Abstract
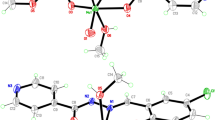
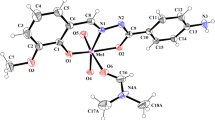
Novel dioxomolybdenum Schiff base complexes, [MoO2(Ln)(CH3OH)], were synthesized by the reaction of MoO2(acac)2 and ONO donor Schiff base ligands (H2Ln) derived by the condensation of 4-aminobenzohydrazide and substituted salicylaldehydes. The synthesized ligands and their complexes were characterized by various spectroscopic techniques like FT-IR, 1H NMR, 13C NMR, and elemental analysis (CHN). The geometries around the central metal atom in the complexes were suggested distorted octahedral. Moreover, the catalytic efficiency of the complexes was investigated by oxidizing the aryl and alkyl sulfides in the presence of tert-butyl hydroperoxide in 1,2-dichloroethane under reflux conditions. This method has numerous ascendancies, such as high yield, short reaction time, and excellent selectivity to produce corresponding sulfoxides without overoxidation to sulfones.






Similar content being viewed by others
References
J. Devi, N. Batra, R. Malhotra, Spectrochim. Acta Part A 97, 397 (2012)
M. Gupta, S. Sihag, A.K. Varshney, S. Varshney, J. Chem. 745101 (2013)
E. Yousif, A. Majeed, K. Al-Sammarrae, N. Salih, J. Salimon, B. Abdullah, Arab. J. Chem. 10, S1639 (2017)
H. Kargar, R. Behjatmanesh-Ardakani, V. Torabi, M. Kashani, Z. Chavoshpour-Natanzi, Z. Kazemi, V. Mirkhani, A. Sahraei, M.N. Tahir, M. Ashfaq, K.S. Munawar, Polyhedron 195, 114988 (2021)
H. Kargar, R. Behjatmanesh-Ardakani, V. Torabi, A. Sarvian, Z. Kazemi, Z. Chavoshpour-Natanzi, V. Mirkhani, A. Sahraei, M.N. Tahir, M. Ashfaq, Inorg. Chim. Acta 514, 120004 (2021)
R. Sudheer, M. Sithambaresan, N.R. Sajitha, E. Manoj, M.P. Kurup, Acta Crystallogr. E71, 702 (2015)
P.B. Sreeja, M.R.P. Kurup, A. Kishore, C. Jasmin, Polyhedron 23, 575 (2004)
R.S. Nair, M. Kuriakose, V. Somasundaram, V. Shenoi, M.R.P. Kurup, P. Srinivas, Life Sci. 116, 90 (2014)
E.N. Nfor, A. Husian, F. Majoumo-Mbe, I.N. Njah, O.E. Offiong, S.A. Bourne, Polyhedron 63, 207 (2013)
R. Pal, V. Kumar, A.K. Gupta, V. Beniwal, Med. Chem. Res. 23, 3327 (2014)
M. Wu, D.D. Yang, H.W. Zheng, Q.F. Liang, J.B. Li, Y. Kang, S. Li, C. Jiao, X.J. Zheng, L.P. **, Dalton Trans. 50, 1507 (2021)
R.P. Bakale, G.N. Naik, C.V. Mangannavar, I.S. Muchchandi, I.N. Shcherbakov, C. Frampton, K.B. Gudasi, Eur. J. Med. Chem. 73, 38 (2014)
B. Türkkan, B. Sarıboğa, N. Sarıboğa, Transit. Met. Chem. 36, 679 (2011)
D.M. Boghaei, S. Mohebi, J. Mol. Catal. A: Chem. 179, 41 (2002)
P.G. Cozzi, Chem. Soc. Rev. 33, 410 (2004)
K.S. Munawar, S. Ali, M.N. Tahir, N. Khalid, Q. Abbas, I.Z. Qureshi, S. Hussain, M. Ashfaq, J. Coord. Chem. 73, 2275 (2020)
R. Ramnauth, S. Al-Juaid, M. Motevalli, B.C. Parkin, A.C. Sullivan, Inorg. Chem. 43, 4072 (2004)
H. Li, J. Wu, X. Huang, G. Lu, J. Yang, X. Lu, Q. **ong, H. Zhang, ACS Nano 7, 10344 (2013)
C. Wang, M. Yu, A.F. Kyle, P. Jakubec, D.J. Dixon, R.R. Schrock, A.H. Hoveyda, Chem. Eur. J. 19, 2726 (2013)
K.C. MacLeod, P.L. Holland, Nat. Chem. 5, 559 (2013)
H. Sugimoto, S. Tatemoto, K. Toyota, K. Ashikari, M. Kubo, T. Ogura, S. Itoh, Chem. Commun. 49, 4358 (2013)
H. Sugimoto, M. Sato, L.J. Giles, K. Asano, T. Suzuki, M.L. Kirk, S. Itoh, Dalton Trans. 42, 15927 (2013)
M.E. Judmaier, C.H. Sala, F. Belaj, M. Volpe, N.C. Mösch-Zanetti, New J. Chem. 37, 2139 (2013)
M. Bagherzadeh, L. Tahsini, R. Latifi, A. Ellern, L.K. Woo, Inorg. Chim. Acta 361, 2019 (2008)
M.R. Maurya, M. Kumar, S.J.J. Titinchi, H.S. Abbo, S. Chand, Catal. Lett. 86, 97 (2003)
T.A. Alsalim, J.S. Hadi, E.A. Al-Nasir, H.S. Abbo, S.J.J. Titinchi, Catal. Lett. 136, 228 (2010)
A.P. de Azevedo Marques, E.R. Dockal, F.C. Skrobot, I.L.V. Rosa, Inorg. Chem. Commun. 10, 255 (2007)
S. Bunce, R.J. Cross, L.J. Farrugia, S. Kunchandy, L.L. Meason, K.W. Muir, M. O’Donnel, R.D. Peacock, D. Stirling, S.J. Teat, Polyhedron 17, 4179 (1998)
K.S. Munawar, S. Ali, M.N. Tahir, N. Khalid, Q. Abbas, I. Qureshi, S. Shahzadi, Russ. J. Gen. Chem. 85, 2183 (2015)
R.S. Walmsley, Z.R. Tshentu, S. Afr, J. Chem. 63, 95 (2010)
S. Mohebbi, F. Nikpour, S. Raiati, J. Mol. Catal. A: Chem. 256, 265 (2006)
M.M. Cecchini, F. De Angelis, C. Iacobucci, S. Reale, M. Crucianelli, Appl. Catal. A: Gen. 599, 117622 (2020)
M. Mancka, W. Plass, Inorg. Chem. Commun. 10, 677 (2007)
M. Salavati-Niasari, M. Bazarganipour, J. Mol. Catal. A: Chem. 278, 173 (2007)
Y.D. Li, X.K. Fu, B.W. Gong, X.C. Zou, X.B. Tu, J.X. Chen, J. Mol. Catal. A: Chem. 322, 55 (2010)
Y. Yang, Y. Zhang, S.J. Hao, Q.B. Kan, J. Colloid Interface Sci. 362, 157 (2011)
S.M. Bruno, J.A. Fernandes, L.S. Martins, I.S. Goncalves, M. Pillinger, P. Ribeiro-Claro, J. Rocha, A.A. Valente, Catal. Today 114, 263 (2006)
C.Y. Lorber, S.P. Smidt, J.A. Osborn, Eur. J. Inorg. Chem. 655 (2000)
I. Fernandez, N. Khiar, Chem. Rev. 103, 3651 (2003)
A.M. Khenkin, R. Neumann, J. Am. Chem. Soc. 124, 4198 (2002)
N. Baig, V.K. Madduluri, A.K. Sah, RSC Adv. 6, 28015 (2016)
D.H. Hua, in Advances in Carbanion Chemistry, ed. by V. Snieckus (JAI Press, London, 1992)
P. Renaud, M. Gerster, Angew. Chem. Int. Ed. 37, 2562 (1998)
Y.Z. Wang, P. Wang, C. Li, Y.Q. Su, Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 46, 927 (2016)
P. Suresh, S. Srimurugan, B. Babu, H.N. Pati, Tetrahedron Asymmetry 18, 2820 (2007)
R.D. Chakravarthy, K. Suresh, V. Ramkumar, D.K. Chand, Inorg. Chim. Acta 376, 57 (2011)
M.M. Javadi, M. Moghadam, I. Mohammadpoor-Baltork, S. Tangestaninejad, V. Mirkhani, J. Iran. Chem. Soc. 12, 477 (2015)
V. Mirkhani, S. Tangestaninejad, M. Moghadam, I. Mohammadpoor-Baltork, H. Kargar, J. Mol. Catal. A: Chem. 242, 251 (2005)
M.M. Javadi, M. Moghadam, I. Mohammadpoor-Baltork, S. Tangestaninejad, V. Mirkhani, H. Kargar, M.N. Tahir, Polyhedron 72, 19 (2014)
W. Sheng, J.-L. Shi, H. Hao, X. Li, X. Lang, J. Colloid Interface Sci. 565, 614 (2020)
H. Kargar, Transit. Met. Chem. 39, 811 (2014)
R. Kia, H. Kargar, J. Coord. Chem. 68, 1441 (2015)
A. Sahraei, H. Kargar, M. Hakimi, M.N. Tahir, J. Mol. Struct. 1149, 576 (2017)
A.A. Ardakani, H. Kargar, N. Feizi, M.N. Tahir, J. Iran. Chem. Soc. 15, 1495 (2018)
H. Kargar, V. Torabi, A. Akbari, R. Behjatmanesh-Ardakani, M.N. Tahir, J. Iran. Chem. Soc. 16, 1081 (2019)
H. Kargar, V. Torabi, A. Akbari, R. Behjatmanesh-Ardakani, A. Sahraei, M.N. Tahir, J. Mol. Struct. 1205, 127642 (2020)
H. Kargar, F. Aghaei-Meybodi, R. Behjatmanesh-Ardakani, M.R. Elahifard, V. Torabi, M. Fallah-Mehrjardi, M.N. Tahir, M. Ashfaq, K.S. Munawar, J. Mol. Struct. 1230, 129908 (2021)
H. Kargar, A.A. Ardakani, M.N. Tahir, M. Ashfaq, K.S. Munawar, J. Mol. Struct. 1229, 129842 (2021)
H. Kargar, A.A. Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, J. Iran. Chem. Soc. (2021). https://doi.org/10.1007/s13738-021-02207-x
A.K. Singh, O.P. Pandey, S.K. Sengupta, Spectrochim. Acta Part A 85, 1 (2012)
S. Durmus, A. Atahan, M. Zengin, Spectrochim. Acta Part A 84, 1 (2011)
A.H. Kianfar, M. Paliz, M. Roushani, M. Shamsipur, Spectrochim. Acta Part A 127, 422 (2014)
M. Shebl, Spectrochim. Acta Part A 70, 850 (2008)
S. Gao, X.-F. Zhang, L.-H. Huo, H. Zhao, Acta Crystallogr. E60, m1731 (2004)
A. Rana, R. Dinda, P. Sengupta, S. Ghosh, L.R. Falvello, Polyhedron 21, 1023 (2002)
V. Vrdoljak, M. Cindric, D. Milic, D. Matkovic-Calogovic, P. Novak, B. Kamenar, Polyhedron 24, 1717 (2005)
T.M. Asha, M.R.P. Kurup, J. Chem. Crystallogr. 49, 219 (2019)
F. Ruff, A. Fabian, O. Farkas, A. Kucsman, Eur. J. Org. Chem. 2102 (2009)
B. Yadollahi, Chem. Lett. 32, 1066 (2003)
R. Tan, C. Li, Z. Peng, D. Yin, D. Yin, Catal. Commun. 12, 1488 (2011)
S. Hussain, D. Talukdar, S.K. Bharadwaj, M.K. Chaudhuri, Tetrahedron Lett. 53, 6512 (2012)
M. Nikoorazm, A. Ghorbani-Choghamarani, N. Noori, Appl. Organomet. Chem. 29, 328 (2015)
M. Bagherzadeh, M. Bahjati, A. Mortazavi-Manesh, J. Organomet. Chem. 897, 200 (2019)
A. Ghorbani-Choghamarani, B. Ghasemi, Z. Safari, G. Azadi, Catal. Commun. 60, 70 (2015)
H. Taghiyar, B. Yadollahi, Sci. Total Environ. 708, 134860 (2020)
Acknowledgements
We gratefully acknowledge practical support of this study by Ardakan University and Payame Noor University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kargar, H., Fallah-Mehrjardi, M. Novel dioxomolybdenum complexes containing ONO-tridentate Schiff base ligands derived from 4-aminobenzohydrazide: synthesis, spectral characterization, and application as efficient homogeneous catalysts for selective sulfoxidation. J IRAN CHEM SOC 18, 3443–3456 (2021). https://doi.org/10.1007/s13738-021-02282-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02282-0