Abstract
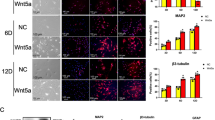
Transplantation of oligodendrocyte progenitor cell (OPC) at the injury site is being developed as a potential therapeutic strategy for promoting remyelination and locomotor function recovery after spinal cord injury (SCI). To this end, the development of expandable and functional human OPCs is crucial for testing their efficacy in SCI. In mice and rats, the endocannabinoid signaling system is crucial for the survival, differentiation, and maturation of OPCs. Similar studies in humans are lacking currently. Endocannabinoids and exogenous cannabinoids exert their effects mainly via cannabinoid receptors (CB1R and CB2R). We demonstrated that these receptors were differentially expressed in iPSC-derived human NSCs and OPCs, and they could be activated by WIN55212-2 (WIN), a potent CB1R/CB2R agonist to upregulate the endocannabinoid signaling during glial induction. WIN primed NSCs generated more OLIG2 + glial progenitors and migratory PDGFRα + OPC in a CB1/CB2 dependent manner compared to unprimed NSCs. Furthermore, WIN-induced OPCs (WIN-OPCs) robustly differentiated into functional oligodendrocytes and myelinate in vitro and in vivo in a mouse spinal cord injury model. RNA-Seq revealed that WIN upregulated the biological process of positive regulation of oligodendrocyte differentiation. Mechanistically, WIN could act as a partial smoothed (SMO) inhibitor or activate CB1/CB2 to form heteromeric complexes with SMO leading to the inhibition of GLI1 in the Sonic hedgehog pathway. The partial and temporal inhibition of GLI1 during glial induction is shown to promote OPCs that differentiate faster than control’s. Thus, CB1R/CB2R activation results in more efficient generation of OPCs that can mature and efficiently myelinate.
Graphical abstract









Similar content being viewed by others
Data Availability
The data are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Ilyasov, A. A., Milligan, C. E., Pharr, E. P., & Howlett, A. C. (2018). The Endocannabinoid System and Oligodendrocytes in Health and Disease. Frontiers in Neuroscience, 12, 733.
McDonald, J. W., & Belegu, V. (2006). Demyelination and remyelination after spinal cord injury. Journal of Neurotrauma, 23(3–4), 345–359.
Singh, A., Tetreault, L., Kalsi-Ryan, S., Nouri, A., & Fehlings, M. G. (2014). Global prevalence and incidence of traumatic spinal cord injury. Clinical Epidemiology, 6, 309–331.
Kerr, C. L., Letzen, B. S., Hill, C. M., Agrawal, G., Thakor, N. V., Sterneckert, J. L., et al. (2010). Efficient Differentiation of Human Embryonic Stem Cells into Oligodendrocyte Progenitors for Application in a Rat Contusion Model of Spinal Cord Injury. International Journal of Neuroscience, 120(4), 305–313.
Yao, Z. F., Wang Y., Lin Y. H., Wu Y., Zhu A. Y., Wang R., et al. (2017). Transplantation of PDGF-AA-Overexpressing Oligodendrocyte Precursor Cells Promotes Recovery in Rat Following Spinal Cord Injury. Frontiers in Cellular Neuroscience, 11.
All, A. H., Gharibani P., Gupta S., Bazley F. A., Pashai N., Chou B. K., et al. (2015). Early Intervention for Spinal Cord Injury with Human Induced Pluripotent Stem Cells Oligodendrocyte Progenitors. PLoS One, 10(1).
Kawabata, S., Takano, M., Numasawa-Kuroiwa, Y., Itakura, G., Kobayashi, Y., Nishiyama, Y., et al. (2016). Grafted Human iPS Cell-Derived Oligodendrocyte Precursor Cells Contribute to Robust Remyelination of Demyelinated Axons after Spinal Cord Injury. Stem Cell Reports, 6(1), 1–8.
Kim, D. S., Jung S. J., Lee J. S., Lim B. Y., Kim H. A., Yoo J. E., et al. (2017). Rapid generation of OPC-like cells from human pluripotent stem cells for treating spinal cord injury. Experimental and Molecular Medicine, 49.
Zheng, W., Li, Q., Zhao, C., Da, Y., Zhang, H. L., & Chen, Z. (2018). Differentiation of Glial Cells From hiPSCs: Potential Applications in Neurological Diseases and Cell Replacement Therapy. Frontiers in Cellular Neuroscience, 12, 239.
Li, P., Li, M., Tang, X., Wang, S., Zhang, Y. A., & Chen, Z. (2016). Accelerated generation of oligodendrocyte progenitor cells from human induced pluripotent stem cells by forced expression of Sox10 and Olig2. Science China Life Sciences, 59(11), 1131–1138.
Ehrlich, M., Mozafari, S., Glatza, M., Starost, L., Velychko, S., Hallmann, A. L., et al. (2017). Rapid and efficient generation of oligodendrocytes from human induced pluripotent stem cells using transcription factors. Proceedings of the National Academy of Sciences of the United States of America, 114(11), E2243–E2252.
Garcia-Leon, J. A., Kumar, M., Boon, R., Chau, D., One, J., Wolfs, E., et al. (2018). SOX10 Single Transcription Factor-Based Fast and Efficient Generation of Oligodendrocytes from Human Pluripotent Stem Cells. Stem Cell Reports, 10(2), 655–672.
Garcia-Leon, J. A., Garcia-Diaz B., Eggermont K., Caceres-Palomo L., Neyrinck K., da Costa R. M., et al. (2020). Generation of oligodendrocytes and establishment of an all-human myelinating platform from human pluripotent stem cells. Nature Protocols, 15(11).
Namchaiw, P., Wen H., Mayrhofer F., Chechneva O., Biswas S., Deng W. B. (2019). Temporal and partial inhibition of GLI1 in neural stem cells (NSCs) results in the early maturation of NSC derived oligodendrocytes in vitro. Stem Cell Research & Therapy, 10(1).
Lu, H. C., & Mackie, K. (2021). Review of the Endocannabinoid System. Biological Psychiatry-Cognitive Neuroscience and Neuroimaging, 6(6), 607–615.
Garcia-Arencibia, M., Molina-Holgado, E., & Molina-Holgado, F. (2019). Effect of endocannabinoid signalling on cell fate: Life, death, differentiation and proliferation of brain cells. British Journal of Pharmacology, 176(10), 1361–1369.
Sun, J., Fang, Y. Q., Ren, H., Chen, T., Guo, J. J., Yan, J., et al. (2013). WIN55,212–2 protects oligodendrocyte precursor cells in stroke penumbra following permanent focal cerebral ischemia in rats. Acta Pharmacologica Sinica, 34(1), 119–128.
Sun, J., Fang, Y. Q., Chen, T., Guo, J. J., Yan, J., Song, S., et al. (2013). WIN55, 212–2 promotes differentiation of oligodendrocyte precursor cells and improve remyelination through regulation of the phosphorylation level of the ERK 1/2 via cannabinoid receptor 1 after stroke-induced demyelination. Brain Research, 1491, 225–235.
Fernandez-Lopez, D., Pradillo, J. M., Garcia-Yebenes, I., Martinez-Orgado, J. A., Moro, M. A., & Lizasoain, I. (2010). The Cannabinoid WIN55212-2 Promotes Neural Repair After Neonatal Hypoxia-Ischemia. Stroke, 41(12), 2956–2964.
Bortolato, M., Bini, V., Frau, R., Devoto, P., Pardu, A., Fan, Y. J., et al. (2014). Juvenile cannabinoid treatment induces frontostriatal gliogenesis in Lewis rats. European Neuropsychopharmacology, 24(6), 974–985.
Tomas-Roig, J., Wirths, O., Salinas-Riester, G., & Havemann-Reinecke, U. (2016). The Cannabinoid CB1/CB2 Agonist WIN55212.2 Promotes Oligodendrocyte Differentiation In Vitro and Neuroprotection During the Cuprizone-Induced Central Nervous System Demyelination. Cns Neuroscience & Therapeutics, 22(5), 387–95.
Galligan, J. J. (2009). Cannabinoid signalling in the enteric nervous system. Neurogastroenterology and Motility, 21(9), 899–902.
Castillo, P. E., Younts, T. J., Chavez, A. E., & Hashimotodani, Y. (2012). Endocannabinoid Signaling and Synaptic Function. Neuron, 76(1), 70–81.
Galve-Roperh, I., Chiurchiu, V., Diaz-Alonso, J., Bari, M., Guzman, M., & Maccarrone, M. (2013). Cannabinoid receptor signaling in progenitor/stem cell proliferation and differentiation. Progress in Lipid Research, 52(4), 633–650.
Mato, S., Alberdi, E., Ledent, C., Watanabe, M., & Matute, C. (2009). CB1 Cannabinoid Receptor-Dependent and -Independent Inhibition of Depolarization-Induced Calcium Influx in Oligodendrocytes. Glia, 57(3), 295–306.
Arevalo-Martin, A., Garcia-Ovejero, D., Rubio-Araiz, A., Gomez, O., Molina-Holgado, F., & Molina-Holgado, E. (2007). Cannabinoids modulate Olig2 and polysialylated neural cell adhesion molecule expression in the subventricular zone of post-natal rats through cannabinoid receptor 1 and cannabinoid receptor 2. European Journal of Neuroscience, 26(6), 1548–1559.
Xapelli, S., Agasse F., Grade S., Bernardino L., Ribeiro F. F., Schitine C. S., et al. (2014). Modulation of subventricular zone oligodendrogenesis: a role for hemopressin? Frontiers in Cellular Neuroscience, 8.
Amankulor, N. M., Hambardzumyan, D., Pyonteck, S. M., Becher, O. J., Joyce, J. A., & Holland, E. C. (2009). Sonic Hedgehog Pathway Activation Is Induced by Acute Brain Injury and Regulated by Injury-Related Inflammation. Journal of Neuroscience, 29(33), 10299–10308.
Takebayashi, H., Nabeshima, Y., Yoshida, S., Chisaka, O., Ikenaka, K., & Nabeshima, Y. (2002). The basic helix-loop-helix factor Olig2 is essential for the development of motoneuron and oligodendrocyte lineages. Current Biology, 12(13), 1157–1163.
Samanta, J., Grund, E. M., Silva, H. M., Lafaille, J. J., Fishell, G., & Salzer, J. L. (2015). Inhibition of Gli1 mobilizes endogenous neural stem cells for remyelination. Nature, 526(7573), 448.
Douvaras, P., & Fossati, V. (2015). Generation and isolation of oligodendrocyte progenitor cells from human pluripotent stem cells. Nature Protocols, 10(8), 1143–1154.
Schoepf, C. L., Zeidler M., Spiecker L., Kern G., Lechner J., Kummer K. K., et al. (2020). Selected Ionotropic Receptors and Voltage-Gated Ion Channels: More Functional Competence for Human Induced Pluripotent Stem Cell (iPSC)-Derived Nociceptors. Brain Sciences, 10(6).
Kim, H. S., Kim J. Y., Song C. L., Jeong J. E., Cho Y. S. (2020). Directly induced human Schwann cell precursors as a valuable source of Schwann cells. Stem Cell Research & Therapy, 11(1).
Basso, D. M., Fisher, L. C., Anderson, A. J., Jakeman, L. B., McTigue, D. M., & Popovich, P. G. (2006). Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. Journal of Neurotrauma, 23(5), 635–659.
Zou, S., Kumar U. (2018). Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. International Journal of Molecular Sciences, 19(3).
Molina-Holgado, E., Vela, J. M., Arevalo-Martin, A., Almazan, G., Molina-Holgado, F., Borrell, J., et al. (2002). Cannabinoids promote oligodendrocyte progenitor survival: Involvement of cannabinoid receptors and phosphatidylinositol-3 kinase/Akt signaling. Journal of Neuroscience, 22(22), 9742–9753.
Rodrigues, R. S., Lourenco D. M., Paulo S. L., Mateus J. M., Ferreira M. F., Mouro F. M., et al. (2019). Cannabinoid Actions on Neural Stem Cells: Implications for Pathophysiology. Molecules, 24(7).
Wegener, A., Deboux, C., Bachelin, C., Frah, M., Kerninon, C., Seilhean, D., et al. (2015). Gain of Olig2 function in oligodendrocyte progenitors promotes remyelination. Brain, 138(Pt 1), 120–135.
Ferent, J., Zimmer, C., Durbec, P., Ruat, M., & Traiffort, E. (2013). Sonic Hedgehog Signaling Is a Positive Oligodendrocyte Regulator during Demyelination. Journal of Neuroscience, 33(5), 1759–1772.
Sanchez, M. A., & Armstrong, R. C. (2018). Postnatal Sonic hedgehog (Shh) responsive cells give rise to oligodendrocyte lineage cells during myelination and in adulthood contribute to remyelination. Experimental Neurology, 299, 122–136.
Hashimoto, H., Jiang, W., Yoshimura, T., Moon, K. H., Bok, J., & Ikenaka, K. (2018). Strong sonic hedgehog signaling in the mouse ventral spinal cord is not required for oligodendrocyte precursor cell (OPC) generation but is necessary for correct timing of its generation. Neurochemistry International, 119, 178–183.
Fish, E. W., Murdaugh, L. B., Zhang, C., Boschen, K. E., Boa-Amponsem, O., Mendoza-Romero, H. N., et al. (2019). Cannabinoids Exacerbate Alcohol Teratogenesis by a CB1-Hedgehog Interaction. Scientific Reports, 9(1), 16057.
Lo, H. F., Hong, M., & Krauss, R. S. (2021). Concepts in Multifactorial Etiology of Developmental Disorders: Gene-Gene and Gene-Environment Interactions in Holoprosencephaly. Frontiers in Cell and Developmental Biology, 9, 795194.
Fuxe, K., Borroto-Escuela, D. O., Marcellino, D., Romero-Fernandez, W., Frankowska, M., Guidolin, D., et al. (2012). GPCR Heteromers and their Allosteric Receptor-Receptor Interactions. Current Medicinal Chemistry, 19(3), 356–363.
Khaliullina, H., Bilgin, M., Sampaio, J. L., Shevchenko, A., & Eaton, S. (2015). Endocannabinoids are conserved inhibitors of the Hedgehog pathway. Proceedings of the National Academy of Sciences of the United States of America, 112(11), 3415–3420.
Forli, S., Huey, R., Pique, M. E., Sanner, M. F., Goodsell, D. S., & Olson, A. J. (2016). Computational protein-ligand docking and virtual drug screening with the AutoDock suite. Nature Protocols, 11(5), 905–919.
Rahman, M. M., Hazan, A., Selway, J. L., Herath, D. S., Harwood, C. A., Pirzado, M. S., et al. (2018). A Novel Mechanism for Activation of GLI1 by Nuclear SMO That Escapes Anti-SMO Inhibitors. Cancer Research, 78(10), 2577–2588.
Kitada, M., & Rowitch, D. H. (2006). Transcription factor co-expression patterns indicate heterogeneity of oligodendroglial subpopulations in adult spinal cord. Glia, 54(1), 35–46.
Sim, F. J., McClain, C. R., Schanz, S. J., Protack, T. L., Windrem, M. S., & Goldman, S. A. (2011). CD140a identifies a population of highly myelinogenic, migration-competent and efficiently engrafting human oligodendrocyte progenitor cells. Nature Biotechnology, 29(10), 934-U123.
Gomez, O., Sanchez-Rodriguez, A., Le, M. Q. U., Sanchez-Caro, C., Molina-Holgado, F., & Molina-Holgado, E. (2011). Cannabinoid receptor agonists modulate oligodendrocyte differentiation by activating PI3K/Akt and the mammalian target of rapamycin (mTOR) pathways. British Journal of Pharmacology, 163(7), 1520–1532.
Bibollet-Bahena, O., & Almazan, G. (2009). IGF-1-stimulated protein synthesis in oligodendrocyte progenitors requires PI3K/mTOR/Akt and MEK/ERK pathways. Journal of Neurochemistry, 109(5), 1440–1451.
Acknowledgements
Authors acknowledge Prof. Xusheng Wang from School of Pharmaceutical Science (Shenzhen), Sun Yat-sen University, Shenzhen, China, for kindly providing the hiPSC cell line, and Dr. **aowei Li from **an University, Guangzhou, China, for guidance and help on the spinal cord injury mice models. Hong Gao would like to thank CSC scholarship for providing the opportunity to study at the University of California, Davis.
Funding
This work was funded by National Natural Science Foundation of China (Grant no. 81772449 and 81971081) and Shenzhen Science and Technology Program (Grant no. KQTD20190929173853397). We would also like to thank the partial support of Jiangxi ShuanQian Team Project #S2019CXTD2090 to Jiangxi Deshang Pharmaceutical Co., Ltd.
Author information
Authors and Affiliations
Contributions
H.G., Y.G., S.B. and Deng.W.B. designed experiments and worked on the manuscript writing. H.G., J.L. and Zhang. H.J. performed cell culture, qPCR, immunostaining and MACS. H.G., Zhang. H.J. and Chen. Z.L. conducted flow cytometry, SCI mice modeling, cell transplantation and TEM. H.G., J.L. and Y.G. performed RNA-Seq analysis and data interpreting. The final version of manuscript was reviewed and approved by all authors.
Corresponding author
Ethics declarations
Ethics Approval
All surgeries and animal experiments were performed with the approval of the Institutional Animal Care and Use Committee of Sun Yat-sen University (SYSU-IACUC-2021–000519), Guangzhou, China.
Consent to Participate
Not applicable.
Consent for Publication
All authors reviewed and approved the final version of the manuscript.
Conflict of Interest
All authors declared there was no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Fig. TEM-1
(PNG 3366 kb)

Fig. TEM-2
(PNG 3399 kb)
Rights and permissions
About this article
Cite this article
Gao, H., Guo, Y., Biswas, S. et al. Promoting Oligodendrocyte Differentiation from Human Induced Pluripotent Stem Cells by Activating Endocannabinoid Signaling for Treating Spinal Cord Injury. Stem Cell Rev and Rep 18, 3033–3049 (2022). https://doi.org/10.1007/s12015-022-10405-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-022-10405-0