Abstract
Background
Tamoxifen (TAM) is recommended as the first-line strategy for men with estrogen receptor (ER)-positive early breast cancer who are candidates for adjuvant endocrine therapy in ASCO guideline. Our study aims to analyze the cost-effectiveness of receiving adjuvant endocrine therapy with TAM compared to no TAM, and to assess the cost-effectiveness of using TAM with high adherence over low adherence for ER-positive early male breast cancer in the USA.
Methods
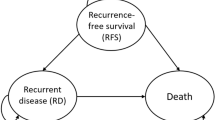
Two Markov models comprising three mutually exclusive health states were constructed: (1) the first Markov model compared the cost-effectiveness of adding TAM with not using TAM (TAM versus Not-TAM); (2) the second model compared the cost-effectiveness of receiving TAM with high adherence and low adherence (High-adherence-TAM versus Low-adherence-TAM). The simulation time horizon for both models was the lifetime of patients. The efficacy and safety data of two models were elicited from the real-world studies. Model inputs were derived from the US website and published literature. The main outcomes of two models both included the total cost, quality-adjusted life-years (QALYs), and incremental cost-effectiveness ratios (ICERs).
Results
In the first model, TAM yielded an ICER of $5707.29 per QALY compared to Not-TAM, which was substantially below the WTP threshold of $50,000.00 per QALY in the USA. Probabilistic sensitivity analysis results demonstrated a 100.00% probability of cost-effectiveness for this strategy. In the second model, High-adherence-TAM was dominated absolutely compared to Low-adherence-TAM. The High-adherence-TAM was cost-effective with a 99.70% probability over Low-adherence-TAM when WTP was set as $50,000.00/QALY. All of these parameters within their plausible ranges did not reversely change the results of our models.
Conclusions
Our study will offer valuable guidance for physicians or patients when making treatment decisions and provide an effective reference for decision-making to consider the appropriate allocation of funds to this special group.



Similar content being viewed by others
Data availability
All data generated or analyzed during this article are included in the published article (and in its supplementary information files).
References
Fox S, Speirs V, Shaaban AM. Male breast cancer: an update. Virchows Arch. 2022;480(1):85–93.
Reddington R, Galer M, Hagedorn A, et al. Incidence of male breast cancer in Scotland over a twenty-five-year period (1992–2017). Eur J Surg Oncol. 2020;46(8):1546–50.
Speirs V, Shaaban AM. The rising incidence of male breast cancer. Breast Cancer Res Treat. 2009;115(2):429–30.
Sasco AJ, Lowenfels AB, Pasker-de JP. Review article: epidemiology of male breast cancer. A meta-analysis of published case-control studies and discussion of selected aetiological factors. Int J Cancer. 1993;53(4):538–49.
Key Statistics for Breast Cancer in Men. 2024. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html. Accessed 6 Mar 2024.
Schröder CP, van Leeuwen-Stok E, Cardoso F, et al. Quality of life in male breast cancer: prospective study of the International Male Breast Cancer Program (EORTC10085/TBCRC029/BIG2-07/NABCG). Oncologist. 2023;28(10):e877–83.
Ruddy KJ, Giobbie-Hurder A, Giordano SH, et al. Quality of life and symptoms in male breast cancer survivors. Breast. 2013;22(2):197–9.
Kornegoor R, Verschuur-Maes AH, Buerger H, et al. Molecular subty** of male breast cancer by immunohistochemistry. Mod Pathol. 2012;25(3):398–404.
Hassett MJ, Somerfield MR, Baker ER, et al. Management of male breast cancer: ASCO guideline. J Clin Oncol. 2020;38(16):1849–63.
Wibowo E, Pollock PA, Hollis N, et al. Tamoxifen in men: a review of adverse events. Andrology. 2016;4(5):776–88.
Palva T, Ranta H, Koivisto AM, et al. A double-blind placebo-controlled study to evaluate endometrial safety and gynaecological symptoms in women treated for up to 5 years with tamoxifen or placebo – a substudy for IBIS I Breast Cancer Prevention Trial. Eur J Cancer. 2013;49(1):45–51.
Setyawan J, Azimi N, Strand V, et al. Reporting of thromboembolic events with JAK inhibitors: analysis of the FAERS Database 2010–2019. Drug Saf. 2021;44(8):889–97.
Visram H, Kanji F, Dent SF. Endocrine therapy for male breast cancer: rates of toxicity and adherence. Curr Oncol. 2010;17(5):17–21.
Anelli TF, Anelli A, Tran KN, et al. Tamoxifen administration is associated with a high rate of treatment-limiting symptoms in male breast cancer patients. Cancer. 1994;74(1):74–7.
Pemmaraju N, Munsell MF, Hortobagyi GN, et al. Retrospective review of male breast cancer patients: analysis of tamoxifen-related side-effects. Ann Oncol. 2012;23(6):1471–4.
Oke O, Niu J, Chavez-MacGregor M, et al. Adjuvant tamoxifen adherence in men with early-stage breast cancer. Cancer. 2022;128(1):59–64.
Falkson CI. Adjuvant tamoxifen adherence in male patients with breast cancer: an ongoing challenge. Cancer. 2022;128(1):22–4.
Xu S, Yang Y, Tao W, et al. Tamoxifen adherence and its relationship to mortality in 116 men with breast cancer. Breast Cancer Res Treat. 2012;136(2):495–502.
Smith TJ, Hillner BE. The efficacy and cost-effectiveness of adjuvant therapy of early breast cancer in premenopausal women. J Clin Oncol. 1993;11(4):771–6.
Butani D, Gupta N, Jyani G, et al. Cost-effectiveness of tamoxifen, aromatase inhibitor, and switch therapy (adjuvant endocrine therapy) for breast cancer in hormone receptor positive postmenopausal women in India. Breast Cancer (Dove Med Press). 2021;13:625–40.
Noah-Vanhoucke J, Green LE, Dinh TA, et al. Cost-effectiveness of chemoprevention of breast cancer using tamoxifen in a postmenopausal US population. Cancer. 2011;117(15):3322–31.
Hong JH, Ha KS, Jung YH, et al. Clinical features of male breast cancer: experiences from seven institutions over 20 years. Cancer Res Treat. 2016;48(4):1389–98.
Cardoso F, Bartlett JMS, Slaets L, et al. Characterization of male breast cancer: results of the EORTC 10085/TBCRC/BIG/NABCG International Male Breast Cancer Program. Ann Oncol. 2018;29(2):405–17.
Smith TJ, Hillner BE, Desch CE. Efficacy and cost-effectiveness of cancer treatment: rational allocation of resources based on decision analysis. J Natl Cancer Inst. 1993;85(18):1460–74.
Weinstein MC, Toy EL, Sandberg EA, et al. Modeling for health care and other policy decisions: uses, roles, and validity. Value Health. 2001;4(5):348–61.
Husereau D, Drummond M, Augustovski F, et al. Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) statement: updated reporting guidance for health economic evaluations. BMC Med. 2022;20(1):23.
Venigalla S, Carmona R, Guttmann DM, et al. Use and effectiveness of adjuvant endocrine therapy for hormone receptor-positive breast cancer in men. JAMA Oncol. 2018;4(10): e181114.
Giordano SH, Perkins GH, Broglio K, et al. Adjuvant systemic therapy for male breast carcinoma. Cancer. 2005;104(11):2359–64.
Guyot P, Ades AE, Ouwens MJ, et al. Enhanced secondary analysis of survival data: reconstructing the data from published Kaplan–Meier survival curves. BMC Med Res Methodol. 2012;12:9.
Drugstore.com Inc. 2024. http://www.drugstore.com/pharmacy. Accessed 22 Feb 2024.
US Bureau of Labor Statistics. Division of Consumer Prices and Price Indexes. Consumer Price Index—All Urban Consumers—US Medical Care, 1947–2024. 2024. https://www.bls.gov/cpi/data.htm. Accessed 18 Mar 2024.
Delea TE, Karnon J, Sofrygin O, et al. Cost-effectiveness of letrozole versus tamoxifen as initial adjuvant therapy in hormone receptor-positive postmenopausal women with early-stage breast cancer. Clin Breast Cancer. 2007;7(8):608–18.
Yang JJ, Park SK, Cho LY, et al. Cost-effectiveness analysis of 5 years of postoperative adjuvant tamoxifen therapy for Korean women with breast cancer: retrospective cohort study of the Korean breast cancer society database. Clin Ther. 2010;32(6):1122–38.
Addo R, Haas M, Goodall S. The cost-effectiveness of adjuvant tamoxifen treatment of hormone receptor-positive early breast cancer among premenopausal and perimenopausal Ghanaian women. Value Health Reg Issues. 2021;25:196–205.
Yao N, Shi W, Liu T, et al. Clinicopathologic characteristics and prognosis for male breast cancer compared to female breast cancer. Sci Rep. 2022;12(1):220.
ClinicalTrials.gov. 2024. https://clinicaltrials.gov/search?cond=Breast%20Cancer&intr=Tamoxifen&viewType=Table&term=Male. Accessed 17 Mar 2024.
Gorin MB, Day R, Costantino JP, et al. Long-term tamoxifen citrate use and potential ocular toxicity. Am J Ophthalmol. 1998;125(4):493–501.
Huang Y, Huang X, Huang X, et al. Cost-effectiveness analysis of ovarian function preservation with GnRH agonist during chemotherapy in premenopausal women with early breast cancer. Hum Reprod. 2023;38(6):1099–110.
Chan-Tack KM. Oral sildenafil in erectile dysfunction. Lancet. 1998;352(9139):1557.
Carvalho de Oliveira CC, Agati LB, Ribeiro CM, et al. Cost-effectiveness analysis of extended thromboprophylaxis with rivaroxaban versus no prophylaxis in high-risk patients after hospitalisation for COVID-19: an economic modelling study. Lancet Reg Health Am. 2023;24: 100543.
Darden M, Espie CA, Carl JR, et al. Cost-effectiveness of digital cognitive behavioral therapy (Sleepio) for insomnia: a Markov simulation model in the United States. Sleep. 2021;44(4): zsaa223.
Counterweight Project Team. Influence of body mass index on prescribing costs and potential cost savings of a weight management programme in primary care. J Health Serv Res Policy. 2008;13(3):158–66.
Greenberg PE, Fournier AA, Sisitsky T, et al. The economic burden of adults with major depressive disorder in the United States (2005 and 2010). J Clin Psychiatry. 2015;76(2):155–62.
Lamerato L, Havstad S, Gandhi S, et al. Economic burden associated with breast cancer recurrence: findings from a retrospective analysis of health system data. Cancer. 2006;106(9):1875–82.
Delea TE, Taneja C, Sofrygin O, et al. Cost-effectiveness of zoledronic acid plus endocrine therapy in premenopausal women with hormone-responsive early breast cancer. Clin Breast Cancer. 2010;10(4):267–74.
Retèl VP, Byng D, Linn SC, et al. Cost-effectiveness analysis of the 70-gene signature compared with clinical assessment in breast cancer based on a randomised controlled trial. Eur J Cancer. 2020;137:193–203.
Peasgood T, Ward SE, Brazier J. Health-state utility values in breast cancer. Expert Rev Pharmacoecon Outcomes Res. 2010;10(5):553–66.
Tsoi B, Blackhouse G, Ferrazzi S, et al. Incorporating ulipristal acetate in the care of symptomatic uterine fibroids: a Canadian cost-utility analysis of pharmacotherapy management. Clinicoecon Outcomes Res. 2015;7:213–25.
Smith KJ, Roberts MS. The cost-effectiveness of sildenafil. Ann Intern Med. 2000;132(12):933–7.
Guy H, Laskier V, Fisher M, et al. Cost-effectiveness of betrixaban compared with enoxaparin for venous thromboembolism prophylaxis in nonsurgical patients with acute medical illness in the United States. Pharmacoeconomics. 2019;37(5):701–14.
Ikeda S, Azuma M, Fujimoto K, et al. PMH8 EQ-5D analysis in patients with insomnia: change of quality of life in lemborexant phase 3 trial sunrise 1. Value Health. 2020;23:S585.
Trueman P, Haynes SM, Felicity Lyons G, et al. Long-term cost-effectiveness of weight management in primary care. Int J Clin Pract. 2010;64(6):775–83.
Saarni SI, Härkänen T, Sintonen H, et al. The impact of 29 chronic conditions on health-related quality of life: a general population survey in Finland using 15D and EQ-5D. Qual Life Res. 2006;15(8):1403–14.
Sullivan JK, Huizinga J, Edwards RR, et al. Cost-effectiveness of duloxetine for knee OA subjects: the role of pain severity. Osteoarthritis Cartilage. 2021;29(1):28–38.
Eggemann H, Bernreiter AL, Reinisch M, et al. Tamoxifen treatment for male breast cancer and risk of thromboembolism: prospective cohort analysis. Br J Cancer. 2019;120(3):301–5.
Reinisch M, Seiler S, Hauzenberger T, et al. Efficacy of endocrine therapy for the treatment of breast cancer in men: results from the MALE phase 2 randomized clinical trial. JAMA Oncol. 2021;7(4):565–72.
Food and Drug Administration. 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/021807s006lbl.pdf. Accessed 13 Mar 2024.
Acknowledgements
None.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
YPH: conception and design, acquisition of data, analysis and interpretation of data, drafting of the article, final approval of the manuscript. CJK: conception and design, analysis and interpretation of data, revision of the article for critically important intellectual content, final approval of the manuscript. JQC: acquisition of data, analysis and interpretation of data, final approval of the manuscript. XXW: acquisition of data, final approval of the manuscript. MHC: acquisition of data, final approval of the manuscript. HS: conception and design, revision of the article for critically important intellectual content, final approval of the manuscript.