Abstract
Septic systems are potentially significant sources of nitrogen to groundwater and surface water. In-stream practices, such as in-stream bioreactors (IBRs), that promote or enhance nitrogen treatment are promising solutions to reduce nitrogen loads to nutrient-sensitive water. More work is needed to evaluate the efficiency of IBRs in new applications, such as residential sub-watersheds with a high-density of septic systems. The goal of this study was to quantify nitrogen treatment by an in-stream bioreactor (IBR) during baseflow conditions. The IBR was constructed in March 2017 when approximately 1 m of streambed sediment was excavated and backfilled with 0.75 m of woodchips capped by 0.2 m of rotary-kiln, expanded slate and boulder-sized riprap. Samples were collected monthly from July 2017 – March 2019 including IBR inflow, monitoring ports within the IBR, groundwater seeps draining to the IBR, and IBR outflow. Water samples were analyzed for total dissolved nitrogen (TDN), nitrate, ammonium, dissolved organic carbon, chloride, and nitrate isotopic fractionation. The IBR reduced the median concentration of TDN and nitrate by 40% and 77%, respectively. The median mass removal of TDN and nitrate was 26 and 5.2 g day− 1, respectively. Nitrogen-chloride ratios and isotopic fractionation data suggest that denitrification was likely a predominant nitrogen reduction mechanism. In addition to nitrogen treatment, the IBR provided other benefits by improving erosion control, streambank stabilization, and increased bank-full storage from 6 m3 to 19.2 m3. Results indicated that the IBR improved water quality and other residential sub-watersheds with septic systems would benefit from similar practices.
Highlights
Nitrogen (N) from septic systems endangers water quality and aquatic ecosystems.
Novel use of an in-stream bioreactor to treat N from septic systems is presented.
The in-stream bioreactor effectively reduced N concentration and masses.
Nitrogen-chloride ratios and nitrate isotope data suggest denitrification occurred.
Applying this approach in other basins may help meet nitrogen management goals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Eutrophication and its cascading effects (e.g., algal blooms, fish kills, habitat degradation, tourism, and other environmental and socioeconomic damages) continue to pose water quality challenges globally. These challenges have been documented in the United States (Rakhimbekova et al. 2021; Herren et al. 2021), Canada (Robertson et al. 2021; Digaletos et al. 2023), Spain (Pérez-Ruzafa et al. 2019), Ethiopia (Menberu et al. 2021), China (Wang et al. 2019), France (Louis et al. 2023), and other countries (Oliver et al. 2018). Elevated nitrogen inputs from anthropogenic sources have been found to negatively affect river ecosystem functioning (Zhang and Chadwick 2022) and can endanger public health by increasing the risk of methemoglobinemia from consumption of elevated nitrate in drinking water (Raju and Singh 2017). These studies suggest that agriculture, wastewater (centralized and/or decentralized), and/or stormwater can be major sources of nitrogen to water resources.
Septic systems are a decentralized form of wastewater management commonly used in rural communities across the globe. UN-Habitat and the WHO (2021) estimated that 24% of domestic wastewater generated worldwide flowed into a septic tank. Past studies have found that septic systems can be potentially significant sources of nutrients to groundwater and/or surface waters (Oldfield et al. 2020; Buszka and Reeves 2021; Robertson et al. 2021; Rakhimbekova et al. 2021; Herren et al. 2021; Digaletos et al. 2023). Groundwater beneath drainfield trenches tends to be mostly nitrate and may be removed by denitrification beneath and/or hydrologically down-gradient from a drainfield. However, soils that facilitate more complete nitrification typically lack sufficient organic carbon to support denitrification, thus nitrate may leach to groundwater (Lusk et al. 2017). Robertson et al. (2021) found a mean nitrate concentration of 34 ± 27 mg/L in groundwater beneath drainfields from 20 sites. They also determined that nitrate-rich plumes containing concentrations that exceed 10 mg/L can persist for up to 30 m downgradient of septic drainfields. Denitrification can rapidly deplete nitrate concentrations in groundwater if plumes discharge to riparian sediments with sufficient organic carbon (Robertson et al. 2021). However, other studies found that watersheds with a high-density of septic systems (> 1 system ha− 1) contained elevated nitrogen concentrations and/or mass exports at the watershed scale (Hoghooghi et al. 2016; Iverson et al. 2018). Thus, areas lacking sufficient organic carbon would benefit from retrofit best management practices designed to curtail nitrogen transport from septic systems via denitrification, such as streams with degraded or missing riparian buffers that drain residential sub-watersheds with a high density of septic systems.
Denitrifying bioreactors may be a viable option to treat nitrogen from residential wastewater by providing dissolved organic carbon (DOC) to facilitate denitrification. Subsurface denitrifying bioreactors are a commonly used best management practice consisting of a trench, bed, or wall of carbonaceous media (e.g., woodchips) installed within the surficial aquifer to intercept nitrate-rich groundwater or tile drainage. These techniques are commonly used in agricultural settings and past literature reviews have found denitrifying bioreactors can be highly efficient nitrate removers (Addy et al. 2016; Christianson et al. 2021). There is growing interest in evaluating techniques designed to promote or enhance denitrification within streams or drainage ditches. One type of practice is the in-ditch or in-stream bioreactor (IBR), which is an engineered technology that typically includes installation of carbonaceous media in the hyporheic zone of a stream (Robertson and Merkley 2009; Elgood et al. 2010; Christianson et al. 2017; Maxwell et al. 2022) or construction of a bioreactor cell filled with carbonaceous media in the stream/ditch anchored by bulkheads, a wire mesh, and/or rocks (Pfannerstill et al. 2016; Sarris and Burbery 2018; Burbery and Abraham 2022). These studies found that IBRs can be effective strategies to reduce nitrogen concentrations and/or masses in surface waters. Goeller et al. (2020) found that IBRs are particularly effective at reducing nitrogen masses in surface waters during baseflow conditions by facilitating microbial transformations and physical retention. However, they found that IBRs were less effective at treating subsurface water or providing nitrogen treatment during stormflow conditions.
Most of the literature examining field-scale denitrifying bioreactors occurred in agricultural settings. However, some studies have found subsurface denitrifying bioreactors (e.g., permeable reactive barriers, denitrification trenches, denitrification walls, etc.) to be effective at reducing nitrate originating from septic system effluent (Robertson et al. 2008; Humphrey et al. 2015). If failing or compromised septic systems also discharge at or near the surface, permeable reactive barriers may be less effective and there are additional challenges associated with scaling treatment from the lot to watershed scale. Removing nitrate at the watershed scale would likely require most residences to install a denitrification trench, which is likely not cost effective. Although IBRs have been shown to effectively remove fertilizer-derived nitrate in agricultural ditches and streams, few studies have tested the treatment efficiency of a field-scale IBR in removing wastewater-derived nitrogen in suburban settings. Septic systems typically have lower nitrogen loading rates compared to fertilizer application at agriculture settings. However, they remain a potentially significant non-point source of nitrogen to surface waters (Lusk et al. 2017). Septic systems discharge ammonium-rich effluent to subsoils where nitrification occurs if there is sufficient vertical separation distance (i.e., vertical distance from drainfield trench bottom to water table). Thus, malfunctioning septic systems and/or shallow water tables can result in leaching of organic nitrogen and/or ammonium to groundwater (Karathanasis et al. 2006; Lusk et al. 2017), and eventually to streams. Furthermore, residential sub-watersheds typically have different and more diverse land cover classes than agricultural sub-watersheds. Increased development in residential sub-watersheds likely results in increased total impervious surfaces, which can alter the natural water cycle and influence stream hydrology. Thus, potential differences in dominant nitrogen speciation and land cover could influence nitrogen treatment efficiency by IBRs in residential streams recharged by septic system effluent.
The goal of this study was to quantify nitrogen treatment by an IBR during baseflow conditions. The IBR was installed within a residential watershed containing a high density of septic systems. Specific objectives of this study were to: (1) quantify the concentration of nitrogen species in IBR inflow and outflow; (2) estimate the mass removal of nitrogen by the IBR; (3) calculate the treatment efficiency of the IBR; and (4) evaluate potential for denitrification using nitrogen-to-chloride (N/Cl) ratios, isotopic fractionation, and environmental parameters. Given the widespread usage of septic systems globally and their potential to contribute to eutrophication, additional work is needed to evaluate management strategies that have potential to reduce nitrogen concentrations and masses at the watershed-scale. If results from this study indicate that IBRs are effective, then this technology can be easily adapted in other streams draining residential sub-watersheds, especially in catchments with a high-density of septic systems and affordable access to woodchips or other carbonaceous media.
2 Materials and Methods
2.1 Study Area
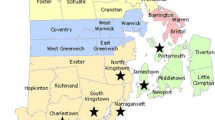
Falls Lake is a reservoir that was impounded in the late 1970s and is in the Piedmont of North Carolina, USA. The Falls Lake Watershed drains approximately 200,000 ha spanning 6 counties in central North Carolina, including Durham, Franklin, Granville, Orange, Person, and Wake Counties. Portions of Falls Lake and its major tributaries are listed on the North Carolina 303(d) list for elevated chlorophyll-a and turbidity, low dissolved oxygen (DO), and/or poor benthic habitat (NC DEQ 2023a). These impairments indicate nutrient enrichment within the Falls Lake Watershed and have led to watershed-scale nutrient reduction efforts (NC DEQ 2023b). The current study occurred within the Lick Creek Watershed and Lick Creek is one of 13 tributaries to Falls Lake (Fig. 1). From 2017 to 2020, mean concentrations of total nitrogen (TN) and chlorophyll-a in the Lick Creek arm of Falls Lake ranged from 0.72 to 0.86 mg L− 1 and 29.8–42.1 µg L− 1, respectively. Furthermore, chlorophyll-a concentrations exceeded water quality standards (> 40 µg L− 1; 15A NCAC 02B .0211) between 0 and 36.4% of the time (NC DEQ 2023b). Currently, North Carolina has not established water quality standards for TN, but monitoring efforts documented that Lick Creek contained TN concentrations that were approximately 2 times greater than reference conditions (streams: 0.4 mg L− 1; lakes: 0.3 mg L− 1) for this ecoregion (US EPA 2000a; 2000b).
The Piedmont geological province is characterized by rolling hills with subsoils originating from Late Proterozoic/Cambrian metamorphic and igneous rocks or Triassic sedimentary rocks. The Falls Lake Watershed is comprised of 3 major geologic settings: the Caroline Terrane, Triassic Basin, and Raleigh Belt (Crabtree, Falls Lake, and Raleigh Terranes) (NC DEQ 2015). The current study occurred within the Triassic Basin where soils formed from weathering of fine-grained red sandstones and/or coarse-grained grey sandstones. The Triassic Basin consists of clayey, saprolitic soils that may exhibit shrink-swell characteristics (e.g., cracks and slickenslides) and may have low hydraulic conductivities (Griffin and Buol 1988).
2.2 Bioreactor Construction
The IBR was installed within a 15.1 ha sub-watershed of Lick Creek that contains 28 septic systems (1.85 systems ha− 1; Fig. 1). This is a residential sub-watershed served entirely by septic systems. The sub-watershed was delineated using USGS (2023) StreamStats 4.0 and consisted of mostly forested (50.0%), developed (44.5%), and herbaceous (5.4%) land cover classes. Collectively, the Mayodan (57.9%) and White Store (25.6%) soil series accounted for > 80% of the sub-watershed. Mayodan (Fine, mixed, semiactive, thermic Typic Hapludults) and White Store (Fine, mixed, active, thermic Oxyaquic Vertic Hapludalfs) typically contain at least 35% clay content at depths of approximately 46 cm with weathered bedrock at its base. Soil texture ranges from sandy clay to sandy loam and clay to sandy loam for Mayodan and White Store, respectively (USDA 2023). The mean annual temperature and precipitation for this region was approximately 15 °C and 122 cm, respectively (NCSCO 2023).
The IBR was installed in a stream receiving drainage from single-family dwellings served by septic systems (Fig. 1). Implementation of the IBR required permission from multiple property owners since several residences bordered the stream. Initially, the plan was to position the IBR immediately adjacent to the sub-watershed outlet (southern portion of the sub-watershed in Fig. 1); however, the property owners in this portion of the sub-watershed were not interested in participating in the study. Thus, the IBR was installed further upstream after acquiring permission from six property owners. This stream is an unnamed, first-order tributary to Lick Creek experiencing substantial erosion causing streambanks to collapse into the stream. During initial meetings with homeowners, there were several requests to modify the design to include stormwater benefits to mitigate localized flooding (e.g., erosion control, installation of riprap instead of stone, streambank stabilization). To incentivize homeowner participation, the design was amended to include these changes which nearly doubled the cost from approximately $23,000 to approximately $40,000.
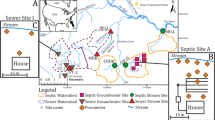
To install the IBR, native sediment was excavated to approximately 1 m deep along a 61 m stream reach. The trench was backfilled with approximately 0.75 m of woodchips and capped by 0.20 m of rotary-kiln, expanded slate, and boulder size riprap was used to stabilize streambanks and prevent erosion of the media (Fig. 2). Before riprap was installed, streambanks were sloped (approximately 3:1) to mitigate erosion and sediment from washing into the IBR. The design flow rate of the IBR was 69.7 L min− 1, which was designed based on stream discharge estimates in a previous study (Iverson et al. 2018). Woodchips were locally sourced, assorted tree species with varying sizes to promote short- and long-term nitrate removal (pre- and post-IBR construction imagery and estimates of woodchip size available in Supplementary Material (SM) Figs. SM1 – SM3). Species of woodchips were not identified, but common trees in the study area include Pinus spp., Acer spp., Quercus spp., and Liquidambar spp. A check-dam was installed immediately upgradient of the IBR to abate energy and facilitate sedimentation of fine-grained sediments upstream of the IBR, which was an issue noted by Robertson and Merkley (2009), Christianson et al. (2017), and Maxwell et al. (2022). Two monitoring ports (P-1 and P-2) were installed in July 2017 within the woodchip layer of the IBR to monitor water levels within the IBR and collect samples. Ports were constructed of polyvinyl chloride (PVC) pipe (3.18 cm diameter) coupled to a screened section of PVC pipe and installed approximately 0.5 m deep. There were also 2 small groundwater seeps (S-1 and S-2) that drained to the downstream portion of the IBR (Fig. 2). A wooden weir was affixed to a preexisting concrete drop box to encourage infiltration into the bioreactor in downstream reaches of the IBR (Fig. 2). Any bypassing water that did not infiltrate into the woodchips flowed along the surface of the IBR and eventually into the drop box. Thus, nitrogen concentrations of bypass water were accounted for in IBR outflow samples.
Cross-sectional diagram of the in-stream bioreactor along its longitudinal axis depicting locations of grab sampling points, groundwater seeps, and monitoring ports (P-1 and P-2) (a). Photos of the seeps (S-1 [b] and S-2 [c]), with S-2 being downstream of S1. A notched wooden weir was installed adjacent to a preexisting concrete culvert to control water level and flow (d)
2.3 Sampling Protocol and Laboratory Analysis
Monthly sampling began in June 2017 (approximately 3 months after installation to allow time to establish) and continued through March 2019 (n = 23). During each sampling event, physicochemical parameters (pH, specific conductance [SC], temperature, DO, oxidation-reduction potential [ORP]) were measured in field using a YSI-556 MultiProbe meter. A Hach turbidimeter was used to measure turbidity. Outlet flow (L min− 1) was measured immediately downstream of the IBR where water flows over a weir into a concrete drop box (Fig. 2). Flow was estimated by using a volumetric bottle and a stopwatch to measure the duration for the bottle to fill to a measured volume. Discharge from groundwater seeps also contributed to IBR flow rate; however, discharge could not be reliably measured at the surface since velocity was minimal and seeps only flowed intermittently. Water samples were collected using clean, polypropylene bottles from the IBR inflow, monitoring ports, groundwater seeps, and IBR outflow (Fig. 2). Bottles were dipped into the stream to collect water samples. Clean, PVC bailers were used to collect water from the monitoring ports installed in the woodchip layer. Samples were stored in an iced cooler and transported to the Environmental Research Laboratory at East Carolina University. Samples were filtered (pore size: 1.5 and 0.7 μm) using vacuum filtration and the filtrate was immediately analyzed or frozen for future analysis. The Environmental Research Laboratory uses glass filters with the forementioned pore sizes as they allow pre-cleaning (combustion in a muffle furnace at 500 °C), have a higher loading capacity, and are more suitable for the SmartChem, which was used to measure nitrate, ammonium, and Cl concentrations. A Shimadzu TNM-L was used to quantify total dissolved nitrogen (TDN) and dissolved organic carbon (DOC) concentrations. A subset of filtrate was shipped to the Stable Isotope Facility at the University of California at Davis for isotopic fractionation of nitrate-nitrogen to enumerate δ15N and δ18O of nitrate, identify potential nitrate sources, and assess if denitrification occurred (Kendall and McDonnell 1998; Silva et al. 2002). Isotopic fractionation samples were only collected in January and February 2018.
2.4 Data and Statistical Analysis
Mass removal of nitrogen was estimated by taking the difference in nitrogen concentration between the IBR inflow and outflow multiplied by the flow at the IBR outflow. Mass removal data were normalized by dividing by the volume of woodchips within the IBR. The surface area of the IBR was estimated using ArcGIS Pro (172 m2). This area was multiplied by the depth of woodchips (0.75 m) to estimate the volume of woodchips within the IBR (129 m3). N/Cl ratios were calculated for each species of nitrogen to better understand possible removal mechanisms for nitrogen. In freshwater environments, Cl can be used to estimate dilution as described by Humphrey et al. (2016). Precipitation data were also downloaded from the NCSCO (2023) and summarized for the sampling period.
Figures and statistical analyses were conducted using the R Statistical Package with the RStudio Interface (R Core Team 2022; Posit Team 2022) using the following packages: readxl (Wickham and Bryan 2022), ggplot2 (Wickham 2016), cowplot (Wilke 2020), and ggbreak (Xu et al. 2021). Data were not normally distributed even after log10 and Box-Cox power transformation, thus non-parametric statistics were used to evaluate differences in comparison groups. For comparison groups with > 2 categorical variables, a Kruskal-Wallis H test was used. If a significant p-value was found (p ≤ 0.05), then post-hoc tests were conducted (Wilcoxon Rank Sum test with the Holm p adjustment factor). For comparisons between 2 variables, a Wilcoxon Rank Sum test was used to test for significance. Spearman correlation was used to assess the significance of relationships between nitrogen treatment and other water quality parameters.
3 Results
3.1 Treatment of Dissolved Nitrogen Species
Water collected from the outflow and within the IBR typically contained lower concentrations of TDN and nitrate relative to IBR inflow (Table 1). IBR outflow contained a median TDN concentration of 1.37 mg L− 1, which was 40% lower than median TDN in IBR inflow. However, inflow and outflow concentrations of TDN did not significantly differ (p = 0.06), which was found from a two-sided test. A significant result (p = 0.01) was found when comparing if outflow TDN concentrations were significantly lower than inflow concentrations. The median concentration of TDN for water within the IBR was also lower than inflow, but the difference was not statistically significant (p = 0.15). The lack of significance in two-sided tests likely occurred due to the elevated inputs of ammonium to the IBR (Table 1). Ammonium was the dominant species of TDN in IBR inflow and outflow, and the IBR was not designed to treat ammonium. However, nitrate concentrations consistently declined after passing through the IBR. Median concentration of nitrate was reduced by 77% between IBR inflow (0.96 mg L− 1) and outflow (0.22 mg L− 1). The median nitrate concentration of water collected from within the IBR was 99% lower than inflow, suggesting near complete removal of nitrate. Nitrate concentrations in the IBR were more than an order of magnitude lower than inflow and outflow (p ≤ 0.01). Additionally, median nitrate concentrations within the IBR remained low throughout the entire study period (see “Port” in Fig. 3) indicating that the IBR consistently reduced nitrate concentrations to < 0.05 mg L− 1. Both TDN (r = 0.70; p < 0.01) and nitrate (r = 0.67; p < 0.01) exhibited a moderate correlation between concentration reduction and inflow concentrations.
Ammonium concentrations typically decreased after passing through the IBR (Table 1). The median concentration of ammonium was 32% lower in IBR outflow compared to inflow (p = 0.74) and outflow concentrations were more variable. Unlike TDN and nitrate, a clear relationship between ammonium treatment and inflow ammonium concentrations was not established (r = 0.11; p = 0.65). Furthermore, ammonium concentrations tended to increase between IBR inflow and water within the IBR. The median ammonium concentration increased by about 0.5 mg L− 1 between these sampling locations, but this difference was not statistically significant (p = 0.09). Despite this increase, the IBR outflow contained a median ammonium concentration that was 52% lower than water within the IBR. Ammonium concentrations exhibited statistically significant differences between IBR outflow and water within the IBR (p < 0.01). Furthermore, a moderate correlation existed between ammonium concentrations within the IBR and treatment efficiency (r = 0.50; p = 0.04).
3.2 Reactor Flow and Nitrogen Mass Removal
Flow measured at the IBR outflow ranged from 0 to 1185 L min− 1 (Fig. 4; Table 1). The IBR was designed to provide nitrogen treatment during baseflow conditions, thus most sampling events occurred during baseflow conditions (< ca. 800 L min− 1). However, there were 2 events (29 Jan and 26 Feb 2018) that coincided with storm conditions resulting in elevated flow (ca. 800–1200 L min− 1). Median reactor flow was 18 L min− 1 and 52% of flow events fell between approximately 10 and 100 L min− 1. During summer months (Jul – Sep), flow decreased to < 2 L min− 1 or surface water levels near the outlet dropped below the weir reducing flow to 0 L min− 1 (Fig. 4). Additionally, the upstream reach of the IBR was dry during 4 sampling events (Jul – Oct 2017; Jul 2018) and water samples could not be collected at the IBR inflow.
The IBR was effective at reducing masses of TDN, nitrate, and ammonium (Fig. 5). The median mass removal of TDN, nitrate, and ammonium was 26, 5, and 13 g day− 1, respectively (Table 1). After normalizing by woodchip volume (129 m3), the median mass removal of TDN, nitrate, and ammonium was 0.20, 0.04, and 0.10 g m− 3 day− 1. Mass removal for the studied nitrogen species was highly variable. TDN, nitrate, and ammonium mass removal ranged from -2362 to 2249 g day− 1 (-18.3 to 17.4 g m− 3 day− 1), -933 to 2831 g day− 1 (-10.2 to 21.9 g m− 3 day− 1), and -1429 to43 g day− 1 (-10.9 to 0.3 g m− 3 day− 1), respectively. Negative values indicated periods when the IBR exported nitrogen, thus there was no removal of nitrogen. This phenomenon occurred 5 times (22% of the time) for TDN and nitrate, whereas this occurred 7 times (30% of the time) for ammonium. The highest mass removal rates were observed during 2 events in February 2018 when concentration difference between inflow and outflow was elevated (> 2 mg L− 1) and temperatures were supportive of microbial activity (approximately 15 °C) (Fig. 3). When inflow nitrate concentrations spiked, nitrate mass removal also typically increased. The inflow nitrogen concentration was positively and significantly correlated with mass removal of TDN (r = 0.75; p < 0.01), nitrate (r = 0.73; p < 0.01), and ammonium (r = 0.49; p = 0.04). The worst nitrogen treatment occurred in January 2018 when a storm passed through the study area resulting in 4.1 cm of precipitation over a 24-hour period before sampling. This storm coincided with the highest flow (1185 L min− 1) and nitrogen concentrations in IBR outflow exceeded IBR inflow. Nitrate concentrations within the IBR remained low (Fig. 3), indicating that nitrate treatment within the IBR was unaffected. However, elevated nitrogen concentrations in IBR outflow suggested water likely bypassed the IBR resulting in nitrogen export. The opposite trend in nitrogen treatment occurred during the 26 February 2018 storm; however, the precipitation amount was substantially lower (0.23 cm). Thus, storms that generate more than 0.23 cm of precipitation may negatively affect nitrogen treatment. During the sampling period, there were 137 days (21.4% of the sampling period) where the daily precipitation exceeded 0.23 cm. There were only 9 days (1.4% of the sampling period) where daily rainfall exceeded 4 cm (similar storm as January 2018). The IBR was not designed to provide nitrogen treatment during storms, thus it is likely that nitrogen export occurred during larger storms, but more research is needed to confirm. While storms likely negatively affected nitrogen treatment, the IBR provided other stormwater management benefits to the residential neighborhood which are explained in Sect. 4.3.
Boxplot of nitrogen mass removal by the in-stream bioreactor. Negative values indicate when nitrogen was exported from the bioreactor. Note that the y-axis contains several breaks to visualize high and low outliers (*). TDN = total dissolved nitrogen; NO3 = nitrate-nitrogen; and NH4 = ammonium-nitrogen
3.3 Nitrogen/Chloride Ratios and Nitrate Isotopic Fractionation
Ratios of N/Cl suggested that mass removal processes occurred within the IBR (Table 2). Decreases in nitrogen-to-chloride ratios suggest that processes other than dilution and dispersion contributed to reductions in nitrogen concentration. Nitrate/Cl ratios decreased by a median 70% between IBR inflow and outflow. When comparing to the median concentration reduction (77%), nitrate/Cl ratio reductions suggest that mass removal processes accounted for most of nitrate reduction, presumably due to denitrification. The median ratio of TDN/Cl ratio was approximately 20% between IBR inflow and outflow (Table 2), which was about half of the median concentration reduction of TDN (40%). These data suggest that mass removal processes were substantive in TDN and nitrate reductions. The ammonium/Cl ratio did not change much between IBR inflow and outflow suggesting that mass reductions of ammonium were minimal. Nitrate isotopic data also support that denitrification occurred within the IBR. The median δ15N and δ18O values were approximately 1.7 and 8, respectively, times greater in the IBR outflow compared to inflow.
3.4 Dissolved Organic Carbon and Physicochemical Parameters
DOC concentrations were similar (p = 0.80) between inflow and outflow (Table 1). The greatest DOC concentrations were observed in IBR outflow and the port from Jul – Sep 2017 (Fig. 3). The IBR inflow location was dry during this period, thus the DOC concentration could not be quantified at the inlet. Carbon-to-nitrogen ratios suggest that the IBR contained an abundance of carbon to facilitate microbial processes that remove nitrogen masses (Table 1). These data also show that water within the IBR was nitrate-limited throughout much of the study period. The median ratio of carbon-to-nitrate was > 12 for all sampling locations (except for the seep), with the highest being within the IBR.
Environmental factors within the IBR and in IBR outflow suggest that low DO conditions occurred (Table 3). IBR inflow contained a median DO concentration of 6.3 mg L− 1, whereas water within the IBR and downstream of the IBR contained median DO concentrations that were approximately 50% lower. DO concentrations tended to be < 5 mg L− 1 within the IBR and in IBR outflow (Table 3). Concentrations of DO were observed to be < 5 mg L− 1 in water within the IBR and IBR outflow on approximately 71% and 80% of sampling events, respectively. There was 1 event where DO concentrations exceeded 10 mg L− 1 (Jan 2018) at all sampling locations, which may indicate that new water entering the IBR bypassed the woodchips thereby inhibiting treatment. The interquartile range of DO for IBR inflow, water within the IBR, and IBR outflow was 5.46–7.64 mg L− 1, 2.7–4.18 mg L− 1, and 1.8–6.6 mg L− 1, respectively. Thus, DO concentrations suggest that reducing conditions were common within the IBR and in IBR outflow, which was also supported by ORP data. ORP values substantially declined from IBR inflow to outflow from 13 to -50 mV indicating that reducing conditions occurred. The remaining parameters (temperature, SC, pH, and turbidity) were similar between IBR inflow and outflow. Mass removal of TDN (r = 0.15; p = 0.54), nitrate (r = 0.30; p = 0.22), and ammonium (r= -0.23; p = 0.36) did not exhibit a strong correlation with temperature suggesting that nitrogen treatment was not temperature-limited.
3.5 Groundwater Seeps
Groundwater seeps can be potentially significant sources of nitrogen (Table 1). The median concentration of TDN in Seep 1 was 8.77 mg L− 1, which was nearly 4 times greater than IBR inflow, and this difference was significant (p = 0.02). Ammonium was typically the dominant species of TDN in Seep 1 (median: 2.64 mg L− 1). Furthermore, the low median concentration of nitrate in Seep 1 suggest that DON was also a dominant species of TDN (Table 1). Seep 1 also contained elevated DOC concentrations (median: 18.60 mg L− 1) relative to all other sampling locations (Table 1). These data may indicate that some septic systems in the sub-watershed are not adequately nitrifying effluent beneath the drainfield trenches, likely resulting in leaching of ammonium and DON to groundwater. Seep 2 exhibited different trends in both concentration and speciation of nitrogen relative to Seep 1 (Table 1). Both Seep 2 and IBR inflow contained similar concentrations of TDN (p = 0.38) with median concentrations of approximately 2.2 mg L− 1. Most of the TDN observed in Seep 2 occurred as nitrate with a median of 2.11 mg L− 1. Additionally, DOC concentrations were similar between Seep 2 and IBR inflow (median: 8.04 mg L− 1). These data suggest that septic systems drained by Seep 2 are adequately nitrifying wastewater effluent before discharging to the seep. However, Seep 2 discharges into the IBR approximately 15 m upstream from the IBR outlet structure, which may reduce performance by contributing additional nitrogen loads between the inlet and outlet.
4 Discussion
4.1 Quantifying Nitrogen Treatment
Nitrogen concentrations typically declined after passing through the IBR. The median concentration of TDN in IBR inflow was 2.27 mg L− 1 and reduced to 1.37 mg L− 1 in the outflow (median reduction: ca. 40%). Treatment of dissolved inorganic nitrogen (DIN; sum of ammonium and nitrate) was slightly greater, with a median concentration reduction of 46%. The median concentration of DIN declined from 2.08 mg L− 1 in IBR inflow to 1.13 mg L− 1 in the outflow. The median concentration of DON was estimated by taking the difference in TDN and DIN, and concentrations in IBR inflow and outflow were both approximately 0.20 mg L− 1. These data indicate that DON was unreactive within the IBR, which was expected. Thus, most of the nitrogen treatment occurred due to reduction of DIN species. Inflow typically contained similar concentrations of nitrate and ammonium with a median of 0.96 mg L− 1 and 1.05 mg L− 1, respectively. Concentrations of nitrate and ammonium typically decreased in IBR outflow, with median values of 0.22 mg L− 1 and 0.72 mg L− 1, respectively. The IBR was most effective at reducing nitrate concentrations, which aligned with past research focusing on bioreactors designed to treat nitrogen.
Most of these studies focused on quantifying nitrate treatment in agricultural settings. Overall, IBRs (or in-ditch bioreactors) exhibited variability in nitrate concentration reductions ranging from -1 to 78% (Robertson and Merkley 2009; Elgood et al. 2010; Pfannerstill et al. 2016; Sarris and Burbery 2018; Christianson et al. 2017; Maxwell et al. 2022). Three of these studies (Robertson and Merkley 2009; Elgood et al. 2010; Christianson et al. 2017) found that nitrate concentration reductions tended to be > 50%. Robertson and Merkley (2009) reported a 78% reduction of nitrate concentrations from 4.8 to 1.04 mg L− 1. Elgood et al. (2010) studied the same IBR as Robertson and Merkley (2009) but during a different period (2008–2009). They found that the mean concentration reduction of nitrate was 53% between the inflow and outflow (2.8 to 1.3 mg L− 1). Christianson et al. (2017) reported a 65% reduction in nitrate concentrations from 2.53 to 0.88 mg L− 1 between the inlet and outlet, respectively. Pfannerstill et al. (2016) found nitrate concentration reductions to be more variable ranging from 7 to 41%, with an overall mean nitrate concentration reduction of 28% (8.87 to 6.43 mg L− 1). Maxwell et al. (2022) reported the lowest mean reductions of nitrate concentrations at -1% ± 16%. Mean nitrate concentrations in the inlet and outlet were 5.9 ± 2.4 mg L− 1 and 6.1 ± 2.2 mg L− 1, respectively. In the current study, the median and mean nitrate concentration in IBR outflow was 77% and 61% lower than inflow, respectively and this aligned with past studies. The median (0.96 mg L− 1) and mean (1.00 mg L− 1) nitrate concentration in IBR inflow was lower than the forementioned studies. Furthermore, nitrate concentrations in water sampled from within the IBR (e.g., Port 1 and 2 in Table 1; Fig. 3) were typically < 0.05 mg L− 1. Thus, the similarity in concentration reductions and the low nitrate concentration within the IBR suggests that this practice was effective at treating low inflow concentrations of nitrate during baseflow.
Median mass removal of TDN, nitrate, and ammonium ranged from about 5–26 g day− 1 (0.04–0.20 g m− 3 day− 1). DON concentrations were similar between IBR inflow and outflow, thus TDN and DIN mass removal was similar. Most studies focused on quantifying nitrate treatment by IBRs and found that mass removal ranged from 4 to 1496.4 g-NO3−-N day− 1 or 0.3 to 37.4 g-NO3−-N m− 3 day− 1 (Robertson and Merkley 2009; Elgood et al. 2010; Pfannerstill et al. 2016; Maxwell et al. 2022). In the current study, the median nitrate removal rate was 5.2 g day− 1 (0.04 g m− 3 day− 1). The median daily nitrate mass removal within range of past studies; however, the volume-normalized nitrate removal rate was much lower. In the forementioned studies, the mean nitrate concentration in inflow ranged from 2.53 to 8.87 mg L− 1, which was 2.5–8.9 times greater than the mean inflow concentration in the current study. Inflow nitrate concentrations in this study’s IBR were typically < 2 mg L− 1, only exceeding this on 2 occasions. When inflow nitrate concentrations exceeded 2 mg L− 1, the median mass removal of nitrate and TDN was 1777.8 g day− 1 (13.8 g m− 3 day− 1) and 1373 g day− 1 (10.6 g m− 3 day− 1), respectively. The decline in mass reduction when comparing nitrate and TDN on these 2 sampling events indicated that the IBR was a source of ammonium and/or DON. There were 9 occasions when inflow nitrate was > 1 mg L− 1 and median mass removal was 16.4 g day− 1 (0.13 g m− 3 day− 1). On these days, the median mass removal of TDN was 38.4 g day− 1 (0.3 g m− 3 day− 1). Thus, the nitrogen removal rate is likely inhibited during periods of nitrate-limitation (concentrations < 1 mg L− 1). A similar finding was reported by Elgood et al. (2010) where they found that low nitrate concentrations (< 1 mg L− 1) also limited nitrate removal rates. Furthermore, Addy et al. (2016) reported diminished nitrogen treatment by subsurface denitrifying bioreactors during periods of nitrate limitation.
N/Cl ratio and isotopic fractionation of nitrate data suggest that mass removal processes (i.e., all nitrogen reduction pathways except for dilution and dispersion) accounted for a substantial portion of nitrogen treatment. Differences in the nitrate/Cl ratio between IBR inflow and outflow imply that mass removal processes accounted for most of the nitrate reductions (Table 2). TDN/Cl ratios suggest mass removal processes accounted for 20% of TDN reduction, which is congruent with past literature. Bryant et al. (2019) recommended that properly installed and maintained denitrifying bioreactors receive a 20% TN load reduction credit based on a review of numerous denitrifying bioreactors. Isotopic fractionation of nitrate suggest that denitrification could account for nitrate mass removal (Kendall and McDonnell 1998; Silva et al. 2002; Hu et al. 2020). Hu et al. (2020) found an inverse relationship between nitrate concentrations and values of δ15N. The current study found a similar trend on 16 Feb 2018 when nitrate treatment was high. IBR outflow contained δ15N and δ18O values that increased by 8.95 and 7.19‰, respectively. The nitrate concentration reduction on this same event was 84.7% (2.75 to 0.42 mg L− 1) and mass removal was 725 g day− 1 (5.6 g m− 3 day− 1). However, this trend was not observed on 29 Jan 2018 when treatment was inhibited. IBR outflow (δ15N: 4.70‰; δ18O: 1.87‰) contained similar values of δ15N and δ18O as IBR inflow (d15N: 3.26‰; d18O: 1.84‰). Nitrate concentration reduction on this day was -53.1% (1.03 to 1.58 mg L− 1) and mass removal of nitrate was -933 g day− 1 (-7.2 g m− 3 day− 1) indicating that the IBR exported nitrate during this storm. Both DO concentration and flow rate were also highest during this sampling event. Collectively, these data likely indicate that runoff bypassed the IBR by flowing through the channel without entering underlying media and/or displaced water within the IBR thereby reducing treatment. More research is necessary to assess hydraulic retention time and flow dynamics under varying flow regimes, particularly during storms that generate high flows (> 800 L min− 1).
4.2 Nitrogen Transformation and Controlling Factors
There are several nitrogen transformation processes that may explain nitrogen treatment occurring in the IBR. The IBR was engineered to facilitate denitrification by installing woodchips within the hyporheic zone of a stream. Denitrification can be an effective nitrate removal process, especially in reducing environments with available carbon and adequate temperatures (Robertson and Merkley 2009; Elgood et al. 2010; Addy et al. 2016; Pfannerstill et al. 2016; Goeller et al. 2020; Christianson et al. 2021). Zhi and Ji (2014) found that a C/N ratio of > 6 was necessary to facilitate complete denitrification. The current study contained a median DOC/nitrate ratio of 12 in inflow water, thus denitrification was not likely limited by availability of DOC. Past studies have established that temperature can be a limiting factor for denitrification, especially if temperatures routinely decline below 10 °C or seasonally freeze (Robertson and Merkley 2009; Elgood et al. 2010; Addy et al. 2016; Pfannerstill et al. 2016; Goeller et al. 2020). Pfannerstill et al. (2016) found that nitrate mass removal rates tended to improve when temperatures were high (5–15 °C, especially > 10 °C) and stream discharge was lower (< 6.7 L min− 1). Temperature rarely decreased below 10 °C on sampling days in the current study (Fig. 3) and there was not a strong association between mass removal and temperature, which likely occurred because the study location is in a milder climate than some other studies. These results suggest that denitrification could have been a primary driver of nitrate treatment.
Another pathway for nitrate transformation could be due to dissimilatory nitrate reduction to ammonium (DNRA). Ammonium concentrations within the IBR increased by about 44% between IBR inflow and water within the IBR. DNRA can occur when the carbon-to-nitrogen ratio is high (e.g., > 12) and in reducing environments where nitrate is limited as an electron acceptor (Healy et al. 2015; Fenton et al. 2016). Water within the IBR exhibited a sharp increase in the DOC/nitrate ratio indicating that nitrate limitation commonly occurred within the IBR. Thus, DNRA cannot be discounted at this time. It is also possible that the increase in ammonium concentrations could be due to inputs from groundwater seeps and/or ammonification of DON. Past studies found elevated ammonium concentrations in outflow from denitrifying bioreactors, which was attributed to mineralization of DON from decay of carbonaceous material (Fenton et al. 2016; Pfannerstill et al. 2016; Manca et al. 2021). Most studies on IBRs reported similar ammonium concentrations between inflow and outflow (Robertson and Merkley 2009; Elgood et al. 2010; Maxwell et al. 2022; Burbery and Abraham 2022) suggesting that DNRA and biodegradation was minimal. These studies reported low ammonium inputs to the IBR, whereas in the current study ammonium inputs were comparable to nitrate. This likely explains the elevated ammonium concentrations in outflow (> 0.3 mg-NH4 L− 1) compared to other studies. Seep 1 contained elevated concentrations of ammonium likely originating from malfunctioning septic systems. Past studies found elevated ammonium in water downgradient from septic systems experiencing malfunction or those with insufficient vertical separation distance between the trench bottom and water table (Karathanasis et al. 2006; Humphrey et al. 2010; O’Driscoll et al. 2014). This study area is characterized by Triassic Basin soils, which have reduced infiltrative capacity that may contribute to inadequate nitrification within drainfields before reaching groundwater (Humphrey et al. 2016).
Ammonium concentrations in outflow tended to be lower than concentrations in IBR inflow and water within the IBR. It is possible that anaerobic ammonium oxidation (anammox) accounted for some ammonium treatment. Smith et al. (2015) found anammox can readily occur in anaerobic, freshwater aquifers and this process is not reliant upon DOC. Thus, ammonium can be directly converted to dinitrogen or nitrous oxide without first being nitrified to nitrate and later denitrified. It is possible that mass removal of ammonium may be attributed to anammox, but more research is needed to confirm. Quantification of enzymatic expressions from microbiological processes would allow confirmation and differentiation of the extent to which denitrification, DNRA, and/or anammox occurred within the IBR (Sparacino-Watkins et al. 2014; Smith et al. 2015; Liu et al. 2021). Furthermore, partnering these efforts with microbial community assemblage efforts would also strengthen understanding of nitrogen transformations.
Nitrogen treatment by the IBR in the current study likely would have been better if groundwater seeps were not present or if the IBR were sited further downstream. Groundwater seeps can be conduits that deliver nitrogen and other pollutants to surface waters (O’Driscoll et al. 2019). Maxwell et al. (2022) reported low nitrate treatment by an in-ditch bioreactor, which they surmised could have been due to intermittent contributions of nitrate from adjacent tile drainage. They found nitrate concentrations in the tile drain to be significantly higher than concentrations upstream of the bioreactor. In the current study, groundwater seeps played a similar role where TDN and ammonium/nitrate concentrations were elevated compared to IBR inflow (Table 1). While the overall nitrogen treatment was effective, concentration and mass reduction estimates do not account for these additional sources, thus nitrogen treatment was underestimated. Future studies adopting similar technologies should investigate the presence of groundwater seeps and, if present, quantify nitrogen concentrations. These seeps may flow seasonally, thus identification efforts should occur during wetter periods. However, it is also important to acknowledge that site selection can be constrained by homeowner preference. The authors contend that the ideal placement for the IBR would have been further downstream near the sub-watershed outlet. Before the IBR was installed, the median TDN concentration at the sub-watershed outlet was 2.86 mg L− 1 (n = 19), which was mostly nitrate (median: 2.05 mg L− 1, n = 19) (Iverson 2019). Thus, an IBR at this location would have likely performed better. However, implementation of engineered solutions on private property requires support from landowners, which can offer a unique set of challenges that affect IBR placement. While the location of the IBR in the current study may not have been optimal for nitrogen treatment, this location provided other environmental benefits.
4.3 Evaluating Other Benefits
Additional environmental benefits provided by the IBR include increased storage capacity, erosion control, and flood management. Before installing the IBR, erosion was undercutting the streambanks and homeowners mentioned issues with nuisance flooding during storms. To mitigate erosion, the streambanks were smoothed and sloped at approximately 3:1. Riprap was installed to stabilize streambanks and prevent underlying media from washing away (Figs. SM1 – SM2). Additionally, the storage capacity of the IBR substantially increased after installing the IBR. To analyze bank-full storage, 4 cross-sections analyses were conducted along the stream reach. The mean volume of bank-full storage before the IBR was about 6 m3, which increased to 19.2 m3 after the IBR was completed (Table SM1). Furthermore, one homeowner informed researchers that the IBR was effective at reducing localized flooding. The homeowner reported that a storm passed through the study area shortly after IBR construction and noted that the stream did not enter flood stage. This homeowner mentioned that past storms with similar intensity caused localized flooding within the neighborhood. Reduced flooding and deeper groundwater associated with drainage improvements could have also increased vertical separation distance and treatment effectiveness of adjacent septic systems. More research is warranted to assess how IBRs may affect stormwater management, especially in nutrient-sensitive sub-watersheds with a suspected or known source of nitrate.
4.4 Study Limitations
While results from this study show promise for using IBRs to improve nitrogen treatment in residential sub-watersheds served by septic systems, this study was not without its limitations. Past studies (Maxwell et al. 2020; Qi et al. 2022) recommend high frequency sampling to improve efforts to quantify water quality parameters. Maxwell et al. (2020) found that concentration reductions of nitrate varied by up to 25% within a single event. Furthermore, they found that high frequency sampling improved estimates of residence time and volumetric removal rates, especially if the bioreactor has long hydraulic retention time and significant lag between inlet and outlet. Similarly, Qi et al. (2022) also found that lower frequency sampling could underestimate pollutant concentrations by missing large events. However, they also state that high frequency sampling can also result in positive relative bias due to overrepresentation of large events. In the current study, the worst treatment by the IBR occurred during a 4.1 cm storm in January 2018, which could have been due to bypass flow or low frequency sampling. The mechanism behind this reduced treatment is unknown at this point. It is possible that this storm event mobilized nitrate from nearby septic systems and conveyed elevated concentrations to the IBR (Pouye et al. 2023). If this occurred and the IBR inlet sample was collected after the nitrate pulse entered the IBR, that could explain the poor treatment on that day. It was beyond the scope of the current study to evaluate storm influences on nitrogen treatment, although future analysis should incorporate high-frequency monitoring to assess nitrogen delivery to the IBR during baseflow and storms at finer temporal resolutions.
High-frequency data collection would also improve understanding of flow dynamics within the IBR by allowing estimation of the percentage of annual flow that the IBR treated. The authors initially planned to deploy pressure transducers to monitor water levels at the IBR inlet, within the IBR, and at the IBR outlet. However, this effort became too costly due to modifications to the IBR design. During meetings with the property owners, there were several requests to strengthen erosion control and streambank stabilization, which nearly doubled the cost of the IBR. Thus, the high frequency monitoring component of the project was cut to cover the increased costs. However, about a year into the project, the authors received an expansion grant and installed a Solinst Levelogger in the IBR outlet from June – December 2018, which was programmed to record water level every 20 min. A total of 13,030 observations were collected during this 7-month period. A moderate, exponential relationship (y = 0.1706 * 2.720.1006x; R2 = 0.76; Fig. SM4) was found between flow rate and water level at the IBR outlet. This equation was used to estimate the water level when the IBR exceeds the design flow of 69.7 L min− 1, which is 57.2 cm. There were 296 observations where water level exceeded 57.2 cm, which represented 2.3% of the observations during the 7-month period. When flow rate exceeds the design flow, there is an increased likelihood for reduced nitrogen treatment from bypass flow. During this same period, 36.3% of the water level observations were ≤ 24.1 cm which is at the base of the notch in the weir. Thus, no outflow occurred which likely promoted more complete denitrification (similar to findings by Elgood et al. (2010). The percentage of the annual flow resulting in bypass is likely underestimated since the logger deployment period did not include most of the wetter, winter season (November–March). Partnering deployable multiprobe sondes that can autonomously log water quality parameters (e.g., nitrate, ammonium, temperature, DO, SC, ORP) alongside water level loggers is recommended to better characterize variability in flow rate and other environmental conditions that affect nitrogen treatment.
5 Conclusions
The current study assessed the efficiency of an IBR retrofitted within a watershed with a high density of septic systems. Denitrifying bioreactors and IBRs have potential to be effective management solutions to remediate elevated TDN from septic system effluent; however, there is a lack of field-scale studies that evaluated the efficiency of these technologies in areas served by septic systems. Results from this study indicated that the IBR was successful at reducing concentrations and masses of TDN, mostly via nitrate reductions. While nitrogen treatment exhibited variability, the IBR performed best when inflow concentrations of nitrate exceeded 2 mg L− 1. Water quality and environmental parameters suggested that several nitrogen transformation pathways could be responsible for treatment, which include denitrification. Furthermore, IBRs also have potential to provide other benefits (e.g., erosion control, stormwater management) beyond nitrogen treatment that warrant future investigation.
There are limited options for improving nitrogen treatment performance of septic systems, especially in nutrient-sensitive watersheds. An IBR is one potential approach for low-order streams draining suburban watersheds where retrofit options may be limited by land availability or cost. There are numerous low-lying areas with low-order streams, conveyance ditches, canals, tile drains, or other hydrologic features that could be retrofitted with IBRs to attenuate nitrogen transport to nutrient-sensitive waters. Nitrogen treatment will likely be optimized by siting IBRs in downstream reaches of watersheds near the outlet. Geographic information system approaches can be used to identify low-order streams that drain to nutrient-sensitive waters that may serve as ideal retrofit candidates. Similar studies should be conducted in other geologic settings to assess treatment efficiency of IBRs that receive drainage from septic systems, especially in streams with elevated nitrate concentrations. It is likely that greater concentration reductions and mass removal would occur if near complete nitrification occurred prior to discharging to the IBR. Additionally, future work should evaluate nitrogen concentrations and flow rates at finer temporal scales to better assess variability in nitrogen treatment under different flow conditions (e.g., baseflow vs. storms). Finally, long-term performance of IBRs should be studied by assessing DOC availability over time as denitrification may become carbon-limited.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Addy K, Gold AJ, Christianson LE, David MB, Schipper LA, Ratigan NA (2016) Denitrifying bioreactors for Nitrate removal: a Meta-analysis. J Environ Qual 45:873–881. https://doi.org/10.2134/jeq2015.07.0399
Bryant R, Baldwin A, Cahall B, Christianson L, Jaynes D, Penn C, Schwartz S (2019) Best Management Practices for Agricultural Ditch Management in the Phase 6 Chesapeake Bay Watershed Model. Report No. CBP/TRS-326-19, Chesapeake Bay Program https://d18lev1ok5leia.cloudfront.net/chesapeakebay/documents/ag_ditches_bmp_panel_report_draft_for_cbp_review_04sep2019.pdf Accessed on 28 April 2023
Burbery L, Abraham P (2022) Results from an In-Stream Woodchip Denitrifying Bioreactor Field Trial in South Canterbury. Annual Farmed Landscapes Research Centre Workshop 11 February 2022, Online Zoom Webinar, conference paper http://flrc.massey.ac.nz/publications.html Accessed 3 April 2024
Buszka TT, Reeves DM (2021) Pathways and timescales associated with nitrogen transport from septic systems in coastal aquifers intersected by canals. Hydrogeol J 29:1953–1964. https://doi.org/10.1007/s10040-021-02362-8
Christianson LE, Collick AS, Bryant RB, Rosen T, Bock EM, Allen AL, Kleinman PJA, May EB, Buda AR, Robinson J, Folmar GJ, Easton ZM (2017) Enhanced denitrification bioreactors hold Promise for Mid-atlantic Ditch Drainage. Ag Environ Lett 2:1–5. https://doi.org/10.2134/ael2017.09.0032
Christianson LE, Cooke RA, Hay CH, Helmers MJ, Feyereisen GW, Ranaivoson AZ, McMaine JT, McDaniel R, Rosen TR, Pluer WT, Schipper LA, Dougherty H, Robinson RJ, Layden IA, Irvine-Brown SM, Manca F, Dhaese K, Nelissen V, von Ahnen M (2021) Effectiveness of denitrifying bioreactors on Water Pollutant reduction from Agricultural Areas. T ASABE 64:641–658. https://doi.org/10.13031/trans.14011
R Core Team (2022) R: A language and environment for statistical computing, (Version: 4.2.2). R Foundation for Statistical Computing: Vienna, Austria. URL: https://www.R-project.org/
Digaletos M, Ptacek CJ, Thomas J, Liu Y (2023) Chemical and biological tracers to identify source and transport pathways of septic system contamination to streams in areas with low permeability soils. Sci Total Environ 870:161866. https://doi.org/10.1016/j.scitotenv.2023.161866
Elgood Z, Robertson WD, Schiff SL, Elgood R (2010) Nitrate removal and greenhouse gas production in a stream-bed denitrifying bioreactor. Ecol Engi 36:1575–1580. https://doi.org/10.1016/j.ecoleng.2010.03.011
Fenton O, Healy MG, Brennan FP, Thornton SF, Lanigan GJ, Ibrahim TG (2016) Holistic evaluation of field-scale denitrifying bioreactors as a basis to Improve Environmental sustainability. J Environ Qual 45:788–795. https://doi.org/10.2134/jeq2015.10.0500
Goeller BC, Febria CM, McKergow LA, Harding JS, Matheson FE, Tanner CC, McIntosh AR (2020) Combining tools from Edge-of-Field to In-Stream to attenuate reactive Nitrogen along Small Agricultural waterways. Water 12:383. https://doi.org/10.3390/w12020383
Griffin RW, Buol SW (1988) Soil and saprolite characteristics of Vertic and Aquic Hapludults derived from Triassic Basin sandstones. Soil Sci Soc Am J 52:1094–1099. https://doi.org/10.2136/sssaj1988.03615995005200040037x
Habitat UN (2021) WHO Progress on wastewater treatment - Global status and acceleration needs for SDG indicator 6.3.1. United Nations Human Settlements Programme (UN-Habitat) and World Health Organization (WHO) https://unhabitat.org/sites/default/files/2021/08/sdg6_indicator_report_631_progress_on_wastewater_treatment_2021_english_pages.pdf Accessed on 7 April 2024
Healy MG, Barrett M, Lanigan GJ, João Serrenho A, Ibrahim TG, Thornton SF, Rolfe SA, Huang WE, Fenton O (2015) Optimizing nitrate removal and evaluating pollution swap** trade-offs from laboratory denitrification bioreactors. Ecol Engi 74:290–301. https://doi.org/10.1016/j.ecoleng.2014.10.005
Herren LW, Brewton RA, Wilking LE, Tarnowski ME, Vogel MA, Lapointe BE (2021) Septic systems drive nutrient enrichment of groundwaters and eutrophication in the urbanized Indian river Lagoon, Florida. Mar Pollut Bull 172:112928. https://doi.org/10.1016/j.marpolbul.2021.112928
Hoghooghi N, Radcliffe DE, Habteselassie MY, Clarke JS (2016) Confirmation of the Impact of Onsite Wastewater Treatment Systems on Stream Base-Flow Nitrogen Concentrations in Urban Watersheds of Metropolitan Atlanta, GA. J Environ Qual 45:1740–1748. https://doi.org/10.2134/jeq2016.04.0139
Hu Y, ** Z, Hu Q, Hu J, Ni C, Li F (2020) Using stable isotopes to identify nitrogen transformations and estimate denitrification in a semi-constructed wetland. Sci Total Environ 720:137628. https://doi.org/10.1016/j.scitotenv.2020.137628
Humphrey CP, O’Driscoll MA, Zarate MA (2010) Controls on groundwater nitrogen contributions from on-site wastewater systems in coastal North Carolina. Water Sci Technol 62:1448–1455. https://doi.org/10.2166/wst.2010.417
Humphrey CP, Pradhan S, Bean E, O’Driscoll M, Iverson G (2015) Preliminary evaluation of a Permeable Reactive Barrier for Reducing Groundwater Nitrate Transport from a large Onsite Wastewater System. Am J Environ Sci 11:216–226. https://doi.org/10.3844/ajessp.2015.216.226
Humphrey CP, Jernigan J, Iverson G, Serozi B, O’Driscoll M, Pradhan S, Bean E (2016) Field evaluation of Nitrogen Treatment by Conventional and single-pass sand Filter Onsite Wastewater Systems in the North Carolina Piedmont. Water Air Soil Pollut 227:1. https://doi.org/10.1007/s11270-016-2958-0
Iverson G (2019) Nutrient contributions from septic systems in nutrient-sensitive watersheds: Quantifying nutrient inputs, reduction methods, and economic feasibility. Dissertation, East Carolina University
Iverson G, Humphrey CP, O’Driscoll MA, Sanderford C, Jernigan J, Serozi B (2018) Nutrient exports from watersheds with varying septic system densities in the North Carolina Piedmont. J Environ Manag 211:206–217. https://doi.org/10.1016/j.jenvman.2018.01.063
Karathanasis AD, Mueller TG, Boone B, Thompson YL (2006) Nutrient removal from septic effluents as affected by soil thickness and texture. J Water Health 4:177–195. https://doi.org/10.2166/wh.2006.067
Kendall C, McDonnell JJ (1998) Isotope tracers in catchment hydrology. Elsevier, Amsterdam
Liu S, Dai J, Wei H, Li S, Wang P, Zhu T, Zhou J, Qiu D (2021) Dissimilatory Nitrate reduction to ammonium (DNRA) and denitrification pathways are leveraged by Cyclic AMP Receptor Protein (CRP) Paralogues Based on Electron Donor/Acceptor Limitation in Shewanella loihica PV-4. Appl Environ Microb 87:1. https://doi.org/10.1128/AEM.01964-20
Louis J, Ballu S, Rossi N, Lasbleiz M, Perrot T, Daniel C, Cellier L, Hénaff F, Richier S (2023) Multi-year renewal of green tides: 18 years of algal mat monitoring (2003–2020) on French coastline (Brittany region). Mar Pollut Bull 193:115173. https://doi.org/10.1016/j.marpolbul.2023.115173
Lusk MG, Toor GS, Yang Y, Mechtensimer S, De M, Obreza TA (2017) A review of the fate and transport of nitrogen, phosphorus, pathogens, and trace organic chemicals in septic systems. Crit Rev Env Sci Tec 47:455–541. https://doi.org/10.1080/10643389.2017.1327787
Manca F, Wegscheidl C, Robinson R, Argent S, Algar C, De Rosa D, Griffiths M, George F, Rowlings D, Schipper L, Grace P (2021) Nitrate removal performance of denitrifying woodchip bioreactors in Tropical climates. Water 13:3608. https://doi.org/10.3390/w13243608
Maxwell BM, Birgand F, Schipper LA, Barkle G, Rivas AA, Helmers MJ, Christianson LE (2020) High-frequency, in situ sampling of field woodchip bioreactors reveals sources of sampling error and hydraulic inefficiencies. J Environ Manag 272:110996. https://doi.org/10.1016/j.jenvman.2020.110996
Maxwell BM, Christianson L, Cooke RAC, Foltz M, Wickramarathne N, Chacon R, Christianson R (2022) Nitrate removal and Woodchip Properties across a paired denitrifying bioreactor treating Centralized Agricultural Ditch flows. Water 14:56. https://doi.org/10.3390/w14010056
Menberu Z, Mogesse B, Reddythota D (2021) Evaluation of water quality and eutrophication status of Hawassa Lake based on different water quality indices. Appl Water Sci 11:1–10. https://doi.org/10.1007/s13201-021-01385-6
NC DEQ (2023b) Falls and Jordan Lakes Monitoring. North Carolina Department of Environmental Quality. https://www.deq.nc.gov/about/divisions/water-resources/water-sciences/intensive-survey-branch/falls-and-jordan-lakes-monitoring Accessed 17 April 2023
NC DEQ (2023a) Water Quality Data Assessment. North Carolina Department of Environmental Quality. https://www.deq.nc.gov/about/divisions/water-resources/water-planning/modeling-assessment/water-quality-data-assessment/integrated-report-files Accessed 17 April 2023
NC DEQ (2015) The Geology of North Carolina: A Story Map. North Carolina Department of Environmental Quality. https://ncdenr.maps.arcgis.com/apps/MapSeries/index.html?appid=0a7ccd9394734ff6aa2434d2528ddf12 Accessed 18 Apr 2023
NCSCO (2023) North Carolina State Climate Office. https://climate.ncsu.edu/ Accessed 18 Apr 2023
O’Driscoll MA, Humphrey CP, Deal NE, Lindbo DL, Zarate-Bermudez MA (2014) Meteorological Influences on Nitrogen Dynamics of a Coastal Onsite Wastewater Treatment System. J Environ Qual 43:1873–1885. https://doi.org/10.2134/jeq2014.05.0227
O’Driscoll M, DeWalle D, Humphrey C, Iverson G (2019) Groundwater seeps: portholes to Evaluate Groundwater’s influence on Stream Water Quality. J Contemp Water Res Ed 166:57–78. https://doi.org/10.1111/j.1936-704X.2019.03302.x
Oldfield LE, Roy JW, Robinson CE (2020) Investigating the use of the artificial sweetener acesulfame to evaluate septic system inputs and their nutrient loads to streams at the watershed scale. J Hydrol 587:124918. https://doi.org/10.1016/j.jhydrol.2020.124918
Oliver S, Corburn J, Ribeiro H (2018) Challenges regarding Water Quality of Eutrophic reservoirs in Urban landscapes: a map** literature review. Int J Env Pub He 16:40. https://doi.org/10.3390/ijerph16010040
Pérez-Ruzafa A, Campillo S, Fernández-Palacios JM, García-Lacunza A, García-Oliva M, Ibañez H, Navarro-Martínez PC, Pérez-Marcos M, Pérez-Ruzafa IM, Quispe-Becerra JI, Sala-Mirete A, Sánchez O, Marcos C (2019) Long-term dynamic in nutrients, Chlorophyll a, and Water Quality parameters in a Coastal lagoon during a process of Eutrophication for decades, a Sudden Break and a relatively Rapid Recovery. Front Mar Sci 6:1–23. https://doi.org/10.3389/fmars.2019.00026
Pfannerstill M, Kühling I, Hugenschmidt C, Trepel M, Fohrer N (2016) Reactive ditches: a simple approach to implement denitrifying wood chip bioreactors to reduce nitrate exports into aquatic ecosystems? Environ Earth Sci 75:1. https://doi.org/10.1007/s12665-016-5856-2
Posit Team (2022) RStudio: Integrated Development Environment for R, Posit Software, PBC: Boston, MA. URL: http://www.posit.co/
Pouye A, Cissé Faye S, Diédhiou M, Gaye CB, Taylor RG (2023) Nitrate contamination of urban groundwater and heavy rainfall: observations from Dakar, Senegal. Vadose Zone J 22:e20239. https://doi.org/10.1002/vzj2.20239
Qi J, Li S, Benoy G, **ng Z, Gao L, Meng F (2022) Impacts of sampling frequency on the estimation accuracy of exceedance for suspended solids and nitrates in streams in small to medium-sized watersheds. J Hydrol X 17:100139. https://doi.org/10.1016/j.hydroa.2022.100139
Raju A, Singh A (2017) Assessment of Groundwater Quality and Map** Human Health risk in Central Ganga Alluvial Plain, Northern India. Environ Process 4:375–397. https://doi.org/10.1007/s40710-017-0232-0
Rakhimbekova S, O’Carroll DM, Oldfield LE, Ptacek CJ, Robinson CE (2021) Spatiotemporal controls on septic system derived nutrients in a nearshore aquifer and their discharge to a large lake. Sci Total Environ 752:141262. https://doi.org/10.1016/j.scitotenv.2020.141262
Robertson WD, Merkley LC (2009) In-Stream Bioreactor for Agricultural Nitrate Treatment. J Environ Qual 38:230–237. https://doi.org/10.2134/jeq2008.0100
Robertson WD, Vogan JL, Lombardo PS (2008) Nitrate removal rates in a 15-Year-old permeable reactive barrier treating septic system nitrate. Ground Water Monit R 28:65–72. https://doi.org/10.1111/j.1745-6592.2008.00205.x
Robertson WD, Van Stempvoort DR, Schiff SL (2021) Nitrogen Attenuation in Septic System Plumes. Ground Water 59:369–380. https://doi.org/10.1111/gwat.13065
Sarris TS, Burbery LF (2018) Stochastic multi-objective performance optimization of an in-stream woodchip denitrifying bioreactor. Ecol Engi 124:38–50. https://doi.org/10.1016/j.ecoleng.2018.09.006
Silva SR, Ging PB, Lee RW, Ebbert JC, Tesoriero AJ, Inkpen EL (2002) Forensic applications of Nitrogen and Oxygen isotopes in tracing Nitrate sources in Urban environments. Environ Forensics 3:125–130. https://doi.org/10.1006/enfo.2002.0086
Smith RL, Böhlke JK, Song B, Tobias CR (2015) Role of anaerobic ammonium oxidation (Anammox) in Nitrogen removal from a Freshwater Aquifer. Environ Sci Technol 49:12169–12177. https://doi.org/10.1021/acs.est.5b02488
Sparacino-Watkins C, Stolz JF, Basu P (2014) Nitrate and periplasmic nitrate reductases. Chem Soc Rev 43:676–676. https://doi.org/10.1039/c3cs60249d
US EPA (2000b) Ambient Water Quality Criteria Recommendations: Lakes and Reservoirs in Ecoregion IX. Office of Water https://www.epa.gov/sites/default/files/documents/lakes9.pdf Accessed on 27 April 2023
US EPA (2000a) Ambient Water Quality Criteria Recommendations: Rivers and Streams in Ecoregion IX. Office of Water https://www.epa.gov/sites/default/files/documents/rivers9.pdf Accessed on 27 April 2023
USDA (2023) Web Soil Survey. United States Department of Agriculture. https://websoilsurvey.nrcs.usda.gov/app/ Accessed 18 Apr 2023
USGS (2023) StreamStats Web Application. United States Geological Survey. https://streamstats.usgs.gov/ss/ Accessed 18 Apr 2023
Wang J, Fu Z, Qiao H, Liu F (2019) Assessment of eutrophication and water quality in the estuarine area of Lake Wuli, Lake Taihu, China. Sci Total Environ 650:1392–1402. https://doi.org/10.1016/j.scitotenv.2018.09.137
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-, New York. https://ggplot2.tidyverse.org
Wickham H, Bryan J (2022) readxl: Read Excel Files, (Version: 1.4.1). https://readxl.tidyverse.org, https://github.com/tidyverse/readxl
Wilke C (2020) cowplot: Streamlined Plot Theme and Plot Annotations for ‘ggplot2’, (Version: 1.1.1). Austin, TX, USA
Xu S, Chen M, Feng T, Zhan L, Zhou L, Yu G (2021) Use ggbreak to effectively utilize plotting space to deal with large datasets and outliers. Front Genet 12:774846. https://doi.org/10.3389/fgene.2021.774846
Zhang M, Chadwick MA (2022) Influences of Elevated Nutrients and Water temperature from Wastewater Effluent on River Ecosystem Metabolism. Environ Process 9:43. https://doi.org/10.1007/s40710-022-00597-5
Zhi W, Ji G (2014) Quantitative response relationships between nitrogen transformation rates and nitrogen functional genes in a tidal flow constructed wetland under C/N ratio constraints. Water Res 64:32–41. https://doi.org/10.1016/j.watres.2014.06.035
Acknowledgements
The authors enthusiastically acknowledge the North Carolina Department of Environmental Quality 319 Non-Point Source Program (Contract 6201) for sponsoring this research. The authors also acknowledge the 6 homeowners who participated in the current study. The authors extend their gratitude to Caitlin Skibiel, Laura McKenna, Beau Benfield, Suelen Tullio, John Haywood, the Environmental Research Laboratory student staff, and Patrick Eaton and other Durham County Health Department staff for assisting with project development, field sampling, laboratory analysis, and/or other contributions in support of this research.
Funding
This study was funded by the North Carolina Department of Environmental Quality 319 Non-Point Source Program (Contract 6201). The funding source did not contribute to study design, data collection, analysis and interpretation of data, report writing, nor decision to submit this manuscript for publication.
Open access funding provided by the Carolinas Consortium.
Author information
Authors and Affiliations
Contributions
GI, CPHJ, and MO were responsible for conceptualization of project. GI and CPHJ developed methodologies for sampling and data analysis. EB supervised the design of the in-stream bioreactor with assistance from GI and CPHJ. GI, CPHJ, CS, and WJU contributed towards acquisition of data. GI led efforts to organize, analyze, and interpret data. GI, CPHJ, and MO were responsible for data validation. CPHJ and MO served as supervisors on the project. CPHJ led efforts to acquire and administer research funds. GI drafted the original manuscript and all authors reviewed and assisted with editing the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Human Participants, Their Data or Biological Material
The current study did not include any human participants or use any of their data or biological material nor did it use animal subjects, their data or biological material. Thus, this study is not subject to an ethics review committee and does not require any informed consent.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iverson, G., Humphrey, C.P., O’Driscoll, M. et al. Quantifying Nitrogen Treatment by an In-stream Bioreactor in a Watershed Served by Septic Systems. Environ. Process. 11, 22 (2024). https://doi.org/10.1007/s40710-024-00701-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40710-024-00701-x