Abstract
Backgrounds
Given their wide availability and range of bioactive reducing metabolites, plant extracts are a promising candidate for the green production of silver nanoparticles (Ag NPs). The objective of the current investigation was to assess Ag NPs’ gastroprotective potential against ethanol-induced stomach ulcers. Rose foetida Herrm. extract was reacted with silver nitrate solution to create the Ag NPs.
Methods
The produced nanoparticles have been satisfactorily characterized using UV–Vis spectroscopy, Fourier transform infrared, X-ray diffraction, scanning electron microscopy, energy-dispersive X-ray, transmission electron microscopy, dynamic light scattering, and atomic force microscopy studies. Five groups of rats—normal, ethanolic, comparison control (receiving ranitidine 50 mg/kg), experimental groups 1 and 2, and Ag NPs at dosages of 5 and 20 mg/kg, respectively—were randomly assigned to the animals. The ulcer index, histological examination, assessment of oxidative stress indicators, antioxidant systems, and certain gastric protection variables were used to look into the gastroprotective effects of Ag NPs.
Results
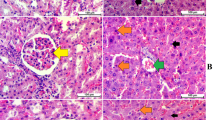
Pretreatment of ulcerated rats with two doses of Ag NPs specifically reduced the negative effects of stomach damage caused by ethanol, as indicated by a decrease in the ulcer index and an increase in the pH of the gastric juice. Furthermore, the findings demonstrated that Ag NPs raised tissue NO, glutathione, catalase activity, and superoxide dismutase while lowering tissue reactive oxygen species, serum nitric oxide, protein carbonyl, and malondialdehyde levels.
Conclusion
In summary, the available data point to Ag NPs as a novel and potential anti-ulcer agent for use in stomach ulcer therapy in the future.
Graphical abstract






Similar content being viewed by others
Data availability
This study’s data are included in the article, and the corresponding author can provide the primary data.
References
Adefisayo MA, Akomolafe RO, Akinsomisoye SO, Alabi QK, Ogundipe OL, Omole JG, Olamilosoye KP (2017) Gastro-protective effect of methanol extract of Vernonia amygdalina (del.) leaf on aspirin-induced gastric ulcer in Wistar rats. Toxicol Rep 4:625–633. https://doi.org/10.1016/j.toxrep.2017.11.004
Amaral GP, de Carvalho NR, Barcelos RP, Dobrachinski F, Portella R, da Silva MH, Lugokenski TH, Dias GR, da Luz SC, Boligon AA, Athayde ML, Villetti MA, Antunes Soares FA, Fachinetto R (2013) Protective action of ethanolic extract of Rosmarinus officinalis L. in gastric ulcer prevention induced by ethanol in rats. Food Chem Toxicol 55:48–55. https://doi.org/10.1016/j.fct.2012.12.038
Matah Marte VM, Ateufack G, Mbiantcha M, Atsamo AD, Adjouzem CF, Djuichou Nguemnang SF, Tsafack EG, Yousseu Nana W, Madjo Kouam YK, Ngoufack Azanze E (2020) Methanolic extract of distemonanthus benthamianus (caesalpiniaceae) stem bark suppresses ethanol/indomethacin-induced chronic gastric injury in rats. Gastroenterol Res Pract 2020:1–14. https://doi.org/10.1155/2020/8180323
Ibrahim IAA, Hussein AI, Muter MS, Mohammed AT, Al-Medhtiy MH, Shareef SH, Aziz PY, Agha NFS, Abdulla MA (2022) Effect of nano silver on gastroprotective activity against ethanol-induced stomach ulcer in rats. Biomed Pharmacother 154:113550. https://doi.org/10.1016/j.biopha.2022.113550
Zhou D, Yang Q, Tian T, Chang Y, Li Y, Duan LR, Li H, Wang SW (2020) Gastroprotective effect of gallic acid against ethanol-induced gastric ulcer in rats: involvement of the Nrf2/HO-1 signaling and anti-apoptosis role. Biomed Pharmacother 126:110075. https://doi.org/10.1016/j.biopha.2020.110075
Mohamed YT, Naguib IA, Abo-Saif AA, Elkomy MH, Alghamdi BS, Mohamed WR (2022) Role of ADMA/DDAH-1 and iNOS/eNOS signaling in the gastroprotective effect of tadalafil against indomethacin-induced gastric injury. Biomed Pharmacother 150:113026. https://doi.org/10.1016/j.biopha.2022.113026
Jain N, Jain P, Rajput D, Patil UK (2021) Green synthesized plant-based silver nanoparticles: therapeutic prospective for anticancer and antiviral activity. Micro Nano Lett 9(1):5. https://doi.org/10.1186/s40486-021-00131-6
Zhang XF, Liu ZG, Shen W, Gurunathan S (2016) Silver nanoparticles: synthesis, characterization, properties, applications, and therapeutic approaches. Int J Mol Sci 17(9):1534. https://doi.org/10.3390/ijms17091534
Akhoondi R, Mirjalili MH, Hadian J (2015) Quantitative and qualitative variations in the essential oil of Rosa foetida Herrm (Rosaceae) flowers as affected by different drying methods. J Essent Oil Res 27(5):421–427. https://doi.org/10.1080/10412905.2015.1025918
Abbasian S, Karimi F, Moghaddam G, Soroush A, Moloudian H, Ahosseini MS (2013) Antioxidant properties of different black tea samples and some Iranian native plants. Pharm Glob 4(2):1–5
Asgarpanah J, Ziarati P, Safialdinardebily M (2014) The volatile oil composition of Rosa foetida Herrm flowers growing wild in Kurdistan province (Iran). J Essent Oil-Bear Plants 17(1):169–172. https://doi.org/10.1080/0972060X.2014.884765
Ayati Z, Ramezani M, Amiri MS, Sahebkar A, Emami SA (2021) Genus Rosa: a review of ethnobotany, phytochemistry and traditional aspects according to islamic traditional medicine (ITM). Adv Exp Med Biol 1308:353–401. https://doi.org/10.1007/978-3-030-64872-5_23
Rezghi M, Hoseinidoust SR, Asgarpanah J (2015) Rosa foetida Herrm. flowers as a future natural antibacterial agent against the main cause of skin burn wound infections Pseudomonas aeroginosa. J Med Herb 5(4):209–213
Slinkard K, Singleton VL (1977) Total phenol analysis: automation and comparison with manual methods. Am J Enol Vitic 28(1):49–55. https://doi.org/10.5344/ajev.1977.28.1.49
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food chem 64(4):555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Dashputre NL, Naikwade NS (2011) Evaluation of anti-ulcer activity of methanolic extract of Abutilon indicum Linn leaves in experimental rats. Int J Pharm Sci Drug Res 3(2):97–100
Takagi K, Okabe S (1968) The effects of drugs on the production and recovery processes of the stress ulcer. Jpn J Pharmacol 18:9–18. https://doi.org/10.1254/jjp.18.9
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1006/abio.1976.9999
Bahramikia S, Ardestani A, Yazdanparast R (2009) Protective effects of four Iranian medicinal plants against free radical-mediated protein oxidation. Food Chem 115(1):37–42. https://doi.org/10.1016/j.foodchem.2008.11.054
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid Peroxidation. In: Methods in enzymology (Vol. 186, pp. 421–431). Academic press. https://doi.org/10.1016/0076-6879(90)86135-I
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. In: Methods in enzymology (Vol. 233, pp. 357–363). Academic press. https://doi.org/10.1016/S0076-6879(94)33041-7
Hortelano S, Dewez B, Genaro AM, Diaz-Guerra MJ, Bosca L (1995) Nitric oxide is released in regenerating liver after partial hepatectomy. J Hepatol 21(3):776–786
Aebi H (1984) Catalase in vitro. In:Methods in enzymology (Vol. 105, pp. 121–126). Academic press. https://doi.org/10.1016/S0076-6879(84)05016-3
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys 21(2):130–132
Jollow DJ, Mitchell JR, Zampaglione NA, Gillette JR (1974) Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3, 4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacol 11(3):151–169. https://doi.org/10.1159/000136485
Roopan SM, Madhumitha G, Rahuman AA, Kamaraj C, Bharathi A, Surendra TV (2013) Low-cost and eco-friendly phyto-synthesis of silver nanoparticles using Cocos nucifera coir extract and its larvicidal activity. Ind Crops Prod 43:631–635. https://doi.org/10.1016/j.indcrop.2012.08.013
Adinortey MB, Ansah C, Galyuon I, Nyarko A (2013) In vivo models used for evaluation of potential antigastroduodenal ulcer agents. Ulcers. https://doi.org/10.1155/2013/796405
Ren S, Wei Y, Niu M, Li R, Wang R, Wei S, Wen J, Wang D, Yang T, Chen X, Wu S, Tong Y, **g M, Li H, Wang M, Zhao Y (2021) Mechanism of rutaecarpine on ethanol-induced acute gastric ulcer using integrated metabolomics and network pharmacology. Biomed Pharmacother 138:111490. https://doi.org/10.1016/j.biopha.2021.111490
Périco LL, Emílio-Silva MT, Ohara R, Rodrigues VP, Bueno G, Barbosa-Filho JM, Rocha LRMD, Batista LM, Hiruma-Lima CA (2020) Systematic analysis of monoterpenes: advances and challenges in the treatment of peptic ulcer diseases. Biomolecules 10(2):265. https://doi.org/10.3390/biom10020265
Aboul Naser A, Younis E, El-Feky A, Elbatanony M, Hamed M (2020) Management of Citrus sinensis peels for protection and treatment against gastric ulcer induced by ethanol in rats. Biomarkers 25(4):349–359. https://doi.org/10.1080/1354750X.2020.1759693
Mousa AM, El-Sammad NM, Hassan SK, Madboli AENA, Hashim AN, Moustafa ES, Bakry SM, Elsayed EA (2019) Antiulcerogenic effect of Cuphea ignea extract against ethanol-induced gastric ulcer in rats. BMC Complement Altern Med 19(1):345. https://doi.org/10.1186/s12906-019-2760-9
Beiranvand M, Bahramikia S (2020) Ameliorating and protective effects mesalazine on ethanol-induced gastric ulcers in experimental rats. Eur J Pharmacol 888:173573. https://doi.org/10.1016/j.ejphar.2020.173573
Alimi H, Hfaiedh N, Bouoni Z, Sakly M, Rhouma KB (2011) Evaluation of antioxidant and antiulcerogenic activities of Opuntia ficus indica f. inermis flowers extract in rats. Environ Toxicol Pharmacol 32(3):406–416. https://doi.org/10.1016/j.etap.2011.08.007
Song YR, Kim JK, Lee HS, Kim SG, Choi EK (2020) Serum levels of protein carbonyl, a marker of oxidative stress, are associated with overhydration, sarcopenia and mortality in hemodialysis patients. BMC Nephrol 21:1–11. https://doi.org/10.1186/s12882-020-01937-z
Pradeep AR, Ramchandraprasad MV, Bajaj P, Rao NS, Agarwal E (2013) Protein carbonyl: an oxidative stress marker in gingival crevicular fluid in healthy, gingivitis, and chronic periodontitis subjects. Contemp Clin Dent 4(1):27. https://doi.org/10.4103/0976-237X.111589
Kahraman A, Erkasap N, Köken T, Serteser M, Aktepe F, Erkasap S (2003) The antioxidative and antihistaminic properties of quercetin in ethanol-induced gastric lesions. Toxicology 183(1–3):133–142. https://doi.org/10.1016/s0300-483x(02)00514-0
Zakaria ZA, Balan T, Azemi AK, Omar MH, Mohtarrudin N, Ahmad Z, Abdullah MN, Desa MN, Teh LK, Salleh MZ (2016) Mechanism(s) of action underlying the gastroprotective effect of ethyl acetate fraction obtained from the crude methanolic leaves extract of Muntingia calabura. BMC Complement Altern Med 16:78. https://doi.org/10.1186/s12906-016-1041-0
Shams SGE, Eissa RG (2022) Amelioration of ethanol-induced gastric ulcer in rats by quercetin: implication of Nrf2/HO1 and HMGB1/TLR4/NF-κB pathways. Heliyon 8(10):e11159. https://doi.org/10.1016/j.heliyon.2022.e11159
Liu B, Feng X, Zhang J, Wei Y, Zhao X (2019) Preventive effect of Anji white tea flavonoids on alcohol-induced gastric injury through their antioxidant effects in Kunming mice. Biomolecules 9(4):137. https://doi.org/10.3390/biom9040137
Chen XY, Chen HM, Liu YH, Zhang ZB, Zheng YF, Su ZQ, Zhang X, **e JH, Liang YZ, Fu LD, Lai XP, Su ZR, Huang XQ (2016) The gastroprotective effect of pogostone from Pogostemonis Herba against indomethacin-induced gastric ulcer in rats. Exp Biol Med (Maywood) 241(2):193–204. https://doi.org/10.1177/1535370215600099
Yu C, Mei XT, Zheng YP, Xu DH (2014) Gastroprotective effect of taurine zinc solid dispersions against absolute ethanol-induced gastric lesions is mediated by enhancement of antioxidant activity and endogenous PGE2 production and attenuation of NO production. Eur J Pharmacol 740:329–336. https://doi.org/10.1016/j.ejphar.2014.07.014
de Almeida CLF, Brito SA, de Santana TI, Costa HBA, de Carvalho Júnior CHR, da Silva MV, de Almeida LL, Rolim LA, Dos Santos VL, Wanderley AG, da Silva TG (2017) Spondias purpurea L. (Anacardiaceae): antioxidant and antiulcer activities of the leaf hexane extract. Oxid Med Cell Longev. https://doi.org/10.1155/2017/6593073
Li Q, Hu X, Xuan Y, Ying J, Fei Y, Rong J, Zhang Y, Zhang J, Liu C, Liu Z (2018) Kaempferol protects ethanol-induced gastric ulcers in mice via pro-inflammatory cytokines and NO. Acta Biochim Biophys Sin (Shanghai) 50(3):246–253. https://doi.org/10.1093/abbs/gmy002
Lebda MA, El-Far AH, Noreldin AE, Elewa YH, Al Jaouni SK, Mousa SA (2018) Protective effects of miswak (Salvadora persica) against experimentally induced gastric ulcers in rats. Oxid Med Cell Longev. https://doi.org/10.1155/2018/6703296
Pan LR, Tang Q, Fu Q, Hu BR, **ang JZ, Qian JQ (2005) Roles of nitric oxide in protective effect of berberine in ethanol-induced gastric ulcer mice. Acta Pharmacol Sin 26(11):1334–1338. https://doi.org/10.1111/j.1745-7254.2005.00186.x
Acknowledgements
The authors appreciate the financial support of this investigation by Lorestan University. This project did not receive any specific grant from funding agencies.
Author information
Authors and Affiliations
Contributions
The core idea of this study came from S. B. He also directed the other authors and analyzed the collected papers. S. M, F. H, and M. N-A conducted the experiments and wrote the manuscript. S. B supervised the study. All authors have declared that no conflict of interest exists. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Shima Mostaed, Seifollah Bahramikia, Faranak Hadi and Mohsen Nabi Afjadi declare that they have no conflict of interest in the manuscript.
Ethical approval
All procedures performed in this study involving animals were following the ethical standards and the international regulations of the usage and welfare of laboratory animals and were approved by the clinical ethics committee of the Faculty of Veterinary Medicine of Lorestan University with (Ethical code: (LU.ECRA.2021.33).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mostaed, S., Bahramikia, S., Hadi, F. et al. Gastroprotective effects of the silver nanoparticles synthesized from Rosa foetida Herrm. against ethanol-induced stomach ulcers. Toxicol. Environ. Health Sci. (2024). https://doi.org/10.1007/s13530-024-00219-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s13530-024-00219-7