Abstract
Both reduced nicotinamide adenine dinucleotide phosphate (NADPH) and β-nicotinamide adenine dinucleotide hydrate (NAD+) have been reported to have potent neuroprotective effects against ischemic neuronal injury. Both NADPH and NAD+ are essential cofactors for anti-oxidation and cellular energy metabolism. We investigated if combined NADPH and NAD+ could offer better neuroprotective effects on cellular and animal models of ischemic stroke. In vitro studies with primary cultured neurons demonstrated that NAD+ was effective in protecting neurons against oxygen–glucose deprivation/reoxygenation (OGD/R) injury when given during the early time period of reoxygenation. In vivo studies in mice also suggested that NAD+ was effective for ameliorating ischemic brain damage when administered within 2 h after reperfusion. The combination of NADPH and NAD+ provided not only greater beneficial effects but also larger therapeutic window in both cellular and animal models of stroke. The combination of NADPH and NAD+ significantly increased the levels of adenosine triphosphate (ATP) and reduced the levels of reactive oxygen species (ROS) and oxidative damage of macromolecules. Furthermore, the combined medication significantly reduced long-term mortality, improved the functional recovery, and inhibited signaling pathways involved in apoptosis and necroptosis after ischemic stroke. The present study indicates that the combination of NAD+ and NADPH can produce greater therapeutic effects with smaller dose of NADPH; on the other hand, NADPH can significantly prolong the therapeutic window of NAD+. The current results suggest that the combination of NADPH and NAD+ may provide a novel effective therapy for ischemic stroke.






Similar content being viewed by others
References
Lyden PD (2001) Further randomized controlled trials of tPA within 3 hours are required-not. Stroke 32(11):2709–2710
Moro MA, Almeida A, Bolanos JP, Lizasoain I (2005) Mitochondrial respiratory chain and free radical generation in stroke. Free Radic Biol Med 39(10):1291–1304. https://doi.org/10.1016/j.freeradbiomed.2005.07.010
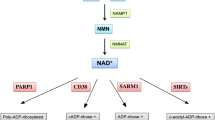
Canto C, Menzies KJ, Auwerx J (2015) NAD(+) metabolism and the control of energy homeostasis: A balancing act between mitochondria and the nucleus. Cell Metab 22(1):31–53. https://doi.org/10.1016/j.cmet.2015.05.023
Alano CC, Garnier P, Ying W, Higashi Y, Kauppinen TM, Swanson RA (2010) NAD+ depletion is necessary and sufficient for poly(ADP-ribose) polymerase-1-mediated neuronal death. J Neurosci 30(8):2967–2978. https://doi.org/10.1523/JNEUROSCI.5552-09.2010
Sheline CT, Behrens MM, Choi DW (2000) Zinc-induced cortical neuronal death: contribution of energy failure attributable to loss of NAD(+) and inhibition of glycolysis. J Neurosci 20(9):3139–3146
Becatti M, Taddei N, Cecchi C, Nassi N, Nassi PA, Fiorillo C (2012) SIRT1 modulates MAPK pathways in ischemic-reperfused cardiomyocytes. Cell Mol Life Sci 69(13):2245–2260. https://doi.org/10.1007/s00018-012-0925-5
Hsu CP, Oka S, Shao D, Hariharan N, Sadoshima J (2009) Nicotinamide phosphoribosyltransferase regulates cell survival through NAD+ synthesis in cardiac myocytes. Circ Res 105(5):481–491. https://doi.org/10.1161/CIRCRESAHA.109.203703
Alano CC, Ying W, Swanson RA (2004) Poly(ADP-ribose) polymerase-1-mediated cell death in astrocytes requires NAD+ depletion and mitochondrial permeability transition. J Biol Chem 279(18):18895–18902. https://doi.org/10.1074/jbc.M313329200
Ying W, Wei G, Wang D, Wang Q, Tang X, Shi J, Zhang P, Lu H (2007) Intranasal administration with NAD+ profoundly decreases brain injury in a rat model of transient focal ischemia. Front Biosci 12:2728–2734
Ying W (2008) NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences. Antioxid Redox Signal 10(2):179–206. https://doi.org/10.1089/ars.2007.1672
Li M, Zhou ZP, Sun M, Cao L, Chen J, Qin YY, JH G, Han F et al (2016) Reduced nicotinamide adenine dinucleotide phosphate, a pentose phosphate pathway product, might be a novel drug candidate for ischemic stroke. Stroke 47(1):187–195. https://doi.org/10.1161/STROKEAHA.115.009687
Marutani E, Kosugi S, Tokuda K, Khatri A, Nguyen R, Atochin DN, Kida K, Van Leyen K et al (2012) A novel hydrogen sulfide-releasing N-methyl-D-aspartate receptor antagonist prevents ischemic neuronal death. J Biol Chem 287(38):32124–32135. https://doi.org/10.1074/jbc.M112.374124
Cao G, **ng J, **ao X, Liou AK, Gao Y, Yin XM, Clark RS, Graham SH et al (2007) Critical role of calpain I in mitochondrial release of apoptosis-inducing factor in ischemic neuronal injury. J Neurosci 27(35):9278–9293. https://doi.org/10.1523/JNEUROSCI.2826-07.2007
Clark WM, Lessov NS, Dixon MP, Eckenstein F (1997) Monofilament intraluminal middle cerebral artery occlusion in the mouse. Neurol Res 19(6):641–648
Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M, Chopp M (2001) Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke 32(4):1005–1011
Muller HD, Hanumanthiah KM, Diederich K, Schwab S, Schabitz WR, Sommer C (2008) Brain-derived neurotrophic factor but not forced arm use improves long-term outcome after photothrombotic stroke and transiently upregulates binding densities of excitatory glutamate receptors in the rat brain. Stroke 39(3):1012–1021. https://doi.org/10.1161/STROKEAHA.107.495069
Yamada K, Tanaka T, Zou LB, Senzaki K, Yano K, Osada T, Ana O, Ren X et al (1999) Long-term deprivation of oestrogens by ovariectomy potentiates beta-amyloid-induced working memory deficits in rats. Br J Pharmacol 128(2):419–427. https://doi.org/10.1038/sj.bjp.0702811
Sasaki YF, Tsuda S, Izumiyama F, Nishidate E (1997) Detection of chemically induced DNA lesions in multiple mouse organs (liver, lung, spleen, kidney, and bone marrow) using the alkaline single cell gel electrophoresis (comet) assay. Mutat Res 388(1):33–44
Zhang F, Wang S, Signore AP, Chen J (2007) Neuroprotective effects of leptin against ischemic injury induced by oxygen-glucose deprivation and transient cerebral ischemia. Stroke 38(8):2329–2336. https://doi.org/10.1161/STROKEAHA.107.482786
Kang X, Chen J, Xu Z, Li H, Wang B (2007) Protective effects of Ginkgo biloba extract on paraquat-induced apoptosis of PC12 cells. Toxicol In Vitro 21(6):1003–1009. https://doi.org/10.1016/j.tiv.2007.02.004
Won SJ, Choi BY, Yoo BH, Sohn M, Ying W, Swanson RA, Suh SW (2012) Prevention of traumatic brain injury-induced neuron death by intranasal delivery of nicotinamide adenine dinucleotide. J Neurotrauma 29(7):1401–1409. https://doi.org/10.1089/neu.2011.2228
Lo EH, Dalkara T, Moskowitz MA (2003) Mechanisms, challenges and opportunities in stroke. Nat Rev Neurosci 4(5):399–415. https://doi.org/10.1038/nrn1106
Chen WW, Yu H, Fan HB, Zhang CC, Zhang M, Zhang C, Cheng Y, Kong J et al (2012) RIP1 mediates the protection of geldanamycin on neuronal injury induced by oxygen-glucose deprivation combined with zVAD in primary cortical neurons. J Neurochem 120(1):70–77. https://doi.org/10.1111/j.1471-4159.2011.07526.x
Han J, Shi S, Min L, Wu T, **a W, Ying W (2011) NAD+ treatment induces delayed autophagy in Neuro2a cells partially by increasing oxidative stress. Neurochem Res 36(12):2270–2277. https://doi.org/10.1007/s11064-011-0551-x
Funding
This study was supported by the Natural Science 583 foundation of China (Nos. 31500822, 81730092, 81271459, 31030034), the Priority Academic Program Development of Jiangsu Higher Education Institutes (PAPD), the Graduate Education Innovation Project of Jiangsu Province (CXZZ12_0850), and the Jiangsu Provincial Medical Youth Talent (QNRC2016762).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal experiments were performed in accordance with the institutional guidelines for animal use and care, and the study protocol was approved by the ethical committee of Soochow University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOC 5521 kb)
Rights and permissions
About this article
Cite this article
Huang, Q., Sun, M., Li, M. et al. Combination of NAD+ and NADPH Offers Greater Neuroprotection in Ischemic Stroke Models by Relieving Metabolic Stress. Mol Neurobiol 55, 6063–6075 (2018). https://doi.org/10.1007/s12035-017-0809-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0809-7