Abstract
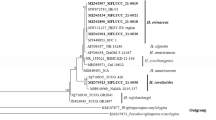
A bacterial strain PB1 with antagonistic activity against pathogenic fungi was isolated from marine soil and was identified as Paenibacillus elgii based on phenotypic and genotypic characterization. The isolate showed good antifungal activity against “Aspergillus niger (MTCC 282), Trichophyton rubrum (MTCC 791), Microsporum gypseum (MTCC 2819), Candida albicans (MTCC 227), and Saccharomyces cerevisiae (MTCC 170)”. Chitinase and beta 1, 4-endoglucanase are known for their capability to degrade fungal cell wall, thus we analyzed its productivity in PB1 strain using Plackett-Burman and Central Composite Design. The factors that affect the productivity of chitinase and beta 1, 4-endoglucanase were identified and optimized. A 7.77-fold increase (3.157 to 24.53 ± 1.33 U/mL) in chitinase and 7.422-fold increase (6.476 to 48.066 ± 0.676 U/mL) in beta 1, 4-endoglucanase versus basal medium was achieved. Chitinase and beta 1, 4-endoglucanase produced by Paenibacillus elgii strain PB1 represents the new source for biotechnological, medical, and agricultural applications.




Similar content being viewed by others
References
Abdel-Naby, M. A., El-Shayeb, N. M., & Sherief, A. A. (1992). Purification and some properties of chitinase from Aspergillus carneus. Applied Biochemistry and Biotechnology, 37(2), 141–154.
Anuradha, V., & Revathi, K. (2013). Purification and characterization of bacterial chitinase isolated from crustacean shells. International Journal of Pure and Applied Bioscience, 1(4), 1–11.
Aounallah, M. A., Slimene-Debez, I. B., Djebali, K., Gharbi, D., Hammami, M., Azaiez, S., Limam, F., & Tabbene, O. (2017). Enhancement of exochitinase production by Bacillus licheniformis AT6 strain and improvement of N-acetylglucosamine production. Applied Biochemistry and Biotechnology, 181(2), 650–666.
Bhat. (2000). Cellulases and related enzymes in biotechnology. Biotechnology Advances, 18(5), 355–383.
Brzezinska, M. S., Jankiewicz, U., Burkowska, A., & Walczak, M. (2014). Chitinolytic microorganisms and their possible application in environmental protection. Current Microbiology, 68(1), 71–81.
Budi, S. W., Van Tuinen, D., Arnould, C., Dumas-Gaudot, E., Gianinazzi-Pearson, V., & Gianinazzi, S. (2010). Hydrolytic enzyme activity of Paenibacillus sp. strain B2 and effects of the antagonistic bacterium on cell integrity of two soil-borne pathogenic fungi. Applied Soil Ecology, 15(2), 191–199.
Chang, W. T., Chen, M. L., & Wang, S. L. (2010). An antifungal chitinase produced by Bacillus subtilis using chitin waste as a carbon source. World Journal of Microbiology and Biotechnology, 26(5), 945–950.
Chen, H. B., Kao, P. M., Huang, H. C., Shieh, C. J., Chen, C. I., & Liu, Y. C. (2010). Effects of using various bioreactors on chitinolytic enzymes production by Paenibacillus taichungensis. Biochemical Engineering Journal, 49(3), 337–342.
Chrisnasari, R., Yasaputera, S., Christianto, P., Santoso, V. I., & Pantjajani, T. (2016). Production and characterization of chitinases from thermophilic bacteria isolated from Prataan hot spring, East Java. Journal of Mathematical and Fundamental Sciences, 48(2), 149–163.
Cruz, J., Hidalgo-Gallego, A., Lora, J. M., Benitez, T., Pintor-Toro, J. A., & Lobell, A. (1992). Isolation and characterization of three chitinases from Trichoderma harzianum. European Journal of Biochemistry, 206(3), 859–867.
Das, S. N., Neeraja, C., Sarma, P. V., Prakash, J. M., Purushotham, P., Kaur, M., Dutta, S., & Podile, A. R. (2012). Microbial chitinases for chitin waste management. In Microorganisms in environment management (pp. 135–150). Dordrechet: Springer.
De Nicola, R., Hall, N., Melville, S. G., & Walker, G. M. (2009). Influence of zinc on distiller’s yeast: cellular accumulation of zinc and impact on spirit congeners. Journal of the Institute of Brewing, 115(3), 265–271.
Ferreira, S., Duarte, A. P., Ribeiro, M. H., Queiroz, J. A., & Domingues, F. C. (2009). Response surface optimization of enzymatic hydrolysis of Cistus ladanifer and Cytisus striatus for bioethanol production. Biochemical Engineering Journal, 45(3), 192–200.
Ghose. (1987). Measurement of cellulase activities. Pure and Applied Chemistry, 59(2), 57–268.
Hsu, S. C., & Lockwood, J. L. (1975). Powdered chitin agar as a selective medium for enumeration of actinomycetes in water and soil. Applied and Environmental Microbiology, 29(3), 422–426.
Kaya, M., Baublys, V., Can, E., Šatkauskienė, I., Bitim, B., Tubelytė, V., & Baran, T. (2014). Comparison of physicochemical properties of chitins isolated from an insect (Melolontha melolontha) and a crustacean species (Oniscus asellus). Zoomorphology, 13(3), 285–293.
Kim, D. S., Bae, C. Y., Jeon, J. J., Chun, S. J., Oh, H. W., Hong, S. G., Baek, K. S., Moon, E. Y., & Bae, K. S. (2004). Paenibacillus elgii sp. nov., with broad antimicrobial activity. International Journal of Systematic and Evolutionary Microbiology, 54(6), 2031–2035.
Kim, Y. H., Park, S. K., Hur, J. Y., & Kim, Y. C. (2017). Purification and characterization of a major extracellular chitinase from a biocontrol bacterium, Paenibacillus elgii HOA73. The Plant Pathology Journal, 33(3), 318–328.
Kumar, M., Brar, A., Vivekanand, V., & Pareek, N. (2017). Production of chitinase from thermophilic Humicola grisea and its application in production of bioactive chitooligosaccharides. International Journal of Biological Macromolecules, 104, 1641–1647.
Kumar, S. N., Jacob, J., Reshma, U. R., Rajesh, R. O., & Kumar, B. S. (2015). Molecular characterization of forest soil based Paenibacillus elgii and optimization of various culture conditions for its improved antimicrobial activity. Frontiers in Microbiology, 6, 1167–1178.
Larroque, M., Barriot, R., Bottin, A., Barre, A., Rougé, P., Dumas, B., & Gaulin, E. (2012). The unique architecture and function of cellulose-interacting proteins in oomycetes revealed by genomic and structural analyses. BMC Genomics, 13(1), 605–620.
Li, J., Liu, W., Luo, L., Dong, D., Liu, T., Zhang, T., Lu, C., Liu, D., Zhang, D., & Wu, H. (2015). Expression of Paenibacillus polymyxa β-1, 3-1, 4-glucanase in Streptomyces lydicus A01 improves its biocontrol effect against Botrytis cinerea. Biological Control, 90, 141–147.
Liu, S., Sun, J., Yu, L., Zhang, C., Bi, J., Zhu, F., Qu, M., Jiang, C., & Yang, Q. (2012). Extraction and characterization of chitin from the beetle Holotrichia parallela Motschulsky. Molecules, 17(4), 4604–4611.
Miller. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31(3), 426–428.
Meruvu, H., & Donthireddy, S. R. R. (2014). Purification and characterization of an antifungal Chitinase from Citrobacter freundii str. Nov. haritD11. Applied Biochemistry and Biotechnology, 172(1), 196–205.
Narasimhan, A., & Shivakumar, S. (2012). Optimization of chitinase produced by a biocontrol strain of Bacillus subtilis using Plackett-Burman design. European Journal of Experimental Biology, 2(4), 861–865.
Niranjane, A. P., Madhou, P., & Stevenson, T. W. (2007). The effect of carbohydrate carbon sources on the production of cellulase by Phlebia gigantea. Enzyme and Microbial Technology, 40(6), 1464–1468.
Pal, D., Kumar, R. M., Kaur, N., Kumar, N., Kaur, G., Singh, N. K., Krishnamurthi, S., & Mayilraj, S. (2017). Bacillus maritimus sp. nov., a novel member of the genus Bacillus isolated from marine sediment. International Journal of Systematic and Evolutionary Microbiology, 67(1), 60–66.
Patel, B., Gohel, V., & Raol, B. (2007). Statistical optimisation of medium components for chitinase production by Paenibacillus sabina strain JD2. Annals of Microbiology, 57(4), 589–597.
Plackett, R. L., & Burman, J. P. (1946). The design of optimum multifactorial experiments. Biometrika, 33(4), 305–325.
Premalatha, N., Gopal, N. O., Jose, P. A., Anandham, R., & Kwon, S. W. (2015). Optimization of cellulase production by Enhydrobacter sp. ACCA2 and its application in biomass saccharification. Frontiers in. Microbiology., 6, 1046–1057.
Rathore, A. S., & Gupta, R. D. (2015). Chitinases from bacteria to human: Properties, applications, and future perspectives. Enzyme Research, 2015, 1–8.
Sadhu, S., Ghosh, P. K., Aditya, G., & Maiti, T. K. (2014). Optimization and strain improvement by mutation for enhanced cellulase production by Bacillus sp.(MTCC10046) isolated from cow dung. Journal of King Saud University-Science, 26(4), 323–332.
Saima, K. M., & Ahmad, I. Z. (2013). Isolation of novel chitinolytic bacteria and production optimization of extracellular chitinase. Journal, Genetic Engineering & Biotechnology, 11(1), 39–46.
Sandhya, C., Binod, P., Nampoothiri, K. M., Szakacs, G., & Pandey, A. (2005). Microbial synthesis of chitinase in solid cultures and its potential as a biocontrol agent against phytopathogenic fungus Colletotrichum gloeosporioides. Applied Biochemistry and Biotechnology, 127(1), 1–15.
Singh, A. K., Mehta, G., & Chhatpar, H. S. (2009). Optimization of medium constituents for improved chitinase production by Paenibacillus sp. D1 using statistical approach. Letters in Applied Microbiology, 49(6), 708–714.
Sreena, C. P., & Sebastian, D. (2018). Augmented cellulase production by Bacillus subtilis strain MU S1 using different statistical experimental designs. Journal, Genetic Engineering & Biotechnology, 16(1), 9–16.
Tasharrofi, N., Adrangi, S., Fazeli, M., Rastegar, H., Khoshayand, M. R., & Faramarzi, M. A. (2011). Optimization of chitinase production by Bacillus pumilus using Plackett-Burman. Iranian Journal of Pharmaceutical Research, 10(4), 759–768.
Velho-Pereira, S., & Kamat, N. M. (2011). Antimicrobial screening of actinobacteria using a modified cross-streak method. Indian Journal of Pharmaceutical Sciences, 73(2), 223–228.
Warcup, J. H. (1950). The soil-plate method for isolation of fungi from soil. Nature, 166, 117–118.
Acknowledgements
We acknowledge the help rendered by Ms. Lucy Milne, an undergraduate student from Griffith University, Australia for her help in the screening of microorganisms producing chitinase enzyme during her project work at our department.
Funding
The study was supported by Department of Biotechnology, Ministry of Science and Technology, Government of India (project ID: BT/PR10827/AAQ/3/661/2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1834 kb)
Rights and permissions
About this article
Cite this article
Philip, N.V., Koteshwara, A., Kiran, G.A. et al. Statistical Optimization for Coproduction of Chitinase and Beta 1, 4-Endoglucanase by Chitinolytic Paenibacillus elgii PB1 Having Antifungal Activity. Appl Biochem Biotechnol 191, 135–150 (2020). https://doi.org/10.1007/s12010-020-03235-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-020-03235-8