Abstract
The quest for medications to reduce intra-pancreatic fat deposition is now quarter a century old. While no specific medication has been approved for the treatment of fatty change of the pancreas, drug repurposing shows promise in reducing the burden of the most common disorder of the pancreas. This leading article outlines the 12 classes of medications that have been investigated to date with a view to reducing intra-pancreatic fat deposition. Information is presented hierarchically—from preclinical studies to retrospective findings in humans to prospective interventional studies to randomised controlled trials. This lays the grounds for shepherding the most propitious drugs into medical practice through well-designed basic science studies and adequately powered randomised controlled trials.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The PANDORA hypothesis identifies fatty change of the pancreas as the shared driving force for all non-heritable diseases of the pancreas. |
Newer glucose-lowering medications (glucagon-like peptide-1 receptor agonists, dipeptidyl peptidase-4 inhibitors, sodium-glucose cotransporter-2 inhibitors) have the potential to reduce intra-pancreatic fat deposition. |
Other possible classes of medications for fatty change of the pancreas are statins and angiotensin II receptor blockers. |
1 Introduction
Approximately one-fifth of world’s population has fatty change of the pancreas—disorder characterised by excess intra-pancreatic fat deposition (IPFD) scattered throughout the pancreas, making it the most common pathology of the pancreas [1, 2]. Fatty change of the pancreas has long been thought to be associated with type 2 diabetes, the most common disease involving the endocrine pancreas [3,4,5,6]. In addition, the role of fatty change of the pancreas in pancreatitis and pancreatic cancer—the most common diseases of the exocrine pancreas—has recently become appreciated [7,8,9,10,11]. Given that the endocrine and exocrine portions of the pancreas are intimately intertwined and taking into account that fatty change of the pancreas is more common than diabetes mellitus, pancreatitis and pancreatic cancer altogether, IPFD is positioned for powerful influence on both portions of the organ.
The PANcreatic Diseases Originating from intRa-pancreatic fAt (PANDORA) hypothesis takes the multidisciplinary field of Pancreatology to the next level by identifying fatty change of the pancreas as the fundamental target for modifying the natural course of diabetes mellitus, pancreatitis and pancreatic cancer [12]. PANDORA ushers in a new era of unprecedented opportunity to improve clinical outcomes in all non-heritable diseases of the pancreas, beyond the bounds of Medicine at the end of the first quarter of the 21st century. One treatment implication of the integrated PANDORA model is that interventions effectively and timely reducing IPFD will have the potential to become the mainstay of prevention (or interception) for common diseases of not only the endocrine pancreas but also the exocrine pancreas. This is important as, while treatments targeting the pathogenesis of type 2 diabetes are galore, there are currently no interventions for intercepting the natural course of pancreatitis or sporadic pancreatic cancer. Long-term dietary changes and bariatric surgery are the types of interventions that have received most attention in regard to fatty change of the pancreas to date [1, 13,14,15]. Clinical investigations of these interventions have generally demonstrated that IPFD is labile and its amenability to treatment is often a function of duration of the study period [1, 12]. For example, a very low-energy diet administered for 6 months reduced IPFD by 42% [16]. Similarly, a reduction in IPFD by around 40% from before bariatric surgery to 6 months after surgery was demonstrated [17,18,19,20,21]. At the same time, a very low-energy diet administered for 8 weeks reduced IPFD only by 14% [22]. Comparable reduction in IPFD by 18% was observed in 8 weeks following bariatric surgery [23].
However, as the groups of people that can benefit from bariatric surgery or long-term dietary changes are quite small, only pharmacological interventions could potentially offer the benefits of reducing IPFD to the largest population [24]. It is fitting to note that a comprehensive landscape description of medications for fatty change of the pancreas is lacking in literature. Therefore, this leading article aims to map both preclinical and clinical evidence on the usefulness of various pharmacological interventions in reducing IPFD. In vivo animal studies, clinical cohort studies and randomised controlled trials were considered, whereas in vitro studies and case reports were not covered in this review.
2 Glucose-Lowering Medications
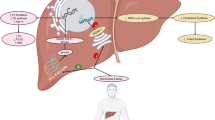
A total of six classes of glucose-lowering medications have been investigated in regard to their potential to reduce IPFD: biguanides, sulfonylureas, thiazolidinediones, glucagon-like peptide-1 (GLP-1) receptor agonists, dipeptidyl peptidase-4 (DPP-4) inhibitors and sodium-glucose cotransporter-2 (SGLT-2) inhibitors (Fig. 1). Insulin promotes the storage of excess glucose as fat and therefore cannot be expected to benefit individuals with fatty change of the pancreas [25]. Other currently available classes of glucose-lowering medications—α-glucosidase inhibitors, amylin analogues, D2 receptor agonists, meglitinides, bile acid sequestrants and dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor co-agonists—are yet to be investigated in the therapeutic context of IPFD.
Types of studies that investigated changes in intra-pancreatic fat deposition following the use of medications. ‘Preclinical’ refers to in vivo animal studies. ‘Non-RCT’ refers to retrospective or prospective clinical studies (other than randomised controlled trials). The green tick-box indicates availability of a given type of study in the literature. DPP-4 dipeptidyl peptidase-4, GLP-1 glucagon-like peptide-1, RCT randomised controlled trial, SGLT-2 sodium-glucose cotransporter-2
2.1 Biguanides
One preclinical study investigated changes in IPFD following the administration of biguanide in a high-fat diet (60% energy from fat for 10 weeks prior to group allocation) mouse model [26]. On histology, IPFD in the untreated group was 12.4%, whereas IPFD in the group receiving 310 mg/kg/day of metformin for 6 weeks was 3.8% post-intervention (p < 0.05). IPFD in the control group receiving standard chow was 1.5%. The absolute weight of the pancreas was 0.36 g in the untreated group, 0.27 g after administration of metformin (p < 0.05, when compared with the untreated group) and 0.31 g in the control group [26].
One clinical study investigated changes in IPFD following the administration of biguanide [27]. It was a prospective investigation of 17 drug-naïve individuals with T2DM diagnosed within a month prior to enrolment. The study found no significant change in IPFD (determined with the use of computed tomography) following 16 weeks of metformin use [27].
2.2 Sulfonylureas
One retrospective clinical study investigated changes in IPFD (determined with the use of computed tomography) following the administration of sulfonylureas [28]. It identified 29 individuals with T2DM on sulfonylureas in Japan over an 11-year study period, with individuals receiving GLP-1 receptor agonists and SGLT-2 inhibitors being excluded (though other glucose-lowering medications, including insulin, were allowed). The study found no significant difference in IPFD before and after administration of glimepiride [28].
2.3 Thiazolidinediones
Two preclinical studies investigated changes in IPFD following the administration of thiazolidinediones (Table 1). A study from the USA used a genetically obese (loss-of-function mutation in the leptin receptor) rat model [29]. On histology, IPFD (specifically, lipid droplets in the islets of Langerhans) was significantly lower in the intervention group receiving 200 mg/kg/day of troglitazone for 7 weeks in comparison with the untreated group (p < 0.005). The significant reduction in IPFD after administration of troglitazone was accompanied by a significant reduction in fasting plasma glucose, triglycerides and non-esterified fatty acids [29]. As a side note, while by design the loss-of-function mutation in the leptin receptor in this study precluded the action of leptin on preventing lipid deposition in the pancreas, the same research group showed that activation of AMP-activated protein kinase (a key enzyme involved in lipid synthesis and metabolism) bypasses the leptin receptor defect in the used rat model and reduces lipid droplets in the islets of Langerhans [30]. A study from Brazil used a high-fat/high-carbohydrate diet (42% energy from fat and 44% energy from carbohydrate for 6 weeks prior to group allocation) mouse model [31]. On histology, IPFD (especially lipid droplets in acinar cells) was significantly higher in the group receiving 9.4 mg/kg/day of rosiglitazone for 5 weeks in comparison with both the high-fat/high-carbohydrate chow alone group and the control group receiving standard chow (p < 0.05) [31].
One clinical study investigated changes in IPFD following the administration of thiazolidinediones [32]. It was a randomised placebo-controlled trial of pioglitazone in healthy individuals. The preliminary analysis of 19 individuals showed a significant reduction in IPFD (determined with the use of magnetic resonance spectroscopy at 1.5 T) following 26 weeks of pioglitazone versus placebo use [32]. Upon the study completion in 2013, a total of 42 individuals were enrolled but the results of this trial have never been published in a peer-reviewed journal [33].
2.4 GLP-1 Receptor Agonists
One preclinical study investigated changes in IPFD following the administration of GLP-1 receptor agonist in a high-fat diet (60% energy from fat for 12 weeks prior to group allocation) mouse model [34]. On histology, the extent of IPFD was significantly lower in the intervention group receiving 0.6 mg/kg/day of liraglutide for 4 weeks in comparison with the group receiving high-fat chow alone (p = 0.001) and was similar to the control group receiving standard chow. The significant reduction in IPFD after the administration of liraglutide was accompanied by ameliorated endoplasmic reticulum stress as evidenced by decreased phosphorylation of eIF2α as well as reduced protein levels of PERK, CHOP and GRP78 in the pancreas. It is fitting to mention at this point that the extent of fibrosis in the pancreas and relative weight of the pancreas (i.e., weight of the pancreas divided by body weight) did not differ significantly between the groups [34].
Five clinical studies (four randomised controlled trials and one retrospective cohort study) investigated changes in IPFD following the administration of GLP-1 receptor agonists in individuals with T2DM [28, 35,36,37,38]. In addition to GLP-1 receptor agonists, these individuals received other glucose-lowering medications in all the five studies (including insulin in three studies [35, 36, 38]). The retrospective study identified 13 individuals with T2DM on liraglutide in Japan over an 11-year study period and found that the change in IPFD (determined with the use of computed tomography) before and after administration of liraglutide just missed the conventional level of statistical significance (p = 0.05) [28]. All the four randomised controlled trials used a magnetic resonance system (at either 1.5T or 3T) to determine IPFD and the sample size of these studies ranged from 36 to 64 individuals [35,36,37,38]. A randomised controlled trial from India found an absolute reduction of 2.1% (a relative reduction of 23%) in IPFD following 24 weeks of dulaglutide use [35]. A randomised controlled trial from France showed an absolute reduction of 5.9% (a relative reduction of 22%) in IPFD following 26 weeks of exenatide use [36]. Both trials used standard care in the control groups, in comparison with IPFD changes in which the above-mentioned reductions in IPFD in the intervention groups were not statistically significant [35, 36]. Two other randomised trials were placebo-controlled, used liraglutide in the intervention groups, and came from the Netherlands and the USA [37, 38]. The Dutch study found an absolute reduction of 2.4% (a relative reduction of 12%) in IPFD following 12 weeks of GLP-1 receptor agonist use [37]. The American study found an absolute reduction of 1.3% (a relative reduction of 10%) in IPFD following 26 weeks of GLP-1 receptor agonist use [38]. These reductions in IPFD were not statistically significant in comparison with IPFD changes in the placebo groups [37, 38]. Magnetic resonance imaging revealed no signs of acute pancreatitis following 12 weeks of liraglutide use in the Dutch trial [37]. Although the other clinical studies mentioned above did not report on the occurrence of acute pancreatitis, there were no signs of this disease on magnetic resonance imaging in a study from Denmark that used liraglutide for 6 weeks (change in IPFD was not investigated) [39].
2.5 DPP-4 Inhibitors
Two preclinical studies investigated changes in IPFD following the administration of DPP-4 inhibitor [26, 40]. A study from Brazil used a high-fat diet (60% energy from fat for 10 weeks prior to group allocation) mouse model [26]. On histology, IPFD in the untreated group was 12.4%, whereas IPFD in the group receiving 1.1 g/kg/day of sitagliptin for 6 weeks was 1.4% post-intervention (p < 0.05). The latter was similar (1.5%) to IPFD in the control group receiving standard chow. The absolute weight of the pancreas was 0.36 g in the untreated group, 0.29 g after administration of sitagliptin (p < 0.05, when compared with the untreated group), and 0.31 g in the control group [26]. The significant reduction in IPFD after administration of sitagliptin was accompanied by a significant reduction in fasting plasma glucose (from 8.5 mmol/L to 7.0 mmol/L) as well as a 30% improvement in glucose tolerance assessed by oral glucose tolerance test, highlighting the role of incretin effect after ingestion [26, 41]. The study was unique for also employing transmission electron microscopy, which showed that mice in the untreated group were characterised by abundance of lipid droplets and paucity of mitochondria in acinar cells as well as by presence of activated pancreatic stellate cells. The cell organisation of mice after administration of sitagliptin resembled that of control group [41]. A study from India used a high-fat diet (65% energy from fat for 2 weeks prior to group allocation) mouse model [40]. Fluorescence photomicrographs showed less IPFD (specifically, lipid droplets in the islets of Langerhans) in the group receiving 20 mg/kg/day of vildagliptin for 14 weeks than in the untreated group. Also, the TLR4-NFkB signalling pathway in the pancreas was significantly suppressed (p < 0.005) and DPP-4 expression in the pancreas was significantly downregulated (p < 0.005) after administration of the intervention, suggesting that vildagliptin may reduce local inflammation [40].
One clinical study investigated changes in IPFD (determined with the use of magnetic resonance imaging at 1.5 T) following the administration of DPP-4 inhibitor [37]. It was a randomised placebo-controlled trial of individuals with T2DM, with sitagliptin being add-on to standard care (biguanides and/or sulfonylureas). The study showed a significant reduced IPFD following 12 weeks of sitagliptin versus placebo use (p = 0.04). Specifically, an absolute reduction in IPFD was 4.2% (and a relative reduction of 16%) in the intervention group. Notably, none of the 19 individuals in the intervention group developed signs of acute pancreatitis on magnetic resonance imaging [37].
2.6 SGLT-2 Inhibitors
Five (four prospective and one retrospective) clinical studies investigated changes in IPFD following the administration of SGLT-2 inhibitors in individuals with T2DM or prediabetes [42,43,44,45,46]. In addition to SGLT-2 inhibitors, individuals with T2DM received other glucose-lowering medications (including insulin in two studies [42, 43]). The retrospective study identified 22 individuals in Japan over a 6-year study period and found no significant difference in IPFD (determined with the use of computed tomography) before and after administration of SGLT-2 inhibitors [42]. All the four prospective studies used a magnetic resonance system at 3T to determine IPFD and the sample size of these studies ranged from 30 to 78 individuals [43,44,45,46]. Three out of four prospective studies were randomised controlled trials, including two placebo-controlled trials [43, 44]. A study from India found an absolute reduction in IPFD by 1.5% (a relative reduction of 20%) following 17 weeks of dapagliflozin use [45], and a study from China showed an absolute reduction in IPFD by 1.2% (a relative reduction of 20%) following 24 weeks of dapagliflozin use [46]. The IPFD change was independent of weight change in the Chinese study [46] and dependent of weight change in the Indian study [45]. The two placebo-controlled trials used empagliflozin as the study intervention and they came from Germany and France [43, 44]. The German study found an absolute reduction of 0.9% (a relative reduction of 13%) in IPFD following 8 weeks of SGLT-2 inhibitor use [44]. The French study found an absolute reduction in IPFD by 2.5% (a relative reduction of 12%) following 12 weeks of SGLT-2 inhibitor use [43]. These reductions in IPFD were not statistically significant in comparison with IPFD changes in the placebo groups (Table 2).
3 Lipid-Regulating Medications
To date, out of all lipid-regulating medications, only statins, fibrates, and pancreatic lipase inhibitors have been investigated in the fatty change of the pancreas context (Fig. 1). Other currently available classes of lipid-regulating medications—nicotinic acid, cholesterol absorption inhibitors, bile acid sequestrants and proprotein convertase subtilisin/kexin type 9 inhibitors—are yet to be investigated in regard to their effect on IPFD.
3.1 Statins
Two preclinical studies investigated changes in IPFD following the administration of statins [47, 48]. A study from China used a high-fat diet (for 20 weeks prior to group allocation; fat content was not specified) mouse model [47]. On histology, triglycerides (but not cholesterol) in the pancreas were significantly lower in the intervention group receiving 30 mg/kg/day of atorvastatin for more than 8 weeks in comparison with the untreated group (p < 0.001). This significant reduction after administration of atorvastatin was accompanied by reduced endoplasmic reticulum stress in the pancreas as evidenced by reduced phosphorylation of eIF2α as well as reduced protein levels of ATF4 and CHOP. The absolute weight of the pancreas was 0.28 g after administration of atorvastatin and 0.23 g in the untreated group (p < 0.05). The greater weight of the pancreas in the intervention group might have been due to increased pancreas cell proliferation following administration of atorvastatin as the study showed a significantly upregulated pancreas duodenum homeobox-1—a critical molecule for pancreas cell proliferation—gene expression in the intervention group [47]. On a related note, decrease in apoptotic cells in the pancreas, as well as activated pancreatic stellate cells (IPFD was not investigated), following the administration of pravastatin into the duodenum was found in a rat model of chronic pancreatitis [49]. The second study of changes in IPFD before and after the use of statins came from Indonesia and employed a high-fat/high-carbohydrate diet (for 4 weeks prior to group allocation; fat and carbohydrate content was not specified) rat model [48]. On histology, the median percentage of lipid-containing cells in the pancreas was 0.27% in the intervention group receiving 10 mg/kg/day of simvastatin for 4 weeks as compared with 0.70% in the untreated group (p = 0.02) [48].
3.2 Fibrates
One preclinical study investigated changes in IPFD following the administration of fibrates in a high-fat/high-carbohydrate diet (42% energy from fat, 44% energy from carbohydrate for 6 weeks prior to group allocation) mouse model [31]. On histology, IPFD did not differ significantly between the group receiving 382 mg/kg/day of bezafibrate for 5 weeks and the high-fat/high-carbohydrate chow alone group. Similarly, IPFD did not differ significantly between the group receiving 163 mg/kg/day of fenofibrate for 5 weeks and the high-fat/high-carbohydrate chow alone group. At the same time, absolute weight of the pancreas was reduced by 41% in the bezafibrate group (p < 0.001) and 24% in the fenofibrate group (p < 0.05) in comparison with the high-fat/high-carbohydrate chow alone group [31].
3.3 Pancreatic Lipase Inhibitors
One preclinical study investigated changes in IPFD following the administration of pancreatic lipase inhibitor in a genetically obese (loss-of-function mutation in the leptin receptor) mice model [50]. A unique aspect of this study was the induction of acute pancreatitis (with the use of cerulein). On histology, IPFD (specifically, necrotic fat) was significantly lower in the intervention group receiving 50 mg/kg/day of orlistat for 2 days in comparison with the untreated group (p < 0.05). The significant reduction in IPFD after administration of orlistat was accompanied by reduced CD68 positivity (a histochemical marker of macrophages) in the intervention group [50].
4 Other Medications
Miscellaneous medications that have been studied in regard to reduction in IPFD include angiotensin II receptor blockers, somatostatin receptor agonists and antioxidants (Fig. 1). Corticosteroids are well-known to stimulate lipogenesis and therefore cannot be expected to benefit individuals with fatty change of the pancreas [51].
4.1 Angiotensin II Receptor Blockers
One preclinical study investigated changes in IPFD following the administration of angiotensin II receptor blocker in a high-fat diet (60% energy from fat for 10 weeks prior to group allocation) mouse model [26]. On histology, IPFD in the untreated group was 12.4%, whereas IPFD in the group receiving 5.2 g/kg/day of telmisartan for 6 weeks was 3.7% post-intervention (p < 0.05). IPFD in the control group receiving standard chow was 1.5%. The absolute weight of the pancreas was 0.36 g in the untreated group, 0.29 g after administration of telmisartan (p < 0.05, when compared with the untreated group) and 0.31 mg in the control group [26]. As a side note, another angiotensin II receptor blocker (candesartan) has been recently shown in a high-fat diet mouse model to have a previously unrecognised beneficial effect of preventing palmitic acid-induced intracellular Ca2+ overload [52]—a key element in the pathogenesis of IPFD [12]. IPFD was not investigated in that study though [52]. Also, accumulating evidence suggests the existence of a local renin–angiotensin system in the pancreas [53].
4.2 Somatostatin Receptor Agonists
One preclinical study investigated changes in IPFD following the administration of somatostatin receptor agonist in a high-fat diet (60% energy from fat for 24 weeks prior to group allocation) rat model [54]. On histology, IPFD (specifically, triglycerides) in the untreated group was 32 mg/g pancreas tissue, whereas IPFD in the group receiving 80 mcg/kg/day of octreotide for 8 days was 24 mg/g pancreas tissue post-intervention (p < 0.05). IPFD in the control group receiving standard chow was 15 mg/g pancreas tissue. Similar findings were observed in regard to free fatty acids in the pancreas. The absolute weight of the pancreas did not differ between the intervention group and the untreated group but was significantly lower in both groups than the absolute weight of the pancreas in the control group receiving standard chow [54]. This might have been due to the used high-fat diet protocol inducing chronic pancreatitis in rats (and leading to the atrophy of the exocrine pancreas). The significant reduction in IPFD after administration of octreotide was accompanied by downregulation of α-smooth muscle actin in the intervention group, suggesting dormancy of pancreatic stellate cells [54].
One randomised controlled trial investigated changes in IPFD (as proxied by texture of the pancreas intraoperatively) following the administration of somatostatin analogue [55]. The study included 25 patients undergoing pancreatic surgery who received either a single bolus of 0.1 mg/mL octreotide or saline solution injected in the gastroduodenal artery. A significantly increased hardness of the organ (quantified with the use of durometer) was observed in the somatostatin group (p = 0.03) [55]. A harder pancreas signifies reduced IPFD [56].
4.3 Antioxidants
One preclinical study investigated changes in IPFD following the administration of antioxidant in a high-fat/high-carbohydrate diet (45% energy from fat for 18 weeks) mouse model [57]. A unique aspect of this study was the use of female mice without functioning ovaries. On histology, IPFD was significantly lower in the intervention group receiving 5% of ascorbic acid in high-fat/high-carbohydrate diet on a weight-by-weight basis for 18 weeks in comparison with the untreated group on high-fat/high-carbohydrate diet alone (p < 0.05). The absolute weight of the pancreas was 0.28 g in the untreated group, 0.18 g after administration of ascorbic acid (p < 0.05, when compared with the untreated group) and 0.18 g in the control group. Also, significant reductions in the absolute weights of heart and liver, but not the absolute weights of kidney and spleen and body weight, were observed in the intervention group [57].
5 Conclusions
From the 1999 publication on the use of a now-defunct drug [29] to the 2024 publication on the use of a blockbuster anti-obesity medication [28], the present article has chronicled the preclinical and clinical investigations of medications that can be helpful in reversing fatty change of the pancreas. There is no specific medication approved for the treatment of fatty change of the pancreas, but certain medications may be worth investigating in purposely designed future studies (Fig. 2). The landscape of these medications is quite diverse, encompassing both newer (e.g. GLP-1 receptor agonists, DPP-4 inhibitors and SGLT-2 inhibitors) and classic (e.g. statins, biguanides and angiotensin II receptor blockers) therapies. One commonality between them is that endoplasmic reticulum stress in the pancreas—the importance of which in the pathogenesis of fatty change of the pancreas was postulated in the PANDORA hypothesis [12]—may be reduced, at least in part, with the use of the above-mentioned classes of medications [34, 47]. This aligns well with the emerging notion of pharmacological modulation of endoplasmic reticulum stress in metabolic diseases [58]. The pharmacological landscape for fatty change of the pancreas is likely to be expanded in the near future as exemplified by the Food and Drug Administration approval of tirzepatide (a dual glucose-dependent insulinotropic polypeptide receptor and GLP-1 receptor co-agonist) for chronic weight management just as the finishing touches on the present article were being put [59, 60]. New opportunities to broaden the indications for approved weight loss medications are definitely on the horizon.
Effects of various classes of medications on IPFD. Only major classes of medications are depicted. Classes of medications that have never been demonstrated to affect IPFD are not presented. DPP-4 dipeptidyl peptidase-4, GLP-1 glucagon-like peptide-1, IPFD intra-pancreatic fat deposition, SGLT-2 sodium-glucose cotransporter-2
References
Petrov MS, Taylor R. Intra-pancreatic fat deposition: bringing hidden fat to the fore. Nat Rev Gastroenterol Hepatol. 2022;19:153–68.
Wong VW, Wong GL, Yeung DK, et al. Fatty pancreas, insulin resistance, and β-cell function: a population study using fat-water magnetic resonance imaging. Am J Gastroenterol. 2014;109:589–97.
Lee Y, Hirose H, Ohneda M, et al. Beta-cell lipotoxicity in the pathogenesis of non-insulin-dependent diabetes mellitus of obese rats: impairment in adipocyte-beta-cell relationships. Proc Natl Acad Sci USA. 1994;91:10878–82.
Singh RG, Yoon HD, Wu LM, et al. Ectopic fat accumulation in the pancreas and its clinical relevance: a systematic review, meta-analysis, and meta-regression. Metabolism. 2017;69:1–13.
Taylor R, Al-Mrabeh A, Sattar N. Understanding the mechanisms of reversal of type 2 diabetes. Lancet Diabetes Endocrinol. 2019;7:726–36.
Al-Mrabeh A, Zhyzhneuskaya SV, Peters C, et al. Hepatic lipoprotein export and remission of human type 2 diabetes after weight loss. Cell Metab. 2020;31:233–49.
Ko J, Al-Ani Z, Long K, et al. Intra-pancreatic, liver, and skeletal muscle fat depositions in first attack of acute pancreatitis versus health. Am J Gastroenterol. 2022;117:1693–701.
Al-Ani Z, Ko J, Petrov MS. Intra-pancreatic fat deposition across the pancreatitis spectrum and the influence of gut hormones. Dig Liver Dis. 2023;55:1081–90.
Ko J, Skudder-Hill L, Priya S, et al. Associations between intra-pancreatic fat deposition, pancreas size, and pancreatic enzymes in health and after an attack of acute pancreatitis. Obes Facts. 2022;15:70–82.
Sreedhar UL, DeSouza SV, Park B, et al. A systematic review of intra-pancreatic fat deposition and pancreatic carcinogenesis. J Gastrointest Surg. 2020;24:2560–9.
Petrov MS. Fateful fat: intra-pancreatic lipids cause pancreatic cancer. Cell Rep Med. 2024;5: 101428.
Petrov MS. Fatty change of the pancreas: the Pandora’s box of pancreatology. Lancet Gastroenterol Hepatol. 2023;8:671–82.
Della Pepa G, Salamone D, Testa R, et al. Intrapancreatic fat deposition and nutritional treatment: the role of various dietary approaches. Nutr Rev. 2023. https://doi.org/10.1093/nutrit/nuad159. (Epub ahead of print).
Ko J, Skudder-Hill L, Tarrant C, et al. Intra-pancreatic fat deposition as a modifier of the relationship between habitual dietary fat intake and insulin resistance. Clin Nutr. 2021;40:4730–7.
Ko J, Kimita W, Skudder-Hill L, et al. Dietary carbohydrate intake and insulin traits in individuals after acute pancreatitis: effect modification by intra-pancreatic fat deposition. Pancreatology. 2021;21:353–62.
Rossi AP, Fantin F, Zamboni GA, et al. Effect of moderate weight loss on hepatic, pancreatic and visceral lipids in obese subjects. Nutr Diabetes. 2012;2: e32.
Honka H, Koffert J, Hannukainen JC, et al. The effects of bariatric surgery on pancreatic lipid metabolism and blood flow. J Clin Endocrinol Metab. 2015;100:2015–23.
Umemura A, Sasaki A, Nitta H, et al. Pancreas volume reduction and metabolic effects in Japanese patients with severe obesity following laparoscopic sleeve gastrectomy. Endocr J. 2017;64:487–98.
Lautenbach A, Wernecke M, Riedel N, et al. Adaptive changes in pancreas post Roux-en-Y gastric bypass induced weight loss. Diabetes Metab Res Rev. 2018;34: e3025.
Gaborit B, Abdesselam I, Kober F, et al. Ectopic fat storage in the pancreas using 1H-MRS: importance of diabetic status and modulation with bariatric surgery-induced weight loss. Int J Obes. 2015;39:480–7.
Hui SC, Wong SK, Ai Q, Yeung DK, Ng EK, Chu WC. Observed changes in brown, white, hepatic and pancreatic fat after bariatric surgery: evaluation with MRI. Eur Radiol. 2019;29:849–56.
Steven S, Hollingsworth KG, Al-Mrabeh A, et al. Very low-calorie diet and 6 months of weight stability in type 2 diabetes: pathophysiological changes in responders and nonresponders. Diabetes Care. 2016;39:808–15.
Steven S, Hollingsworth KG, Small PK, et al. Weight loss decreases excess pancreatic triacylglycerol specifically in type 2 diabetes. Diabetes Care. 2016;39:158–65.
Goodarzi MO, Petrov MS. Diabetes of the exocrine pancreas: implications for pharmacological management. Drugs. 2023;83:1077–90.
Lewis GF, Carpentier A, Adeli K, et al. Disordered fat storage and mobilization in the pathogenesis of insulin resistance and type 2 diabetes. Endocr Rev. 2002;23:201–29.
Souza-Mello V, Gregório BM, Cardoso-de-Lemos FS, et al. Comparative effects of telmisartan, sitagliptin and metformin alone or in combination on obesity, insulin resistance, and liver and pancreas remodelling in C57BL/6 mice fed on a very high-fat diet. Clin Sci (Lond). 2010;119:239–50.
Zsóri G, Illés D, Ivány E, et al. In new-onset diabetes mellitus, metformin reduces fat accumulation in the liver, but not in the pancreas or pericardium. Metab Syndr Relat Disord. 2019;17:289–95.
Kuriyama T, Ishibashi C, Kozawa J, et al. Effects of liraglutide on intrapancreatic fat deposition in patients with type 2 diabetes. Clin Nutr ESPEN. 2024;59:208–13.
Higa M, Zhou YT, Ravazzola M, et al. Troglitazone prevents mitochondrial alterations, beta cell destruction, and diabetes in obese prediabetic rats. Proc Natl Acad Sci USA. 1999;96:11513–8.
Yu X, McCorkle S, Wang M, et al. Leptinomimetic effects of the AMP kinase activator AICAR in leptin-resistant rats: prevention of diabetes and ectopic lipid deposition. Diabetologia. 2004;47:2012–21.
Fernandes-Santos C, Evangelista Carneiro R, de Souza ML, et al. Rosiglitazone aggravates nonalcoholic fatty pancreatic disease in C57BL/6 mice fed high-fat and high-sucrose diet. Pancreas. 2009;38:e80–6.
Lingvay I, Poduri M, Szczepaniak E, et al. Pioglitazone: effect on pancreatic steatosis and beta cell function. Diabetologia. 2012;55(Suppl 1):S327.
Lingvay I. Pioglitazone on pancreatic steatosis and bone health. https://clinicaltrials.gov. Accessed 18 Jan 2024.
Fang T, Huang S, Chen Y, et al. Glucagon like peptide-1 receptor agonists alter pancreatic and hepatic histology and regulation of endoplasmic reticulum stress in high-fat diet mouse model. Exp Clin Endocrinol Diabetes. 2021;129:625–33.
Kuchay MS, Krishan S, Mishra SK, et al. Effect of dulaglutide on liver fat in patients with type 2 diabetes and NAFLD: randomised controlled trial (D-LIFT trial). Diabetologia. 2020;63:2434–45.
Dutour A, Abdesselam I, Ancel P, et al. Exenatide decreases liver fat content and epicardial adipose tissue in patients with obesity and type 2 diabetes: a prospective randomized clinical trial using magnetic resonance imaging and spectroscopy. Diabetes Obes Metab. 2016;18:882–91.
Smits MM, Tonneijck L, Muskiet MH, et al. Pancreatic effects of liraglutide or sitagliptin in overweight patients with type 2 diabetes: a 12-week randomized, placebo-controlled trial. Diabetes Care. 2017;40:301–8.
Vanderheiden A, Harrison LB, Warshauer JT, et al. Mechanisms of action of liraglutide in patients with type 2 diabetes treated with high-dose insulin. J Clin Endocrinol Metab. 2016;101:1798–806.
Svane MS, Johannesen HH, Martinussen C, et al. No effects of a 6-week intervention with a glucagon-like peptide-1 receptor agonist on pancreatic volume and oedema in obese men without diabetes. Diabetes Obes Metab. 2020;22:1837–46.
Nag S, Mandal S, Mukherjee O, et al. Vildagliptin inhibits high fat and fetuin-A mediated DPP-4 expression, intracellular lipid accumulation and improves insulin secretory defects in pancreatic beta cells. Biochim Biophys Acta Mol Basis Dis. 2024;1870: 167047.
Souza-Mello V, Gregório BM, Relvas-Lucas B, et al. Pancreatic ultrastructural enhancement due to telmisartan plus sitagliptin treatment in diet-induced obese C57BL/6 mice. Pancreas. 2011;40:715–22.
Horii T, Kozawa J, Fujita S, et al. Amelioration of pancreatic fat accumulation in Japanese type 2 diabetes patients treated with sodium-glucose cotransporter 2 inhibitors: a retrospective study. Obes Sci Pract. 2021;7:346–52.
Gaborit B, Ancel P, Abdullah AE, et al. Effect of empagliflozin on ectopic fat stores and myocardial energetics in type 2 diabetes: the EMPACEF study. Cardiovasc Diabetol. 2021;20:57.
Hummel J, Machann J, Dannecker C, et al. Eight weeks of empagliflozin does not affect pancreatic fat content and insulin secretion in people with prediabetes. Diabetes Obes Metab. 2022;24:1661–6.
Ghosh A, Dutta K, Bhatt SP, et al. Dapagliflozin improves body fat patterning, and hepatic and pancreatic fat in patients with type 2 diabetes in North India. J Clin Endocrinol Metab. 2022;107:e2267–75.
Shi M, Zhang H, Wang W, et al. Effect of dapagliflozin on liver and pancreatic fat in patients with type 2 diabetes and non-alcoholic fatty liver disease. J Diabetes Complicat. 2023;37: 108610.
Chen ZY, Liu SN, Li CN, et al. Atorvastatin helps preserve pancreatic β cell function in obese C57BL/6 J mice and the effect is related to increased pancreas proliferation and amelioration of endoplasmic-reticulum stress. Lipids Health Dis. 2014;13:98.
Krisnamurti DGB, Farida S, Putri RC, et al. The effect of simvastatin-Acalypha indica Linn. combination on the improvement of fatty pancreas in rats induced with a high fructose and cholesterol diet. J Adv Vet Anim Res. 2022;9:346–50.
Wei L, Yamamoto M, Harada M, et al. Treatment with pravastatin attenuates progression of chronic pancreatitis in rat. Lab Investig. 2011;91:872–84.
Patel K, Trivedi RN, Durgampudi C, et al. Lipolysis of visceral adipocyte triglyceride by pancreatic lipases converts mild acute pancreatitis to severe pancreatitis independent of necrosis and inflammation. Am J Pathol. 2015;185:808–19.
Geer EB, Islam J, Buettner C. Mechanisms of glucocorticoid-induced insulin resistance: focus on adipose tissue function and lipid metabolism. Endocrinol Metab Clin N Am. 2014;43:75–102.
Lee JW, Gu HO, Jung Y, et al. Candesartan, an angiotensin-II receptor blocker, ameliorates insulin resistance and hepatosteatosis by reducing intracellular calcium overload and lipid accumulation. Exp Mol Med. 2023;55:910–25.
Leung PS. The physiology of a local renin-angiotensin system in the pancreas. J Physiol. 2007;580:31–7.
Ye T, Chen YH, Gao JH, et al. Effect of octreotide on pancreatic fibrosis in rats with high-fat diet-induced obesity. Int J Clin Exp Pathol. 2018;11:4784–94.
Belyaev O, Polle C, Herzog T, et al. Effects of intra-arterial octreotide on pancreatic texture: a randomized controlled trial. Scand J Surg. 2013;102:164–70.
Hong TH, Choi JI, Park MY, et al. Pancreatic hardness: correlation of surgeon’s palpation, durometer measurement and preoperative magnetic resonance imaging features. World J Gastroenterol. 2017;23:2044–51.
Jeon S, Lee J, Shin Y, Yoon M. Ascorbic acid reduces insulin resistance and pancreatic steatosis by regulating adipocyte hypertrophy in obese ovariectomized mice. Can J Physiol Pharmacol. 2023;101:294–303.
Marciniak SJ, Chambers JE, Ron D. Pharmacological targeting of endoplasmic reticulum stress in disease. Nat Rev Drug Discov. 2022;21:115–40.
France NL, Syed YY. Tirzepatide: a review in type 2 diabetes. Drugs. 2024. https://doi.org/10.1007/s40265-023-01992-4. (Epub ahead of print).
Sardar MB, Nadeem ZA, Babar M. Tirzepatide: a novel cardiovascular protective agent in type 2 diabetes mellitus and obesity. Curr Probl Cardiol. 2024;49: 102489.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. The research programme of M.S.P. is supported, in part, by the Royal Society of New Zealand in the form of a Rutherford Discovery Fellowship.
Conflict of interest
M.S.P. has no conflicts to declare.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Upon reasonable request to the corresponding author.
Author contributions
M.S.P. conceived and wrote the manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Petrov, M.S. The Pharmacological Landscape for Fatty Change of the Pancreas. Drugs 84, 375–384 (2024). https://doi.org/10.1007/s40265-024-02022-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-024-02022-7