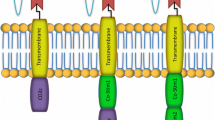
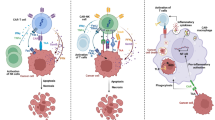
Abstract
Chimeric Antigen Receptor T cell (CAR-T) therapy has revolutionized cancer treatment, but its application to solid tumors is limited. CAR-T cells have poor incapability of entering, surviving, proliferating, and finally exerting function in the tumor microenvironment. This review summarizes the main strategies related to enhancing the infiltration, efficacy, antigen recognition, and production of CAR-T in solid tumors. Additional applications of CAR-γδ T and macrophages are also discussed. We believe CAR-T will be a milestone in treating solid tumors once these problems are solved.


Similar content being viewed by others
Data availability
The articles analyzed during the current study are available in the literature and listed in the references.
Abbreviations
- CAR:
-
Chimeric antigen receptor
- scFv:
-
Single-chain fragment variable
- TAA:
-
Tumor-associated antigen
- TME:
-
Tumor microenvironment
- MDSC:
-
Myeloid-derived suppressor cell
- HRE:
-
Hypoxia response element
- PSMA:
-
Prostate-specific membrane antigen
- CCL2:
-
C–C motif chemokine ligand 2
- DC:
-
Dendritic cells
- CXCL12:
-
C-X-C motif chemokine ligand 12
- CSF1:
-
Colony-stimulating factor 1
- VEGF:
-
Vascular endothelial growth factor
- CAR-M:
-
CAR-engineered macrophages
- CSR:
-
Chimeric switch receptor
- PD1:
-
Programmed death-1
- CEA:
-
Carcinoembryonic antigen
- TAM:
-
Tumor-associated macrophages
- PDAC:
-
Pancreatic ductal adenocarcinoma
- iCAR:
-
Inhibitory CAR
- PSCA:
-
Prostate stem cell antigen
- TRUCK:
-
T cells redirected for universal cytokine-mediated killing
- ZOL:
-
Zoledronate
- CAIX:
-
Carbonic anhydrase IX
- iPSC:
-
Induced pluripotent stem cell
- HPSE:
-
Expression of heparinase
- ECM:
-
Extracellular matrix
- CAF:
-
Cancer-associated fibroblasts
- FAP:
-
Fibroblast activation protein
- DNR:
-
Dominant-negative receptors
- synNotch:
-
Synthetic Notch receptor
- SDF-1α:
-
Secrete stromal cell-derived factor 1α
- CRS:
-
Cytokine Release Syndrome
- CCR:
-
Chimeric costimulatory receptor
- TSAs:
-
Tumor-specific antigens
References
Albelda SM. CAR T cell therapy for patients with solid tumors: key lessons to learn and unlearn. Nat Rev Clin Oncol. 2023;21:47–66.
Li G, et al. IL-7 and CCR2b co-expression-mediated enhanced CAR-T survival and infiltration in solid tumors. Front Oncol. 2021;11: 734593.
Wang Y, et al. Chemokine receptor CCR2b enhanced anti-tumor function of chimeric antigen receptor T cells targeting mesothelin in a non-small-cell lung carcinoma model. Front Immunol. 2021;12: 628906.
Moon EK, et al. Expression of a functional CCR2 receptor enhances tumor localization and tumor eradication by retargeted human T cells expressing a mesothelin-specific chimeric antibody receptor. Clin Cancer Res Off J Am Assoc Cancer Res. 2011;17(14):4719–30.
Whilding LM, et al. CAR T-cells targeting the integrin αvβ6 and co-expressing the chemokine receptor CXCR2 demonstrate enhanced homing and efficacy against several solid malignancies. Cancers. 2019;11(5):674.
Li G, et al. CXCR5 guides migration and tumor eradication of anti-EGFR chimeric antigen receptor T cells. Mol Ther Oncolytics. 2021;22:507–17.
Luo H, et al. Coexpression of IL7 and CCL21 increases efficacy of CAR-T cells in solid tumors without requiring preconditioned lymphodepletion. Clin Cancer Res Off J Am Assoc Cancer Res. 2020;26(20):5494–505.
Hu J-F, et al. Induced expression of CCL19 promotes the anti-tumor ability of CAR-T cells by increasing their infiltration ability. Front Immunol. 2022;13: 958960.
Adachi K, et al. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat Biotechnol. 2018;36(4):346–51.
Lu L-L, et al. GPC3-IL7-CCL19-CAR-T primes immune microenvironment reconstitution for hepatocellular carcinoma therapy. Cell Biol Toxicol. 2023;39(6):3101–19.
Caruana I, et al. Heparanase promotes tumor infiltration and antitumor activity of CAR-redirected T lymphocytes. Nat Med. 2015;21(5):524–9.
Zhao Y, et al. Bioorthogonal equip** CAR-T cells with hyaluronidase and checkpoint blocking antibody for enhanced solid tumor immunotherapy. ACS Cent Sci. 2022;8(5):603–14.
Whittle MC, Hingorani SR. Fibroblasts in pancreatic ductal adenocarcinoma: biological mechanisms and therapeutic targets. Gastroenterology. 2019;156(7):2085–96.
Chen Y, McAndrews KM, Kalluri R. Clinical and therapeutic relevance of cancer-associated fibroblasts. Nat Rev Clin Oncol. 2021;18(12):792–804.
**ao Z, et al. Desmoplastic stroma restricts T cell extravasation and mediates immune exclusion and immunosuppression in solid tumors. Nat Commun. 2023;14(1):5110.
Liu Y, et al. FAP-targeted CAR-T suppresses MDSCs recruitment to improve the antitumor efficacy of claudin18.2-targeted CAR-T against pancreatic cancer. J Transl Med. 2023;21(1):255.
Tran E, et al. Immune targeting of fibroblast activation protein triggers recognition of multipotent bone marrow stromal cells and cachexia. J Exp Med. 2013;210(6):1125–35.
Chen Y, et al. Therapeutic potential of TNFα and IL1β blockade for CRS/ICANS in CAR-T therapy via ameliorating endothelial activation. Front Immunol. 2021;12: 623610.
Chen Y, et al. Eradication of neuroblastoma by T cells redirected with an optimized GD2-specific chimeric antigen receptor and interleukin-15. Clin Cancer Res Off J Am Assoc Cancer Res. 2019;25(9):2915–24.
Lanitis E, et al. Optimized gene engineering of murine CAR-T cells reveals the beneficial effects of IL-15 coexpression. J Exp Med. 2021;218(2): e20192203.
Zhang Z, et al. Gene-edited interleukin CAR-T cells therapy in the treatment of malignancies: present and future. Front Immunol. 2021;12: 718686.
Kim MY, et al. A long-acting interleukin-7, rhIL-7-hyFc, enhances CAR T cell expansion, persistence, and anti-tumor activity. Nat Commun. 2022;13(1):3296.
Aspuria P-J, et al. An orthogonal IL-2 and IL-2Rβ system drives persistence and activation of CAR T cells and clearance of bulky lymphoma. Sci Transl Med. 2021;13(625):eabg7565.
Ramos TL, et al. Prevention of acute GVHD using an orthogonal IL-2/IL-2Rβ system to selectively expand regulatory T cells in vivo. Blood. 2023;141(11):1337–52.
Zhang Q, et al. A human orthogonal IL-2 and IL-2Rβ system enhances CAR T cell expansion and antitumor activity in a murine model of leukemia. Sci Transl Med. 2021;13(625):eabg6986.
Kalbasi A, et al. Potentiating adoptive cell therapy using synthetic IL-9 receptors. Nature. 2022;607(7918):360–5.
Kagoya Y, et al. A novel chimeric antigen receptor containing a JAK-STAT signaling domain mediates superior antitumor effects. Nat Med. 2018;24(3):352–9.
Ebner R, et al. Cloning of a type I TGF-β receptor and its effect on TGF-β binding to the type II receptor. Science. 1993;260(5112):1344–8.
Kloss CC, et al. Dominant-negative TGF-β receptor enhances PSMA-targeted human CAR T cell proliferation and augments prostate cancer eradication. Mol Ther. 2018;26(7):1855–66.
Li K, et al. Dominant-negative transforming growth factor-β receptor-armoured mesothelin-targeted chimeric antigen receptor T cells slow tumour growth in a mouse model of ovarian cancer. Cancer Immunol Immunother. 2023;72(4):917–28.
Stüber T, et al. Inhibition of TGF-β-receptor signaling augments the antitumor function of ROR1-specific CAR T-cells against triple-negative breast cancer. J Immunother Cancer. 2020;8(1): e000676.
Narayan V, et al. PSMA-targeting TGFβ-insensitive armored CAR T cells in metastatic castration-resistant prostate cancer: a phase 1 trial. Nat Med. 2022;28(4):724–34.
Sukumaran S, et al. Enhancing the potency and specificity of engineered T cells for cancer treatment. Cancer Discov. 2018;8(8):972–87.
Mohammed S, et al. Improving chimeric antigen receptor-modified T cell function by reversing the immunosuppressive tumor microenvironment of pancreatic cancer. Mol Ther. 2017;25(1):249–58.
Wilkie S, et al. Selective expansion of chimeric antigen receptor-targeted T-cells with potent effector function using interleukin-4. J Biol Chem. 2010;285(33):25538–44.
Qiao Y, et al. Enhancement of CAR-T cell activity against cholangiocarcinoma by simultaneous knockdown of six inhibitory membrane proteins. Cancer Commun (Lond Engl). 2023;43(7):788–807.
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020;30(8):660–9.
Kim W, et al. PD-1 signaling promotes tumor-infiltrating myeloid-derived suppressor cells and gastric tumorigenesis in mice. Gastroenterology. 2021;160(3):781–96.
Cherkassky L, et al. Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition. J Clin Investig. 2016;126(8):3130–44.
Prosser ME, et al. Tumor PD-L1 co-stimulates primary human CD8(+) cytotoxic T cells modified to express a PD1:CD28 chimeric receptor. Mol Immunol. 2012;51(3–4):263–72.
Liu X, et al. A chimeric switch-receptor targeting PD1 augments the efficacy of second-generation CAR T cells in advanced solid tumors. Cancer Res. 2016;76(6):1578–90.
Agarwal S, et al. Deletion of the inhibitory co-receptor CTLA4 enhances and invigorates chimeric antigen receptor T cells. Immunity. 2023;56(10):2388-2407.e9.
Dötsch S, et al. Long-term persistence and functionality of adoptively transferred antigen-specific T cells with genetically ablated PD-1 expression. Proc Natl Acad Sci USA. 2023;120(10): e2200626120.
Yamamoto TN, et al. T cells genetically engineered to overcome death signaling enhance adoptive cancer immunotherapy. J Clin Investig. 2019;129(4):1551–65.
McKenzie C, et al. Novel Fas-TNFR chimeras that prevent Fas ligand-mediated kill and signal synergistically to enhance CAR T cell efficacy. Mol Ther Nucleic Acids. 2023;32:603–21.
Wang Q, et al. Role of tumor microenvironment in cancer progression and therapeutic strategy. Cancer Med. 2023;12(10):11149–65.
Chen J, et al. Target delivery of a PD-1-TREM2 scFv by CAR-T cells enhances anti-tumor efficacy in colorectal cancer. Mol Cancer. 2023;22(1):131.
Tian X, et al. Editorial: the role of epigenetic modification in MDSC differentiation and function. Front Immunol. 2023;14:1177138.
Lanitis E, et al. Mechanisms regulating T-cell infiltration and activity in solid tumors. Ann Oncol. 2017;28:xii18–32.
Hegde S, Leader AM, Merad M. MDSC: markers, development, states, and unaddressed complexity. Immunity. 2021;54(5):875–84.
Tumino N, et al. Polymorphonuclear myeloid-derived suppressor cells impair the anti-tumor efficacy of GD2.CAR T-cells in patients with neuroblastoma. J Hematol Oncol. 2021;14(1):191.
Charbonneau M-E, O’Riordan MXD. Reducing stress PERKs up anti-tumor immunity. Immunity. 2020;52(4):575–7.
Sun R, et al. Olaparib suppresses MDSC recruitment via SDF1α/CXCR4 axis to improve the anti-tumor efficacy of CAR-T cells on breast cancer in mice. Mol Ther J Am Soc Gene Ther. 2021;29(1):60–74.
Sun R, et al. CXCR4-modified CAR-T cells suppresses MDSCs recruitment via STAT3/NF-κB/SDF-1α axis to enhance efficacy against pancreatic cancer. Mol Ther J Am Soc Gene Ther. 2023;13(11):3193–209.
Ghonim MA, et al. Targeting PARP-1 with metronomic therapy modulates MDSC suppressive function and enhances anti-PD-1 immunotherapy in colon cancer. J Immunother Cancer. 2021;9(1): e001643.
Nalawade SA, et al. Selectively targeting myeloid-derived suppressor cells through TRAIL receptor 2 to enhance the efficacy of CAR T cell therapy for treatment of breast cancer. J Immunother Cancer. 2021;9(11): e003237.
Sun R, Sun Y, Wu C, et al. CXCR4-modified CAR-T cells suppresses MDSCs recruitment via STAT3/NF-κB/SDF-1α axis to enhance efficacy against pancreatic cancer. Mol Ther. 2023;31(11):3193–209.
Ray-Coquard I, et al. Olaparib plus bevacizumab first-line maintenance in ovarian cancer: final overall survival results from the PAOLA-1/ENGOT-ov25 trial. Ann Oncol Off J Eur Soc Med Oncol. 2023;34(8):681–92.
Yu EY, et al. Pembrolizumab plus olaparib in patients with metastatic castration-resistant prostate cancer: long-term results from the Phase 1b/2 KEYNOTE-365 Cohort A Study. Eur Urol. 2023;83(1):15–26.
Golan T, et al. Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N Engl J Med. 2019;381(4):317–27.
Chiu DK-C, et al. Hypoxia inducible factor HIF-1 promotes myeloid-derived suppressor cells accumulation through ENTPD2/CD39L1 in hepatocellular carcinoma. Nat Commun. 2017;8(1):517.
Godet I, et al. Fate-map** post-hypoxic tumor cells reveals a ROS-resistant phenotype that promotes metastasis. Nat Commun. 2019;10(1):4862.
Li H, et al. Antagonistic effects of p53 and HIF1A on microRNA-34a regulation of PPP1R11 and STAT3 and hypoxia-induced epithelial to mesenchymal transition in colorectal cancer cells. Gastroenterology. 2017;153(2):505–20.
Godet I, et al. Post-hypoxic cells promote metastatic recurrence after chemotherapy treatment in TNBC. Cancers. 2021;13(21):5509.
Liao Q, et al. Engineering T cells with hypoxia-inducible chimeric antigen receptor (HiCAR) for selective tumor killing. Biomarker Res. 2020;8(1):56.
He H, et al. Conditioned CAR-T cells by hypoxia-inducible transcription amplification (HiTA) system significantly enhances systemic safety and retains antitumor efficacy. J Immunother Cancer. 2021;9(10): e002755.
Kosti P, et al. Hypoxia-sensing CAR T cells provide safety and efficacy in treating solid tumors. Cell Rep Med. 2021;2(4): 100227.
Drenckhan A, et al. CAIX furthers tumour progression in the hypoxic tumour microenvironment of esophageal carcinoma and is a possible therapeutic target. J Enzyme Inhib Med Chem. 2018;33(1):1024–33.
Lock FE, et al. Targeting carbonic anhydrase IX depletes breast cancer stem cells within the hypoxic niche. Oncogene. 2013;32(44):5210–9.
Li H, et al. CAIX-specific CAR-T cells and sunitinib show synergistic effects against metastatic renal cancer models. J Immunother (Hagerstown Md 1997). 2020;43(1):16–28.
Cui J, et al. Targeting hypoxia downstream signaling protein, CAIX, for CAR T-cell therapy against glioblastoma. Neuro Oncology. 2019;21(11):1436–46.
Lamers CH, et al. Treatment of metastatic renal cell carcinoma with CAIX CAR-engineered T cells: clinical evaluation and management of on-target toxicity. Mol Ther J Am Soc Gene Ther. 2013;21(4):904–12.
Sebestyen Z, et al. Translating gammadelta (γδ) T cells and their receptors into cancer cell therapies. Nat Rev Drug Discov. 2020;19(3):169–84.
Chabab G, et al. Diversity of tumor-infiltrating, γδ T-cell abundance in solid cancers. Cells. 2020;9(6):1537.
Gentles AJ, et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat Med. 2015;21(8):938–45.
Capsomidis A, et al. Chimeric antigen receptor-engineered human gamma delta T cells: enhanced cytotoxicity with retention of cross presentation. Mol Ther J Am Soc Gene Ther. 2018;26(2):354–65.
Frieling JS, et al. γδ-Enriched CAR-T cell therapy for bone metastatic castrate-resistant prostate cancer. Sci Adv. 2023;9(18):eadf0108.
Fisher J, et al. Engineering γδT cells limits tonic signaling associated with chimeric antigen receptors. Sci Signal. 2019;12(598):eaax1872.
Morandi F, et al. Engineering the bridge between innate and adaptive immunity for cancer immunotherapy: focus on γδ T and NK cells. Cells. 2020;9(8):1757.
Ganapathy T, et al. CAR γδ T cells for cancer immunotherapy. Is the field more yellow than green? Cancer Immunol Immunother. 2023;72(2):277–86.
Klichinsky M, et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat Biotechnol. 2020;38(8):947–53.
Young RM, et al. Next-generation CAR T-cell therapies. Cancer Discov. 2022;12(7):1625–33.
Qian H, et al. Dual-aptamer-engineered M1 macrophage with enhanced specific targeting and checkpoint blocking for solid-tumor immunotherapy. Mol Ther J Am Soc Gene Ther. 2022;30(8):2817–27.
Kang M, et al. Nanocomplex-mediated in vivo programming to chimeric antigen receptor-M1 macrophages for cancer therapy. Adv Mater (Deerfield Beach Fla). 2021;33(43): e2103258.
Chen Y, et al. The application of HER2 and CD47 CAR-macrophage in ovarian cancer. J Transl Med. 2023;21(1):654.
Dong X, et al. Efficacy evaluation of chimeric antigen receptor-modified human peritoneal macrophages in the treatment of gastric cancer. Br J Cancer. 2023;129(3):551–62.
Dooling LJ, et al. Cooperative phagocytosis of solid tumours by macrophages triggers durable anti-tumour responses. Nat Biomed Eng. 2023;7(9):1081–96.
Wang S, et al. CAR-macrophage: an extensive immune enhancer to fight cancer. EBioMedicine. 2022;76: 103873.
Zhang L, et al. Pluripotent stem cell-derived CAR-macrophage cells with antigen-dependent anti-cancer cell functions. J Hematol Oncol. 2020;13(1):153.
Wang X, et al. Metabolic Reprogramming via ACOD1 depletion enhances function of human induced pluripotent stem cell-derived CAR-macrophages in solid tumors. Nat Commun. 2023;14(1):5778.
Yan T, Zhu L, Chen J. Current advances and challenges in CAR T-Cell therapy for solid tumors: tumor-associated antigens and the tumor microenvironment. Exp Hematol Oncol. 2023;12(1):14.
Li J, et al. Chimeric antigen receptor T cell (CAR-T) immunotherapy for solid tumors: lessons learned and strategies for moving forward. J Hematol Oncol. 2018;11(1):22.
Feng K-C, et al. Cocktail treatment with EGFR-specific and CD133-specific chimeric antigen receptor-modified T cells in a patient with advanced cholangiocarcinoma. J Hematol Oncol. 2017;10(1):4.
León-Triana O, et al. Dual-target CAR-Ts with on- and off-tumour activity may override immune suppression in solid cancers: a mathematical proof of concept. Cancers. 2021;13(4):703.
Shah NN, et al. Bispecific anti-CD20, anti-CD19 CAR T cells for relapsed B cell malignancies: a phase 1 dose escalation and expansion trial. Nat Med. 2020;26(10):1569–75.
Zurko JC, et al. Long-term outcomes and predictors of early response, late relapse, and survival for patients treated with bispecific LV20.19 CAR T-cells. Am J Hematol. 2022;97(12):1580–8.
Tong C, et al. Optimized tandem CD19/CD20 CAR-engineered T cells in refractory/relapsed B-cell lymphoma. Blood. 2020;136(14):1632–44.
** X, et al. CAR-T cells dual-target CD123 and NKG2DLs to eradicate AML cells and selectively target immunosuppressive cells. Oncoimmunology. 2023;12(1):2248826.
Kloss CC, et al. Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells. Nat Biotechnol. 2013;31(1):71–5.
Lanitis E, et al. Chimeric antigen receptor T cells with dissociated signaling domains exhibit focused antitumor activity with reduced potential for toxicity in vivo. Cancer Immunol Res. 2013;1(1):43–53.
Alvarez-Vallina L, Hawkins RE. Antigen-specific targeting of CD28-mediated T cell co-stimulation using chimeric single-chain antibody variable fragment-CD28 receptors. Eur J Immunol. 1996;26(10):2304–9.
Wilkie S, et al. Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J Clin Immunol. 2012;32(5):1059–70.
Roybal KT, et al. Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell. 2016;164(4):770–9.
Morsut L, et al. Engineering customized cell sensing and response behaviors using synthetic Notch receptors. Cell. 2016;164(4):780–91.
Hyrenius-Wittsten A, et al. SynNotch CAR circuits enhance solid tumor recognition and promote persistent antitumor activity in mouse models. Sci Transl Med. 2021;13(591):eabd8836.
Ruffo E, et al. Post-translational covalent assembly of CAR and synNotch receptors for programmable antigen targeting. Nat Commun. 2023;14(1):2463.
Allen ME, et al. An AND-gated drug and photoactivatable Cre-loxP system for spatiotemporal control in cell-based therapeutics. ACS Synth Biol. 2019;8(10):2359–71.
Kobayashi A, et al. Light-controllable binary switch activation of CAR T cells. ChemMedChem. 2022;17(12): e202100722.
Nguyen NT, et al. Nano-optogenetic engineering of CAR T cells for precision immunotherapy with enhanced safety. Nat Nanotechnol. 2021;16(12):1424–34.
Zhang B, et al. Photoswitchable CAR-T cell function in vitro and in vivo via a cleavable mediator. Cell Chem Biol. 2021;28(1):60-69.e7.
Huang Z, et al. Engineering light-controllable CAR T cells for cancer immunotherapy. Sci Adv. 2020;6(8):eaay9209.
Duplus-Bottin H, et al. A single-chain and fast-responding light-inducible Cre recombinase as a novel optogenetic switch. Elife. 2021;10: e61268.
Fedorov VD, Themeli M, Sadelain M. PD-1- and CTLA-4-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med. 2013;5(215):215ra172.
Cho JH, Collins JJ, Wong WW. Universal chimeric antigen receptors for multiplexed and logical control of T cell responses. Cell. 2018;173(6):1426-1438.e11.
Cho JH, et al. Engineering advanced logic and distributed computing in human CAR immune cells. Nat Commun. 2021;12(1):792.
Depil S, et al. ‘Off-the-shelf’ allogeneic CAR T cells: development and challenges. Nat Rev Drug Discov. 2020;19(3):185–99.
Cripe TP, et al. Leveraging gene therapy to achieve long-term continuous or controllable expression of biotherapeutics. Sci Adv. 2022;8(28):eabm1890.
Liu X, et al. Novel T cells with improved in vivo anti-tumor activity generated by RNA electroporation. Protein Cell. 2017;8(7):514–26.
Funding
This work was supported by the National Natural Science Foundation of China (31870899 and 32070899 to X.Z.).
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, G., Ye, M., Zhang, Y. et al. Challenges and strategies in relation to effective CAR-T cell immunotherapy for solid tumors. Med Oncol 41, 126 (2024). https://doi.org/10.1007/s12032-024-02310-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-024-02310-y