Abstract
An enormous cost is placed on people, communities, and healthcare systems by bacterial infections. Measures of the burden of bacterial infections include morbidity, mortality, economic expenditures, and overall effects on public health. Campylobacteriosis is a bacterial infection imposes a significant economic burden on both individuals and societies due to its prevalence, healthcare costs, and the associated loss of productivity. In our research, we develop a model to analyze the transmission of campylobacteriosis infection, taking into account factors such as vaccination and treatment. We also examine the fundamental characteristics of fractional calculus to understand the model better. The equilibria of the model are studied, and we calculate the reproduction parameter denoted as \(\mathcal{R}_{0}\). Furthermore, we provide proof of stability for the equilibria of the system. Lastly, we conduct numerical investigations to explore the variation of the system’s reproduction parameter with different input parameters. We have established the necessary conditions to ensure the existence and uniqueness of solutions for the proposed model of campylobacteriosis infection. To better understand the complex dynamics of campylobacteriosis infection, we conduct various simulations of the suggested model while modifying the input factors. These simulations allow us to investigate the effects of different input parameters on the dynamics of campylobacteriosis infection. We analyze the dynamic behavior of the system to develop efficient control strategies for managing the infection. Notable improvements have been observed by reducing the order of the fractional derivative. Based on our findings, we propose various factors to the policy makers in the community to mitigate the spread of campylobacteriosis infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Campylobacteriosis is a bacterial infection caused by campylobacter species, primarily campylobacter jejuni and, to a lesser extent, campylobacter coli. These bacteria are commonly found in the intestines of animals, especially poultry (chickens, turkeys), cattle, and other livestock [1]. Campylobacteriosis is a prevalent global food-borne illness that is commonly contracted through the consumption of contaminated food or water. The main sources of contamination are raw or undercooked poultry, unpasteurized milk, and untreated water. Additionally, the infection can spread through direct contact with infected animals or their feces [2]. Symptoms of campylobacteriosis usually manifest between 2 to 5 days following exposure to the bacteria. The symptoms may include abdominal cramps, Diarrhea and pain, nausea and vomiting, fever, muscle pain and headache [3]. In the majority of instances, the illness is self-limiting and typically resolves within a week, but some individuals, particularly young children, elderly, and immuno-compromised individuals, may experience more severe symptoms and complications. Most cases of campylobacteriosis do not require specific treatment, as the infection will clear up on its own. However, it is essential to stay well-hydrated and manage symptoms like diarrhea. In severe cases or for individuals at higher risk of complications, antibiotics may be prescribed by a health-care professional. Most cases of campylobacteriosis do not require specific treatment, as the infection will clear up on its own. However, it is essential to stay well-hydrated and manage symptoms like diarrhea. In severe cases or for individuals at higher risk of complications, antibiotics may be prescribed by a health-care professional [4].
To prevent campylobacteriosis, it is crucial to follow good food safety practices, such as thoroughly cooking poultry and other meats, avoiding cross-contamination between raw and cooked foods, and consuming pasteurized milk and dairy products. Hand-washing after handling raw meat and before eating is also essential [5]. Campylobacteriosis is classified as a zoonotic disease as it can be transmitted from animals to humans. Avian species, including chickens, geese, and ducks, serve as a key reservoir for campylobacter jejuni infections [6]. Additionally, other reservoirs of campylobacter jejuni include humans and animals such as goats, dogs, cats, pets, and swine [6]. It is important to note that campylobacteriosis cannot be directly transmitted from one human to another. Instead, humans typically acquire Campylobacter infections through the fecal-oral route, involving the consumption of contaminated animal-derived food items such as meat, milk, undercooked chicken, water, and feces from infected humans [7,8,9]. Numerous experimental and mathematical models have been employed to recognize the transmission of zoonotic food-borne diseases, including campylobacteriosis, cholera, salmonellosis, yersiniosis, shigellosis, and listeriosis [10, 11]. In addition to this, mathematical models contribute significantly to visualize the transmission dynamics of diseases and provide new strategies for the control and prevention of diseases [12, 13].
The authors in [14] proposed an epidemic model that incorporated both humans and animals to investigate the dynamics and control of campylobacteriosis using a non-standard finite approach. Their findings demonstrated that reducing contact between susceptible humans, infected humans, and infected animals could effectively control campylobacteriosis. Furthermore, the authors in [8] explored the dynamics of the infection to study campylobacteriosis transmission in humans. Their research emphasized the significance of public health education campaigns, treatment, and sanitation in reducing the transmission of campylobacteriosis among humans. Mathematical modeling has emerged as a powerful tool in understanding the spread and control of infectious diseases [15,16,17]. These models play a crucial role in epidemiology, public health planning, and decision-making processes [18, 19]. Building upon this knowledge, our study proposes a mathematical model that incorporates treatment and cumulative public health education campaigns to enhance hygiene practices and minimize the health-care risks associated with campylobacteriosis among humans. This study, therefore, propose a mathematical model to incorporate treatment and cumulative public health education campaigns for improving hygiene practices aiming at minimize the embedded health-care risks of the campylobacteriosis disease among human.
Mathematical modeling serves as a powerful tool for comprehending real-world problems and capturing the entirety of phenomena across various domains [20,21,22,23]. Fractional calculus plays a vital role in investigating the influence of memory on epidemiological models and generating more accurate results. In essence, fractional calculus offers greater flexibility compared to classical calculus by incorporating the inherent characteristics of memory into the models. Several fractional operators have been introduced in the literature [24, 25], and these operators have been effectively applied in modeling various real-world scenarios. Specifically, fractional calculus has been extensively studied and its diverse applications have been demonstrated in numerous research studies [26,27,28]. It is commonly understood that ordinary differential systems are suitable for describing local dynamical systems without any external influences. The complexity and crossover dynamics exhibited by communicable diseases surpass the abilities of conventional differential operators. In contrast, fractional order (FO) derivatives offer a better fit to real-world data when modeling diverse infection dynamics, and they also yield more efficient experimental results across different fields. Several research studies have effectively employed fractional order derivatives, yielding improved outcomes when examining real-world data across a for diseases [29,30,31,32].
Based on the aforementioned research, we have developed a mathematical model to describe the dynamics of campylobacteriosis infection using a fractional order approach, specifically employing the Caputo–Fabrizio derivative with an order \(\imath \in (0,1]\). The subsequent sections of our research are organized as follows: Sect. 2 provides the fundamental definitions and propositions of the fractional theory. In Sect. 3 of our study, we develop a campylobacteriosis infection model that incorporates the effects of vaccination and treatment using the fractional Caputo–Fabrizio (CF) derivatives. We analyze the equilibria of the model and calculate the reproduction number for the proposed system. In Sect. 4, we examine the existence and uniqueness of solutions for our system using fixed point theory. Moving on to Sect. 5, we introduce a numerical scheme to explore the influence of various input factors on the system’s dynamics. Finally, in Sect. 6, we provide concluding remarks to summarize our study.
2 Fractional concepts
Now we will present the basic concepts and outcomes of the Caputo–Fabrizio (CF) fractional derivatives, which will be utilized for analyzing the dynamics of campylobacteriosis infection. The essential definitions of these concepts are provided below:
Definition 2.1
The CF fractional derivative [25] for function \(h \in \mathfrak {H}^1(r_{1}, r_{2})\) is stated as
where \(r_{2}>r_{1}\) and \(\mathfrak {J}(\tau )\) represent the normality [25] such that \(\imath \in [0, 1]\). In the case, if \(h \notin \mathfrak {H}^1(r_{1}, r_{2})\), then we obtain
Remark 2.1
In the case if \(\zeta =\frac{1-\imath }{\imath }\in [0, \infty )\) and \(\imath =\frac{1}{1+\zeta }\in [0, 1]\), then Eq. (2) gives the following
Additionally, we have
Definition 2.2
[33], The fractional integral of a given function f can be represented below in the context of fractional calculus as
where fractional order of the integral is represented by \(\imath\) such that \(0<\imath <1\).
Remark 2.2
Definition 2.2 implies the below stated equation
which shows that \(\mathfrak {J}(\imath )=\frac{2}{2-\imath }\), \(0<\imath <1\). In the paper [33], the authors utilized Eq. (6) to introduce the following
where fractional order is indicated by \(\imath\) with \(0< \imath < 1\).
3 Evaluation of the fractional dynamics
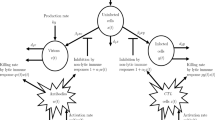
In the formulation of the model, we divided the total humans population into four classes including susceptible \(\texttt {Q}\), vaccinated \(\texttt {K}\), infected \(\texttt {J}\) and recovered class \(\texttt {P}\) while the compybacter population is indicated by \(\texttt {B}\) that refers to all sources of the disease transmissions. The recruitment rate is represented by \(\**\) while \(\mu\) represents the natural death rate. The fraction p represents the vaccinated fraction of the susceptible humans population and \(\beta\) denotes the transmission rate of humans population. In Fig. 1, we illustrated the flow chart of the model to visualize the overall picture of the transmission. Thus from the above stated assumptions we get the following model
with the given values of state variables
The size of population is given by the following
\(\tau\) represent the treatment rate, \(\delta\) represent the disease induced death rate while the recovery rate is represented by \(\gamma\). In addition, the infected humans shed bacteria into the environment at the rate \(\psi \texttt {J}\). Due to the epidemiological aspects of campylobacteriosis disease, individuals who get infected recover temporarily and hence those who loose immunity relapse to the susceptible population at the waning rate \(\nu \texttt {P}\). Indeed, the rate at which bacteria are wiped out from the environment is \(\eta \texttt {B}\) where \(\eta\) defines the natural clearance rate of bacteria from the environment.
Fractional calculus finds extensive utility across various research domains, particularly in the realm of mathematical biology. Empirical evidence indicates that the dynamics of infections can be more accurately represented using fractional calculus compared to classical integer order derivatives. Thus, the aforementioned campylobacteriosis infection model can be expressed within the fractional framework as follows:
where the CF fractional operator order is represented by \(\imath\). The positive invariant region for the proposed fractional system of campylobacteriosis infection is provided below:
Theorem 3.1
Assuming \(\Omega =\{ (\texttt {Q},\texttt {K},\texttt {J},\texttt {P},\texttt {B}) \in R^5_+: 0 \le \texttt {Q}+\texttt {K}+\texttt {J}+\texttt {P}+\texttt {B} \le \textrm{M} \}\), then the set \(\Omega\) remains positively invariant for the proposed system (9) of campylobacteriosis infection.
3.1 Analysis of fractional dynamics
In this section, we will examine the suggested model (9) for campylobacteriosis infection in terms of steady-state and stability. Initially, we investigate the disease-free equilibrium, represented as \(\mathcal{E}_0\), of the suggested system. Considering the aforementioned fractional model of campylobacteriosis infection in steady-states without infection, we get that
To find the reproduction number for the system (9), we will use the following method
The result obtained by evaluating the Jacobian of matrices \(\textrm{F}\) and \(\textrm{V}\) at the disease-free equilibrium point \(\mathcal{E}_{0}\) is as follows
Now calculating \(\texttt {V}^{-1}\), we have
which implies that
The basic reproduction number can be determined by finding the spectral radius of the matrix \(\texttt {F}{} \texttt {V}^{-1}\), which is obtained by calculating the eigenvalues of the \(\texttt {F}{} \texttt {V}^{-1}\) matrix. The reproduction number \(\mathcal{R}_{0}\) is stated as follows
Theorem 3.2
The disease-free equilibrium of the campylobacteriosis infection model (9) exhibits local asymptotic stability when \(\mathcal{R}_{0}<1\), while it is unstable in other cases.
Proof
In order to prove the theorem, we will compute the Jacobian matrix at \(\mathcal{E}_0\) as follows
By evaluating the above stated matrix \(\mathcal{J}_{\mathcal{E}_{0}}\), we achieve that \(\lambda _{1}=-\mu\), \(\lambda _{2}=-(p+\mu )\) and \(\lambda _{3}=-(\mu +\nu )\), which reduces the matrix \(\mathcal{J}_{\mathcal{E}_{0}}\) to a 2 by 2 matrix, that is
In order to show that the system (9) is locally asymptotically stable we will prove that \(Trace(\mathcal{J}_{\mathcal{E}_{1}})<0\) and \(Det(\mathcal{J}_{\mathcal{E}_{1}})>0\). Thus we have
and
Since \(Trace(\mathcal{J}_{\mathcal{E}_{1}})<0\) and \(Det(\mathcal{J}_{\mathcal{E}_{1}})>0\) if \(\mathcal{R}_{0}<1\), which shows that the disease free equilibrium of campylobacteriosis infection model (9) will be locally asymptotically stable if \(\mathcal{R}_{0}<1\) and unstable in other cases.
4 Analysis of the solutions
In this context, our main focus lies in examining the solutions of the proposed fractional system of campylobacteriosis infection. To investigate the existence of solution (9), we employ fixed point theory and proceed as outlined below
By incorporating the concepts introduced in the research [33], we derived the subsequent findings
Moreover, we have
Theorem 4.1
If the below stated condition holds true, then the kernels \(\Re _1, \Re _2, \Re _3, \Re _4\), and \(\Re _5\) satisfy the conditions of Lipschitz and contraction,
Proof 4.1
To achieve the desired outcomes mentioned above, we begin with \(\texttt {Q}\) and \(\texttt {Q}_1\), and proceed with \(\Re _1\) according to the following approach
Further simplifying (13), we achieve that
Here, we have \(\Vert \beta \texttt {B} \Vert \le M\) due to boundedness and assume \(\kappa _{1}= (M+p+\mu )\), then we get
Hence, we have established the Lipschitz condition for \(\Re _1\). Moreover, by satisfying the condition \(0 \le (M+p+\mu ) <1\), we have also obtained the contraction property. Similarly, we can ascertain the Lipschitz conditions as follows
Further simplification of Eq. (11) yields that
moreover, we have
with initial values
We can achieve the difference terms in the following manner
Observing that
Simplifying in the same manner, we achieve that
Equation (21) yields that
Theorem 4.2
If it is possible to find a suitable \(t_0\) such that the below stated condition holds
then, we obtain exact coupled solutions for the proposed fractional system (9).
Proof 4.2
With the Lipschitz condition being met and the boundedness of \(\texttt {Q}(t)\), \(\texttt {K}(t)\), \(\texttt {J}(t), \texttt {P}(t)\) and \(\texttt {B}(t)\), we can infer the following from Eqs. (24) and (25):
As a consequence, we attain existence and continuity of the solutions. Additionally, we need to establish that the aforementioned solution is valid for our system (9). To accomplish this, we proceed as outlined below
Next, we take
Additionally, we have
At time \(t_0\), we get
By following the same procedure and utilizing Eq. (30), we obtain the following
Similarly, we observe that \(\mathfrak {A}2_\aleph (t), \mathfrak {A}3_\aleph (t), \mathfrak {A}4_\aleph (t),\mathfrak {A}5_\aleph (t)\) tends to 0 as \(\aleph\) tends to \(\infty\). \(\square\)
To establish the uniqueness of solutions for the system (9), let \((\texttt {Q}_{1}(t)\), \(\texttt {K}_{1}(t)\), \(\texttt {J}_{1}(t)\), \(\texttt {P}_{1}(t)\), \(\texttt {B}_{1}(t))\) is an alternative solution. In such a case, we proceed in the following manner
By utilizing norm on (31), we get
Based on the fulfillment of the Lipschitz condition, we can derive the following
which implies that
Theorem 4.3
If the below stated condition holds
then the system (9) has unique solution.
Proof 4.3
Suppose that the condition mentioned in Eq. (35) is met, then Eq. (34) yields the following
from the above, we have
Similarly, we achieve that
\(\square\)
In the upcoming section, we will discuss the solution pathways of the recommended model of the infection to illustrate the impact of different input factors on the system. These analysis will help us to predict the most critical factors of the system to health officials or policy makers.
5 Result and discussion
In this section of the paper, we will present the dynamical behaviour of the recommended model of bacterial infection with the variation of different input factors of the system. The behavior of an epidemic model can vary depending on several factors, including the characteristics of the disease, the population, and the implemented control measures. We performed different simulation to show the key factors of the system for the control and prevention of the infection. The values of input parameters and state variables are assumed for simulation purposes. In Table 1, we represent the values of input parameters of the system.
In the first simulation showcased in Figs. 2 and 3, we have depicted the influence of a fractional parameter on the dynamics of campylobacteriosis infection. Our aim was to demonstrate the behavior of the infection level as affected by the fractional parameter. Figure 2 presents a comparative analysis between classical and fractional operators. Through these figures, we observed a substantial reduction in the level of infection when employing the fractional parameter, indicating its potential as an effective control parameter.
Figure 4 demonstrates the second simulation, which focuses on the conceptualization of the role of vaccination. Through this simulation, we observed that vaccination can serve as an effective measure to control campylobacteriosis infection within a society. By assuming various values for the vaccination rate, we were able to assess the system’s behavior and its response to different vaccination scenarios. Figure 5 displays the third simulation, which focuses on visualizing the role of transmission probability. By presenting solution pathways of the system under different values of the transmission probability parameter \(\beta\), we have demonstrated that \(\beta\) plays a critical role in determining the risk of infection within a society. Our findings highlight that higher values of \(\beta\) can significantly increase the risk of infection. Figure 6 depicts the exploration of treatment’s role in the transmission dynamics of campylobacteriosis infection. The visualization reveals a marginal impact of this parameter on the infection dynamics. Furthermore, in the last simulation showcased in Fig. 7, we examined the variation of the input parameter \(\nu\) and its effect on the solution pathways. Our results demonstrated that this parameter poses a significant risk, as it increases the likelihood of infection.
Our findings indicate that the fractional parameter and vaccination play crucial roles in reducing the infection level within a society. These factors have been shown to be effective in mitigating the spread of the infection. Conversely, we observed that the transmission probability \(\beta\) and the rate of immunity loss \(\nu\) pose significant risks, as they can contribute to the propagation of the infection.
6 Concluding remarks
In this work, we structured a model that captures the transmission dynamics of campylobacteriosis infection by incorporating vaccination and treatment factors using the Caputo–Fabrizio fractional operator. We utilized key principles from fractional calculus to analyze the dynamics of the proposed model of the infection. We studied the equilibria of the model and calculated the reproduction parameter represented by \(\mathcal{R}_{0}\) through next-generation matrix method. We have shown that the infection-free steady-state of the proposed system is local asymptotic stability when \(\mathcal{R}_{0}<1\), while it becomes unstable in other conditions. Applying the theory of fractional derivatives, we proved the existence and uniqueness of the solution of the proposed model of campylobacteriosis infection. In addition to this, We presented a numerical scheme to analyze the time series of the system, which allows us to explore the dynamical behavior. Through numerical simulations, we investigated the impact of various input parameters on the dynamics of campylobacteriosis infection. This enabled us to conceptualize the influence of different input parameters on the infection dynamics. Our findings recommended the most pivotal scenario to the public health authorities and policy makers for the prevention of the infection. This will assist them in implementing appropriate infection control measures to address the situation effectively. In our future work, we will use machine learning techniques to analyze large-scale epidemiological data and optimize model parameters, aiding in real-time disease monitoring and control strategies. Moreover, climate change and environmental factors will be included in the model to gain a deeper understanding of disease dynamics and inform public health policies.
Availability of data and materials
Not applicable
References
Zhang Q, Sahin O (2020) Campylobacteriosis. Diseases of poultry 2020: 754–769
Frosth S, Karlsson-Lindsj O, Niazi A, Fernstrm LL, Hansson I (2020) Identification of transmission routes of Campylobacter and on-farm measures to reduce Campylobacter in chicken. Pathogens 9(5):363
Kuhn KG, Falkenhorst G, Emborg HD, Ceper T, Torpdahl M, Krogfelt KA, Ethelberg S, Molbak K (2017) Epidemiological and serological investigation of a waterborne Campylobacter jejuni outbreak in a Danish town. Epidemiol Infect 145(4):701–709
Moffatt CR, Kennedy KJ, ONeill B, Selvey L, Kirk MD (2021) Bacteraemia, antimicrobial susceptibility and treatment among Campylobacter-associated hospitalisations in the Australian Capital Territory: a review. BMC Infect Dis 21(1):1–12
Igwaran A, Okoh AI (2019) Human campylobacteriosis: a public health concern of global importance. Heliyon 5:11
Hansson I, Sandberg M, Habib I, Lowman R, Engvall EO (2018) Knowledge gaps in control of Campylobacter for prevention of campylobacteriosis. Transbound Emerg Dis 65:30–48
Chlebicz A, Slizewska K (2018) Campylobacteriosis, salmonellosis, yersiniosis, and listeriosis as zoonotic foodborne diseases: a review. Int J Environ Res Public Health 15(5):863
Chuma F, Mussa Z (2021) Campylobacteriosis transmission dynamics in humans: modeling the effects of public health education, treatment, and sanitation. Tanzania J Sci 47(1):315–331
Sahin O, Fitzgerald C, Stroika S, Zhao S, Sippy RJ, Kwan P, Plummer PJ, Han J, Yaeger MJ, Zhang Q (2012) Molecular evidence for zoonotic transmission of an emergent, highly pathogenic Campylobacter jejuni clone in the United States. J Clin Microbiol 50(3):680–687
Berhe HW, Makinde OD, Theuri DM (2019) Co-dynamics of measles and dysentery diarrhea diseases with optimal control and cost-effectiveness analysis. Appl Math Comput 347:903–921
Olaniyi S, Abimbade SF, Ajala OA, Chuma FM (2023) Efficiency and economic analysis of intervention strategies for recurrent malaria transmission. Qual Quant 2023: 1–19
Guo Y, Li T (2022) Modeling and dynamic analysis of novel coronavirus pneumonia (COVID-19) in China. J Appl Math Comput 68(4):2641–2666
Guo Y, Li T (2023) Modeling the competitive transmission of the Omicron strain and Delta strain of COVID-19. J Math Anal Appl 526(2):127283
Osman S, Togbenon HA, Otoo D (2020) Modelling the dynamics of campylobacteriosis using nonstandard finite difference approach with optimal control. Comput Math Methods Med 2020:1–12
Zhang J, Jia J, Song X (2014) Analysis of an SEIR epidemic model with saturated incidence and saturated treatment function. Sci World J 2014: 1-11
Liu J (2019) Bifurcation analysis for a delayed SEIR epidemic model with saturated incidence and saturated treatment function. J Biol Dyn 13(1):461–480
Rawson T, Dawkins MS, Bonsall MB (2019) A mathematical model of Campylobacter dynamics within a broiler flock. Front Microbiol 10:1940
Li T, Guo Y (2022) Optimal control and cost-effectiveness analysis of a new COVID-19 model for Omicron strain. Physica A 606:128134
Li T, Guo Y (2022) Modeling and optimal control of mutated COVID-19 (Delta strain) with imperfect vaccination. Chaos Solitons Fractals 156:111825
Shah Z, Bonyah E, Alzahrani E, Jan R, Aedh Alreshidi N (2022) Chaotic phenomena and oscillations in dynamical behaviour of financial system via fractional calculus. Complexity 2022: 1-14
Jan R, Jan A (2017) MSGDTM for solution of fractional order dengue disease model. Int J Sci Res 6(3):1140–1144
Boulaaras S, Choucha A, Cherif B, Alharbi A, Abdalla M (2021) Blow up of solutions for a system of two singular nonlocal viscoelastic equations with dam**, general source terms and a wide class of relaxation functions. AIMS Math 6(5):4664–4676
Khattak S, Hussain I, Gomez-Aguilar JF, Jan R (2021) Analysis of PD-type iterative learning control for discrete-time singular system. Math Methods Appl Sci 2021: 1-14
Samko SG (1993) Fractional integrals and derivatives. Theory Appl 1993
Caputo M, Fabrizio M (2015) A new definition of fractional derivative without singular kernel. Progress Fract Differ Appl 1(2):73–85
Tang TQ, Shah Z, Bonyah E, Jan R, Shutaywi M, Alreshidi N (2022) Modeling and analysis of breast cancer with adverse reactions of chemotherapy treatment through fractional derivative. Comput Math Methods Med 2022: 1-19
Guefaifia R, Boulaaras SM, El-Sayed AAE, Abdalla M, Cherif BB (2021) On existence of sequences of weak solutions of fractional systems with Lipschitz nonlinearity. J Funct Spaces 2021:2017
Tang TQ, Shah Z, Jan R, Alzahrani E (2022) Modeling the dynamics of tumorimmune cells interactions via fractional calculus. Eur Phys J Plus 137(3):367
Jan R, Qureshi S, Boulaaras S, Pham VT, Hincal E, Guefaifia R (2023) Optimization of the fractional-order parameter with the error analysis for human immunodeficiency virus under Caputo operator. Discrete Contin Dyn Syst 2023: 0-0
Qureshi S, Jan R (2021) Modeling of measles epidemic with optimized fractional order under Caputo differential operator. Chaos Solitons Fractals 145:110766
Guo Y, Li T (2023) Fractional-order modeling and optimal control of a new online game addiction model based on real data. Commun Nonlinear Sci Numer Simul 121:107221
Jan R, Boulaaras S, Shah SAA (2022) Fractional-calculus analysis of human immunodeficiency virus and CD4+ T-cells with control interventions. Commun Theor Phys 74(10):105001
Losada J, Nieto JJ (2015) Properties of a new fractional derivative without singular kernel. Progr Fract Differ Appl 1(2):87–92
Acknowledgements
Not applicable
Funding
Not applicable
Author information
Authors and Affiliations
Contributions
HD illustrated the natural phenomena of the model. HD supervised the overall work. RJ formulated the model and carried out analytic analysis. ZR carried out the numerical simulation of the work and wrote the manuscript. SB conceptualized, supervised and validated the proposed model. AJ presented the numerical scheme and find out the numerical results through software. AJ investigated the model and checked accuracy of numerical findings. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
There is no competing interests regarding this work
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Degaichia, H., Jan, R., Rehman, Z.U. et al. Fractional-view analysis of the transmission dynamics of a bacterial infection with nonlocal and nonsingular kernel. SN Appl. Sci. 5, 324 (2023). https://doi.org/10.1007/s42452-023-05538-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-023-05538-x