. Uptake and accumulation of polystyrene microplastics inzebrafish (Danio rerio) and toxic effects in liver. Environ Sci Technol. 2016;50(7):4054–60.
Li Y, Tao L, Wang Q, Wang F, Li G, Song M. Potential health impact of microplastics: a review of environmental distribution, human exposure, and toxic effects. Environ Heal. 2023;1(4):249–57.
Author information
Authors and Affiliations
Contributions
Sonali—Data curation, Investigation, Visualisation, Writing—original draft, review & editing. Sabyasachi—Conceptualization, Investigation, Visualisation, Writing—original draft, review & editing, Resources, Supervision, Project administration, Funding acquisition. Vikram Joshi: Data curation Vandana Pulhani- Supervision, review, editing, and Project administration A. V. Kumar- Supervision, review, editing, and Project administration. All authors reviewed the manuscript Ms. Sonali and Dr. Sabyasachi Rout Both are equally contributed to the Research work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yadav, S., Rout, S., Joshi, V. et al. Unfolding the interaction of radioactive Cs and Sr with polyethylene-derived microplastics in marine environment. Discov Oceans 1, 13 (2024). https://doi.org/10.1007/s44289-024-00015-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44289-024-00015-8