Abstract
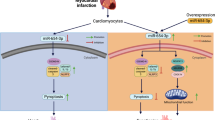
Elevated level of homocysteine (Hcy) called hyperhomocysteinemia (HHcy) is one of the major risk factors for chronic heart failure. Although the role of Hcy in cardiac remodeling is documented, the regulatory mechanism involved therein is still nebulous. MicroRNAs (miRNAs) and dicer have been implicated in regulation of cardiovascular diseases. Dicer is the only known enzyme involved in miRNA maturation. We investigated the involvement of dicer and miRNA in Hcy-induced cardiac remodeling. HL-1 cardiomyocytes were cultured in different doses of Hcy. Total RNA was isolated and RT-PCR and real-time PCR was performed for dicer, MMP-2,-9, TIMP-1,-3, and NOX-4. MiRNA microarray was used for analyzing the differential expression of miRNAs. Individual miRNA assay was also done. Western blotting was used to assess the MMP-9 expression in HHcy cardiomyocytes. The RT-PCR results suggest that dicer expression is enhanced in HHcy cardiomyocytes suggesting its involvement in cardiac remodeling caused due to high dose of Hcy. On the other hand, high dose of Hcy increased NOX-4 expression, a marker for oxidative stress. Additionally, HHcy cardiomyocytes showed elevated levels of MMP-2,-9 and TIMP-1,-3, and reduced expression of TIMP-4, suggesting cardiac remodeling due to oxidative stress. The miRNA microarray assay revealed differential expression of 11 miRNAs and among them miR-188 show dramatic downregulation. These findings suggest that dicer and miRNAs especially miR-188 are involved in Hcy-induced cardiac remodeling.









Similar content being viewed by others
Abbreviations
- Hcy :
-
Homocysteine
- HHcy:
-
Hyperhomocysteinemia
- MMP:
-
Matrix metalloproteinase
- TIMP:
-
Tissue inhibitor of metalloproteinase
- NOX:
-
Nicotinamide adenine diphosphate oxidase
- CHF:
-
Congestive heart failure
- CVD:
-
cardiovascular diseases
- miR/miRNA:
-
microRNA
- RT-PCR:
-
Reverse transcription polymerase chain reaction
References
Herrmann, W., Herrmann, M., & Obeid, R. (2007). Hyperhomocysteinaemia: A critical review of old and new aspects. Current Drug Metabolism, 8(1), 17–31.
Martin-Herrero, F., Jimenez-Candil, J., Martin-Moreiras, J., Pabon, P., Cruz-Gonzalez, I., Martin-Garcia, A., et al. (2008). Homocysteine, cause or consequence? International Journal of Cardiology, 129(2), 276–277.
Refsum, H., Smith, A. D., Ueland, P. M., Nexo, E., Clarke, R., McPartlin, J., et al. (2004). Facts and recommendations about total homocysteine determinations: An expert opinion. Clinical Chemistry, 50(1), 3–32.
Wierzbicki, A. S. (2007). Homocysteine and cardiovascular disease: A review of the evidence. Diabetes & Vascular Disease Research, 4(2), 143–150.
Jhee, K. H., & Kruger, W. D. (2005). The role of cystathionine beta-synthase in homocysteine metabolism. Antioxidants Redox Signaling, 7(5–6), 813–822.
Graham, I. M., Daly, L. E., Refsum, H. M., Robinson, K., Brattstrom, L. E., Ueland, P. M., et al. (1997). Plasma homocysteine as a risk factor for vascular disease. The European Concerted Action Project. JAMA, 277(22), 1775–1781.
Blacher, J., Demuth, K., Guerin, A. P., Vadez, C., Moatti, N., Safar, M. E., et al. (1999). Association between plasma homocysteine concentrations and cardiac hypertrophy in end-stage renal disease. Journal of Nephrology, 12(4), 248–255.
Blacher, J., & Safar, M. E. (2001). Homocysteine, folic acid, B vitamins and cardiovascular risk. Journal of Nutrition, Health & Aging, 5(3), 196–199.
Lee, R. T. (2001). Matrix metalloproteinase inhibition and the prevention of heart failure. Trends in Cardiovascular Medicine, 11(5), 202–205.
Sakata, Y., Yamamoto, K., Mano, T., Nishikawa, N., Yoshida, J., Hori, M., et al. (2004). Activation of matrix metalloproteinases precedes left ventricular remodeling in hypertensive heart failure rats: Its inhibition as a primary effect of angiotensin-converting enzyme inhibitor. Circulation, 109(17), 2143–2149.
Spinale, F. G., Coker, M. L., Bond, B. R., & Zellner, J. L. (2000). Myocardial matrix degradation and metalloproteinase activation in the failing heart: A potential therapeutic target. Cardiovascular Research, 46(2), 225–238.
Tyagi, S. C., Smiley, L. M., Mujumdar, V. S., Clonts, B., & Parker, J. L. (1998). Reduction-oxidation (Redox) and vascular tissue level of homocyst(e)ine in human coronary atherosclerotic lesions and role in extracellular matrix remodeling and vascular tone. Molecular and Cellular Biochemistry, 181(1–2), 107–116.
Lominadze, D., Roberts, A. M., Tyagi, N., Moshal, K. S., & Tyagi, S. C. (2006). Homocysteine causes cerebrovascular leakage in mice. American Journal of Physiology. Heart and Circulatory Physiology, 290(3), H1206–H1213.
Moshal, K. S., Sen, U., Tyagi, N., Henderson, B., Steed, M., Ovechkin, A. V., et al. (2006). Regulation of homocysteine-induced MMP-9 by ERK1/2 pathway. American Journal of Physiology. Cell Physiology, 290(3), C883–C891.
Moshal, K. S., Tipparaju, S. M., Vacek, T. P., Kumar, M., Singh, M., Frank, I. E., et al. (2008). Mitochondrial matrix metalloproteinase activation decreases myocyte contractility in hyperhomocysteinemia. American Journal of Physiology. Heart and Circulatory Physiology, 295(2), H890–H897.
Tyagi, S. C., Rodriguez, W., Patel, A. M., Roberts, A. M., Falcone, J. C., Passmore, J. C., et al. (2005). Hyperhomocysteinemic diabetic cardiomyopathy: Oxidative stress, remodeling, and endothelial-myocyte uncoupling. Journal of Cardiovascular Pharmacology and Therapeutics, 10(1), 1–10.
Cheng, Y., Ji, R., Yue, J., Yang, J., Liu, X., Chen, H., et al. (2007). MicroRNAs are aberrantly expressed in hypertrophic heart: Do they play a role in cardiac hypertrophy? American Journal of Pathology, 170(6), 1831–1840.
Mishra, P. K., Tyagi, N., Kumar, M., & Tyagi, S. C. (2009). MicroRNAs as a therapeutic target for cardiovascular diseases. Journal of Cellular and Molecular Medicine, 13(4), 778–789.
Sayed, D., Hong, C., Chen, I. Y., Lypowy, J., & Abdellatif, M. (2007). MicroRNAs play an essential role in the development of cardiac hypertrophy. Circulation Research, 100(3), 416–424.
Tatsuguchi, M., Seok, H. Y., Callis, T. E., Thomson, J. M., Chen, J. F., Newman, M., et al. (2007). Expression of microRNAs is dynamically regulated during cardiomyocyte hypertrophy. Journal of Molecular and Cellular Cardiology, 42(6), 1137–1141.
Thum, T., Catalucci, D., & Bauersachs, J. (2008). MicroRNAs: Novel regulators in cardiac development and disease. Cardiovascular Research, 79(4), 562–570.
Van Rooij, E., & Olson, E. N. (2007). MicroRNAs: Powerful new regulators of heart disease and provocative therapeutic targets. Journal of Clinical Investigation, 117(9), 2369–2376.
Yang, W. J., Yang, D. D., Na, S., Sandusky, G. E., Zhang, Q., & Zhao, G. (2005). Dicer is required for embryonic angiogenesis during mouse development. Journal of Biological Chemistry, 280(10), 9330–9335.
Bartel, D. P. (2009). MicroRNAs: Target recognition and regulatory functions. Cell, 136(2), 215–233.
Latronico, M. V., Catalucci, D., & Condorelli, G. (2007). Emerging role of microRNAs in cardiovascular biology. Circulation Research, 101(12), 1225–1236.
Lai, E. C. (2002). Micro RNAs are complementary to 3’ UTR sequence motifs that mediate negative post-transcriptional regulation. Nature Genetics, 30(4), 363–364.
Borchert, G. M., Lanier, W., & Davidson, B. L. (2006). RNA polymerase III transcribes human microRNAs. Nature Structural & Molecular Biology, 13(12), 1097–1101.
Zhang, C. (2008). MicroRNAs: Role in cardiovascular biology and disease. Clinical Science, 114(12), 699–706.
Ruby, J. G., Jan, C. H., & Bartel, D. P. (2007). Intronic microRNA precursors that bypass Drosha processing. Nature, 448(7149), 83–86.
Berezikov, E., Guryev, V., van de Belt, J., Wienholds, E., Plasterk, R. H., & Cuppen, E. (2005). Phylogenetic shadowing and computational identification of human microRNA genes. Cell, 120(1), 21–24.
Zhang, C. (2008). MicroRNomics: A newly emerging approach for disease biology. Physiological Genomics, 33(2), 139–147.
Bernstein, E., Kim, S. Y., Carmell, M. A., Murchison, E. P., Alcorn, H., Li, M. Z., et al. (2003). Dicer is essential for mouse development. Nature Genetics, 35(3), 215–217.
Murchison, E. P., Stein, P., Xuan, Z., Pan, H., Zhang, M. Q., Schultz, R. M., et al. (2007). Critical roles for Dicer in the female germline. Genes and Development, 21(6), 682–693.
Lynn, F. C., Skewes-Cox, P., Kosaka, Y., McManus, M. T., Harfe, B. D., & German, M. S. (2007). MicroRNA expression is required for pancreatic islet cell genesis in the mouse. Diabetes, 56(12), 2938–2945.
Koralov, S. B., Muljo, S. A., Galler, G. R., Krek, A., Chakraborty, T., Kanellopoulou, C., et al. (2008). Dicer ablation affects antibody diversity and cell survival in the B lymphocyte lineage. Cell, 132(5), 860–874.
Davis, T. H., Cuellar, T. L., Koch, S. M., Barker, A. J., Harfe, B. D., McManus, M. T., et al. (2008). Conditional loss of Dicer disrupts cellular and tissue morphogenesis in the cortex and hippocampus. Journal of Neuroscience, 28(17), 4322–4330.
Kuehbacher, A., Urbich, C., Zeiher, A. M., & Dimmeler, S. (2007). Role of Dicer and Drosha for endothelial microRNA expression and angiogenesis. Circulation Research, 101(1), 59–68.
Zhao, Y., Ransom, J. F., Li, A., Vedantham, V., von Drehle, M., Muth, A. N., et al. (2007). Dysregulation of cardiogenesis, cardiac conduction, and cell cycle in mice lacking miRNA-1-2. Cell, 129(2), 303–317.
Suarez, Y., Fernandez-Hernando, C., Pober, J. S., & Sessa, W. C. (2007). Dicer dependent microRNAs regulate gene expression and functions in human endothelial cells. Circulation Research, 100(8), 1164–1173.
Chen, J. F., Murchison, E. P., Tang, R., Callis, T. E., Tatsuguchi, M., Deng, Z., et al. (2008). Targeted deletion of Dicer in the heart leads to dilated cardiomyopathy and heart failure. Proceedings of the National Academy of Science of the United States of America, 105(6), 2111–2116.
de Ruijter, W., Westendorp, R. G., Assendelft, W. J., den Elzen, W. P., de Craen, A. J., le Cessie, S., et al. (2009). Use of Framingham risk score, new biomarkers to predict cardiovascular mortality in older people: Population based observational cohort study. BMJ, 338, a3083.
Kundu, S., Kumar, M., Sen, U., Mishra, P. K., Tyagi, N., Metreveli, N., et al. (2009). Nitrotyrosinylation, remodeling and endothelial-myocyte uncoupling in iNOS, cystathionine beta synthase (CBS) knockouts and iNOS/CBS double knockout mice. Journal of Cellular Biochemistry, 106(1), 119–126.
Tyagi, S. C., & Hoit, B. D. (2002). Metalloproteinase in myocardial adaptation and maladaptation. Journal of Cardiovascular Pharmacology and Therapeutics, 7(4), 241–246.
Hsu, C. P., Huang, C. Y., Wang, J. S., Sun, P. C., & Shih, C. C. (2008). Extracellular matrix remodeling attenuated after experimental postinfarct left ventricular aneurysm repair. Annals of Thoracic Surgery, 86(4), 1243–1249.
Claycomb, W. C., Lanson, N. A., Jr., Stallworth, B. S., Egeland, D. B., Delcarpio, J. B., Bahinski, A., et al. (1998). HL-1 cells: A cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proceedings of the National Academy of Science of the United States of America, 95(6), 2979–2984.
Tyagi, S. C. (1999). Homocysteine and heart disease: Pathophysiology of extracellular matrix. Clinical and Experimental Hypertension, 21(3), 181–198.
Acknowledgment
A part of the study was supported by NIH grants HL 71010, HL-74185, and HL-88012.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishra, P.K., Tyagi, N., Kundu, S. et al. MicroRNAs Are Involved in Homocysteine-Induced Cardiac Remodeling. Cell Biochem Biophys 55, 153–162 (2009). https://doi.org/10.1007/s12013-009-9063-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-009-9063-6