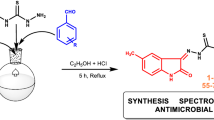
Abstract
Due to the emergence of drug-resistant microorganisms, treating infectious illnesses is a significant challenge in the medical industry. To counter this, researchers are continuously investigating new drugs to combat infectious illnesses. Hence, we explore the production of new SB derivatives (SB1–SB8) obtained from the condensation of 5-hydroxysalicyaldehyde with substituted amines. For structural confirmations of procured compounds, Fourier-transform infrared spectroscopy, 1H and 13C-Nuclear magnetic resonance, single crystal X-ray diffraction, and electrospray ionisation mass spectrometry spectroscopic methods of analysis were employed. The SCXRD analysis of compound SB1 has a triclinic lattice with P-1 and Z = 2 space groups. The antimicrobial attributes of synthesized SB derivatives (SB1–SB8) were further investigated. In comparison with conventional antibiotic ampicillin (250 and 100 µg/mL), the compound SB3 demonstrated enhanced effectiveness against S. aureus (100 µg/mL), E. coli (62.5 µg/mL), and S. pyogenes (50 g/mL), as well as euipotency against the P. aeruginosa (100 µg/mL) microorganisms. Among the synthesized compounds, compounds SB1 and SB5 to SB8 showed notably greater potency (100–250 µg/mL) against the fungus C. albicans in comparison to the conventional drug Griseofulvin (500 µg/mL).






Similar content being viewed by others
References
Frieri M, Kumar K, Boutin A J Infect Public Health., 2017 10, 369
Janowska S, Khylyuk D, Janowski M, Kosikowska U, Strzyga-Łach P (2023) M. Struga, and M. Wujec, Molecules., 28
Akova M (2016) Virulence 7:252
Terreni M, Taccani M, Pregnolato M (2021) Molecules, 26
Mishra N, Yadav R, Kumar K, Pandey H, Pandey R (2020) J Phys Conf Ser 1504:012002
Wady AF, Hussein MB, Mohammed MM (2021) Sch Int J Chem Mater Sci 4:46
Munawar KS, Haroon SM, Hussain SA, Raza H (2018) J Basic Appl Sci 14:217
Tople M, Patel N, Patel P, Purohit A, Ahmad I, Patel H (2022) J Mol Struct 1271:134016
Iacopetta D, Lappano R, Mariconda A, Ceramella J, Sinicropi MS, Saturnino C, Talia M, Cirillo F, Martinelli F, Puoci F (2020) Int J Mol Sci 21:7797
Rana K, Pandurangan A, Singh N, Tiwari AK (2012) Int J Curr Pharm Res 4:5
Hameed A, al-Rashida M, Uroos M, Abid S, Ali, Khan KM (2017) Expert Opin Ther Pat 27:63
GÜmÜŞ A, OkumuŞ V, GÜmÜŞ S (2020) Turk J Chem 44:1200
Waziri I, Yusuf TL, Akintemi E, Kelani MT, and A. Muller (2023) J Mol Struct 1273:134382
Shanty AA, Philip JE, Sneha EJ, Prathapachandra Kurup MR, Balachandran S, Mohanan PV (2017) Bioorg Chem 70:67
Murtaza S, Abbas A, Iftikhar K, Shamim S, Akhtar MS, Razzaq Z, Naseem K, Elgorban AM (2016) Med Chem Res 25:2860
Cinarli A, Gürbüz D, Tavman A, Birteksöz AS (2012) Chin J Chem 30:449
Sancheti RS, Shah HH, Patil AM, Patil RD (2023) and R. S. Bendre
Tadavi SK, Yadav AA, Bendre RS (2018) J Mol Struct 1152:223
BAUER AW, PERRY DM, KIRBY WMM (1959) AMA Arch Intern Med 104:208
Shebl M (2014) Spectrochim Acta Mol Biomol Spectrosc 117:127
Acknowledgements
The authors express gratitude to the esteemed Management, Principal of SNJB’s KKHA Arts, SMGL Commerce, and SPHJ Science College, Chandwad, and Department of Chemistry, KBC, North Maharashtra University, Jalgaon.
Author information
Authors and Affiliations
Contributions
All authors are equally contributed.
Corresponding author
Ethics declarations
Supplementary Information
The CCDC number of compound SB1 is 2208358. This data obtained from https://www.ccdc.cam.ac.uk/data_request/CIF, through emailing on data_request@ccdc.cam.ac.uk.The details of crystallographic data, IR, 1H-NMR and 13C-NMR, mass spectra, and tables are also provided in supplementary data.
Conflict of Interest
No conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Todarwal, M.A., Sancheti, R.S., Shah, H.H. et al. Exploring New Derivatives of 5-Hydroxysalicyaldehyde as Promising Antimicrobial Compounds Towards Drug-Resistant Microorganism. Chemistry Africa (2024). https://doi.org/10.1007/s42250-024-00973-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42250-024-00973-8