Abstract
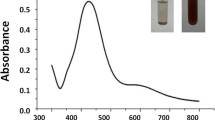
Pre-sowing seed treatment with nanoparticles has a promising role towards the improvement of seed germination and seedling growth. In the present study, silver nanoparticles (AgNPs) were synthesized from silver nitrate (AgNO3) through green route using aqueous extract of Parthenium hysterophorus L. roots. The synthesized nanoparticles were characterized using various analytical instruments such as UV–Vis spectrophotometer, TEM, SEM, EDX, XRD, and FTIR. Further, the impacts of AgNPs, AgNO3, and plant extract on germination, seedling growth, activity of hydrolytic enzymes, and ROS generation of three pulses (Cicer arietinum L., Pisum sativum L., and Vigna radiata L.) were investigated. Characterization of nanoparticles revealed that the green synthesized AgNPs were mostly spherical with an average size of 11–20 nm and crystallinity was 71.3%. The growth experiment revealed that seed germination and seedling growth were increased under AgNPs (10 and 50 mg/L) and AgNO3 (10 mg/L) treatments as compared to control for three tested pulses. Results also demonstrated the increased hydrolytic enzyme activities during early seedling establishment of three pulses under nanoparticle treatments. Meanwhile, dose dependent increase in ROS production was recorded under both AgNPs and AgNO3 treatments and it was always higher in AgNO3 as compared to AgNPs treatments. However, the growth inhibition at higher concentrations of both AgNPs and AgNO3 treatments suggested that the ROS generation at an optimum level might play an important role towards the enhancement of seed germination. Therefore, AgNPs mediated alteration of the activity of hydrolytic enzymes and generation of ROS might regulate early seedling establishment.
Graphical abstract











Similar content being viewed by others
References
Acharya P, Jayaprakasha GK, Crosby KM, Jifon JL, Patil BS (2019) Green-synthesized nanoparticles enhanced seedling growth, yield, and quality of onion (Allium cepa L.). ACS Sustain Chem Eng 7(17):14580–14590. https://doi.org/10.1021/acssuschemeng.9b02180
Alabdallah NM, Hasan MM (2021) Plant-based green synthesis of silver nanoparticles and its effective role in abiotic stress tolerance in crop plants. Saudi J Biol Sci 28(10):5631–5639. https://doi.org/10.1016/j.sjbs.2021.05.081
Almutairi ZM, Alharbi A (2015) Effect of silver nanoparticles on seed germination of crop plants. Int J Sci Res 9(6):594–598
Anusuya S, Banu KN (2016) Silver-chitosan nanoparticles induced biochemicalvariations of chickpea (Cicer arietinum L.). Biocatal Agric Biotechnol 8:39–44. https://doi.org/10.1016/j.bcab.2016.08.005
Avezum L, Rondet E, Mestres C, Achir N, Madode Y, Gibert O, Lefevre C, Hemery Y, Verdeil JL, Rajjou L (2022) Improving the nutritional quality of pulses via germination. Food Rev Int. https://doi.org/10.1080/87559129.2022.2063329
Basheerudeen MAH, Mushtaq SA, Soundhararajan R, Nachimuthu SK (2021) Marine endophytic fungi mediated Silver nanopa application in plant growth promotion in rticles and their Vigna radiata L. Int J Nano Dimens 12(1):1–10
Bhardwaj J, Anand A, Nagarajan S (2012) Biochemical and biophysical changes associated with magnetopriming in germinating cucumber seeds. Plant Physiol Biochem 57:67–73. https://doi.org/10.1016/j.plaphy.2012.05.008
Botcha S, Prattipati SD (2020) Role of amylase and protease in germinating Sterculia urens Roxb. Bangladesh J Sci Ind Res 55(2):107–112. https://doi.org/10.3329/bjsir.v55i2.47631
Chandrasekaran U, Luo X, Wang Q, Shu K (2020) Are there unidentified factors involved in the germination of nanoprimed seeds? Front Plant Sci 11:832. https://doi.org/10.3389/fpls.2020.00832
Chau NH, Doan QH, Chu TH, Nguyen TT, Dao Trong H, Ngo QB (2019) Effects of different nanoscale microelement-containing formulations for presowing seed treatment on growth of soybean seedlings. J Chem 20:19. https://doi.org/10.1155/2019/8060316
Debnath P, Mondal A, Sen K, Mishra D, Mondal NK (2020) Genotoxicity study of nano Al2O3, TiO2 and ZnO along with UV-B exposure: An Allium cepa root tip assay. Sci Total Environ 713:136592. https://doi.org/10.1016/j.scitotenv.2020.136592
Dobias J, Bernier-Latmani R (2013) Silver release from silver nanoparticles in natural waters. Environ Sci Technol 47(9):4140–4146. https://doi.org/10.1021/es304023p
Feizi H, Kamali M, Jafari L, Moghaddam PR (2013) Phytotoxicity and stimulatory impacts of nanosized and bulk titanium dioxide on fennel (Foeniculum vulgare Mill). Chemosphere 91(4):506–511. https://doi.org/10.1016/j.chemosphere.2012.12.012
Guha T, Das H, Mukherjee A, Kundu R (2021) Elucidating ROS signaling networks and physiological changes involved in nanoscale zero valent iron primed rice seed germination sensu stricto. Free Radic Biol Med 171:11–25. https://doi.org/10.1016/j.freeradbiomed.2021.05.005
Honchar L, Mazurenko B, Shutyi O, Pylypenko V, Rakhmetov D (2021) Effect of pre-seed and foliar treatment with nano-particle solutions on seedling development of tiger nut (Cyperus Esculentus L.) plants. Agron Res 19(S1):767–776. https://doi.org/10.15159/ar.21.021
Janmohammadi M, Sabaghnia N (2015) Effect of pre-sowing seed treatments with silicon nanoparticles on germinability of sunflower (Helianthus annuus). Bot Lith 21(1):13–21. https://doi.org/10.1515/botlit-2015-0002
Khan I, Awan SA, Rizwan M, Akram MA, Zia-ur-Rehman M, Wang X, Zhang X, Huang L (2023) Physiological and transcriptome analyses demonstrate the silver nanoparticles mediated alleviation of salt stress in pearl millet (Pennisetum glaucum L). Environ Pollut 318:120863. https://doi.org/10.1016/j.envpol.2022.120863
Kittler S, Greulich C, Diendorf J, Koller M, Epple M (2010) Toxicity of silver nanoparticles increases during storage because of slow dissolution under release of silver ions. Chem Mater 22(16):4548–4554. https://doi.org/10.1021/cm100023p
Kumar D, Dhankher OP, Tripathi RD, Seth CS (2023) Titanium dioxide nanoparticles potentially regulate the mechanism(s) for photosynthetic attributes, genotoxicity, antioxidants defense machinery, and phytochelatins synthesis in relation to hexavalent chromium toxicity in Helianthus annuus L. J Hazard Mater 454:131418. https://doi.org/10.1016/j.jhazmat.2023.131418/cm100023p
Laware SL, Raskar S (2014) Effect of titanium dioxide nanoparticles on hydrolytic and antioxidant enzymes during seed germination in onion. Int J Curr Microbiol App Sci 3(7):749–760
Lakshmi SJ, Bai RR, Sharanagouda H, Ramachandra CT, Nadagouda S, Doddagoudar SR (2017) Biosynthesis and characterization of ZnO nanoparticles from spinach (Spinacia oleracea) leaves and its effect on seed quality parameters of greengram (Vigna radiata). Int J Curr Microbiol Appl Sci 6(9):3376–3384. https://doi.org/10.20546/ijcmas.2017.609.416
Lowry O, Rosebrough N, Farr AL, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Ma C, White JC, Dhankher OP, **ng B (2015) Metal-based nanotoxicity and detoxification pathways in higher plants. Environ Sci Technol 49(12):7109–7122. https://doi.org/10.1021/acs.est.5b00685
Maclachlan S, Zalik S (1963) Plastid structure, chlorophyll concentration, and free amino acid composition of a chlorophyll mutant of barley. Can J Bot 41(7):1053–1062. https://doi.org/10.1139/b63-088
Mahakham W, Theerakulpisut P, Maensiri S, Phumying S, Sarmah AK (2016) Environmentally benign synthesis of phytochemicals-capped gold nanoparticles as nanopriming agent for promoting maize seed germination. Sci Total Environ 573:1089–1102. https://doi.org/10.1016/j.scitotenv.2016.08.120
Mahakham W, Sarmah AK, Maensiri S, Theerakulpisut P (2017) Nanopriming technology for enhancing germination and starch metabolism of aged rice seeds using phytosynthesized silver nanoparticles. Sci Rep 7(1):1–21. https://doi.org/10.1038/s41598-017-08669-5
McCready RM, Guggolz J, Silviera V, Owens HS (1950) Determination of starch and amylose in vegetables. Anal Chem 22(9):1156–1158. https://doi.org/10.1021/ac60045a016
Mondal NK, Chowdhury A, Dey U, Mukhopadhya P, Chatterjee S, Das K, Datta JK (2014) Green synthesis of silver nanoparticles and its application for mosquito control. Asian Pac J Trop Dis 4(1):S204–S210. https://doi.org/10.1016/S2222-1808(14)60440-0
Mondal A, Sen K, Mondal A, Mishra D, Debnath P, Mondal NK (2022) Bio-fabricated silver nanoparticles for controlling dengue and filaria vectors and their characterization, as well as toxicological risk assessment in aquatic mesocosms. Environ Res 212:113309. https://doi.org/10.1016/j.envres.2022.113309
Nair PMG, Chung IM (2014) Assessment of silver nanoparticle-induced physiological and molecular changes in Arabidopsis thaliana. Environ Sci Pollut Res 21:8858–8869. https://doi.org/10.1007/s11356-014-2822-y
Nair PMG, Chung IM (2015) Physiological and molecular level studies on the toxicity of silver nanoparticles in germinating seedlings of mung bean (Vigna radiata L.). Acta Physiol Plant 37(1):1–11. https://doi.org/10.1007/s11738-014-1719-1
Oracz K, Karpinski S (2016) Phytohormones signaling pathways and ROS involvement in seed germination. Front Plant Sci 7:864. https://doi.org/10.3389/fpls.2016.00864
Pereira ADES, Oliveira HC, Fraceto LF (2019) Polymeric nanoparticles as an alternative for application of gibberellic acid in sustainable agriculture: a field study. Sci Rep 9(1):1–10. https://doi.org/10.1016/j.fbio.2021.100978
Pérez-de-Luque A (2017) Interaction of nanomaterials with plants: what do we need for real applications in agriculture? Front Environ Sci. https://doi.org/10.3389/fenvs.2017.00012
Prażak R, Święciło A, Krzepiłko A, Michałek S, Arczewska M (2020) Impact of Ag nanoparticles on seed germination and seedling growth of green beans in normal and chill temperatures. Agriculture 10(8):312. https://doi.org/10.3390/agriculture10080312
Rai-Kalal P, Jajoo A (2021) Priming with zinc oxide nanoparticles improve germination and photosynthetic performance in wheat. Plant Physiol Biochem 160:341–351. https://doi.org/10.1016/j.plaphy.2021.01.032
Rashid H, Khan MA, Amin A, Nawab K, Hussain N, Bhowmik PK (2008) Effect of Parthenium hysterophorus L., root extracts on seed germination and growth of maize and barley. Am J Plant Sci Biotech 2(2):51–55
Rauf A, Khan IA, Alnasser SM, Shah SUA, Rahman MM (2022) Phytochemical analysis and in vitro and in vivo pharmacological evaluation of Parthenium hysterophorus Linn. Evid Based Complem Altern Med 2022:6088585. https://doi.org/10.1155/2022/6088585
Song K, He X (2021) How to improve seed germination with green nanopriming. Seed Sci Technol 49(2):81–92. https://doi.org/10.15258/sst.2021.49.2.01
Stefano DE, Tsopmo A, Oliviero T, Fogliano V, Udenigwe CC (2019) Bioprocessing of common pulses changed seed microstructures, and improved dipeptidyl peptidase-IV and α-glucosidase inhibitory activities. Sci Rep 9(1):1–13. https://doi.org/10.1038/s41598-019-51547-5
Tefera T (2002) Allelopathic effects of Parthenium hysterophorus extracts on seed germination and seedling growth of Eragrostis tef. J Agron Crop Sci 188(5):306–310. https://doi.org/10.1046/j.1439-037X.2002.00564.x
Tomacheski D, Pittol M, Simões DN, Ribeiro VF, Santana RMC (2017) Impact of silver ions and silver nanoparticles on the plant growth and soil microorganisms. Glob J Environ Sci Manag 3(4):341–350. https://doi.org/10.22034/gjesm.2017.03.04.001
Tymoszuk A (2021) Silver nanoparticles effects on in vitro germination, growth, and biochemical activity of tomato, radish, and kale seedlings. Materials 14(18):5340. https://doi.org/10.3390/ma14185340
Vishwakarma K, Shweta UN, Singh J, Liu S, Singh VP, Prasad SM, Chauhan DK, Tripathi DK, Sharma S (2017) Differential phytotoxic impact of plant mediated silver nanoparticles (AgNPs) and silver nitrate (AgNO3) on Brassica sp. Front Plant Sci 8:1501. https://doi.org/10.3389/fpls.2017.01501
Wu YX, von Tiedemann A (2002) Impact of fungicides on active oxygen species and antioxidant enzymes in spring barley (Hordeum vulgare L.) exposed to ozone. Environ Pollut 116(1):37–47. https://doi.org/10.1016/S0269-7491(01)00174-9
Xu L, Zhu Z, Sun DW (2021) Bioinspired nanomodification strategies: moving from hemical-based agrosystems to sustainable agriculture. ACS Nano 15(8):12655–12686. https://doi.org/10.1021/acsnano.1c03948
Yuen CWM, Ku SKA, Choi PSR, Kan CW, Tsang SY (2005) Determining functional groups of commercially available ink-jet printing reactive dyes using infrared pectroscopy. Res J Text Appar 9(2):26–38. https://doi.org/10.1108/RJTA-09-02-2005-B004
Acknowledgements
The authors express their sincere thanks to all the faculty members including technical staff of the Department of Environmental Science and University Science Instrumentation Centre (USIC), The University of Burdwan, for their moral and technical support.
Funding
This work was supported by Swami Vivekananda Merit cum Means Fellowship (WBP211642404696), Govt. of West Bengal, West Bengal, India.
Author information
Authors and Affiliations
Contributions
RK contributed to conceptualization, methodology, investigation, formal analysis, writing—original draft. AM contributed to methodology, investigation, formal analysis, writing—review and editing. NKM contributed to conceptualization, formal analysis, resources, writing—review and editing, supervision.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that there is no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Koley, R., Mondal, A. & Mondal, N.K. Green synthesized silver nanoparticles mediated regulation on hydrolytic enzymes and ROS homeostasis promote growth of three pulses (Cicer arietinum L., Pisum sativum L., and Vigna radiata L.). Energ. Ecol. Environ. 8, 537–555 (2023). https://doi.org/10.1007/s40974-023-00293-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40974-023-00293-6