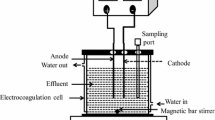
Abstract
The untreated wastewater potentially can be a significant environmental challenge, which requires an effective treatment before discharging into the environment. This paper aimed to study and optimize the removal of the chemical oxygen demand (COD) from industrial wastewater using Electro-Persulfate (EPS) activated by iron electrodes. The Central Composite Design (CCD) was employed for experimental design and statistical analysis of independent operational parameters, including persulfate to COD ratio, pH, treatment time, and applied current on the removal of COD in the wastewater. According to the results of Response Surface Methodology (RSM), the optimal conditions for each critical variable were as follows: \(\frac{{{\text{S}}_{2} {\text{O}}_{8}^{2 - } }}{{{\text{COD}}}} = 1.92\), pH at 9, applied current at 3A, and 40 min of treatment time. The maximum efficiency in the experimental and predicted COD removal was 80 and 78.85%, respectively. The results of Analysis of variance (ANOVA) demonstrated high determination coefficient quantities (R2 = 0.96, Predicted R2 = 0.86 and Adjusted R2 = 0.92) which validated the reliability of the second-order regression model. The amounts of other parameters including total dissolved solids (TDS), total organic carbon (TOC), and biological oxygen demand (BOD) were decreased considerably as well.





Similar content being viewed by others
References
Ahmadi M, Ghanbari F (2016) Optimizing COD removal from greywater by photoelectro-persulfate process using Box-Behnken design: assessment of effluent quality and electrical energy consumption. Environ Sci Pollut Res 23:19350–19361. https://doi.org/10.1007/s11356-016-7139-6
Akbari S, Ghanbari F, Moradi M (2016) Bisphenol A degradation in aqueous solutions by electrogenerated ferrous ion activated ozone, hydrogen peroxide and persulfate: applying low current density for oxidation mechanism. Chem Eng J 294:298–307. https://doi.org/10.1016/j.cej.2016.02.106
Aksoy DO, Sagol E (2016) Application of central composite design method to coal flotation: modelling, optimization and verification. Fuel 183:609–616. https://doi.org/10.1016/J.FUEL.2016.06.111
Asha TT, Gandhimathi R, Ramesh ST, Nidheesh PV (2016) Treatment of stabilized leachate by ferrous-activated persulfate oxidative system. J Hazardous, Toxic, Radioact Waste 21:04016012. https://doi.org/10.1061/(ASCE)HZ.2153-5515.0000328
Bayat A, Shokri A (2021) Degradation of p-Nitrotoluene in aqueous environment by Fe(II)/Peroxymonosulfate using full factorial experimental design. Sep Sci Technol 56:2941–2950. https://doi.org/10.1080/01496395.2020.1861016
Chanikya P, Nidheesh PV, Babu DS, Gopinath A, Kumar MS (2021) Treatment of dyeing wastewater by combined sulfate radical based electrochemical advanced oxidation and electrocoagulation processes. Sep Purif Technol 254:117570. https://doi.org/10.1016/j.seppur.2020.117570
Chen W-S, Jhou Y-C, Huang C-P (2014) Mineralization of dinitrotoluenes in industrial wastewater by electro-activated persulfate oxidation. Chem Eng J 252:166–172. https://doi.org/10.1016/j.cej.2014.05.033
Ganiyu SO, El-Din MG (2020) Insight into in-situ radical and non-radical oxidative degradation of organic compounds in complex real matrix during electrooxidation with boron doped diamond electrode: A case study of oil sands process water treatment. Appl Catal B 279:119366. https://doi.org/10.1016/j.apcatb.2020.119366
Ghanbari F, Martínez-Huitle CA (2019) Electrochemical advanced oxidation processes coupled with peroxymonosulfate for the treatment of real washing machine effluent: a comparative study. J Electroanal Chem 847:113182. https://doi.org/10.1016/j.jelechem.2019.05.064
Giannakis S, Lin KYA, Ghanbari F (2021) A review of the recent advances on the treatment of industrial wastewaters by Sulfate Radical-based Advanced Oxidation Processes (SR-AOPs). Chem Eng J 406:127083. https://doi.org/10.1016/J.CEJ.2020.127083
Görmez F, Görmez Ö, Yabalak E, Gözmen B (2020) Application of the central composite design to mineralization of olive mill wastewater by the electro/FeII/persulfate oxidation method. SN Appl Sci 2:1–11. https://doi.org/10.1007/S42452-020-1986-Y/TABLES/6
Guvenc SY, Varank G (2021) Box-Behnken design optimization of electro-fenton/-persulfate processes following the acidification for TSS removal from biodiesel wastewater. Sigma J Eng Nat Sci 38:1767–1780
Hossein Panahi A, Meshkinian A, Ashrafi SD et al (2020) Survey of sono-activated persulfate process for treatment of real dairy wastewater. Int J Environ Sci Technol 17:93–98. https://doi.org/10.1007/s13762-019-02324-4
Jaafarzadeh N, Omidinasab M, Ghanbari F (2016) Combined electrocoagulation and UV-based sulfate radical oxidation processes for treatment of pulp and paper wastewater. Process Saf Environ Prot 102:462–472. https://doi.org/10.1016/j.psep.2016.04.019
Kattel E, Dulova N (2016) Ferrous ion-activated persulphate process for landfill leachate treatment: removal of organic load, phenolic micropollutants and nitrogen. Environ Technol 38:1223–1231. https://doi.org/10.1080/09593330.2016.1221472
Liu K, Bai L, Shi Y et al (2020) Simultaneous disinfection of E. faecalis and degradation of carbamazepine by sulfate radicals: an experimental and modelling study. Environ Pollut 263:114558. https://doi.org/10.1016/J.ENVPOL.2020.114558
Masomboon N, Ratanatamskul C, Lu MC (2010) Chemical oxidation of 2,6-dimethylaniline by electrochemically generated Fenton’s reagent. J Hazard Mater 176:92–98. https://doi.org/10.1016/J.JHAZMAT.2009.11.003
Montgomery DC (2017) Design and analysis of experiments
Moradi M, Ghanbari F, Minaee Tabrizi E (2015) Removal of acid yellow 36 using Box-Behnken designed photoelectro-Fenton: a study on removal mechanisms. Toxicol Environ Chem 97:700–709. https://doi.org/10.1080/02772248.2015.1060975
Rao YF, Qu L, Yang H, Chu W (2014) Degradation of carbamazepine by Fe(II)-activated persulfate process. J Hazard Mater 268:23–32. https://doi.org/10.1016/J.JHAZMAT.2014.01.010
Romero A, Santos A, Vicente F, González C (2010) Diuron abatement using activated persulphate: effect of pH, Fe(II) and oxidant dosage. Chem Eng J 162:257–265. https://doi.org/10.1016/J.CEJ.2010.05.044
Sahu JN, Acharya J, Meikap BC (2010) Optimization of production conditions for activated carbons from Tamarind wood by zinc chloride using response surface methodology. Bioresour Technol 101:1974–1982. https://doi.org/10.1016/J.BIORTECH.2009.10.031
Shokri A (2018) Application of Sono–photo-Fenton process for degradation of phenol derivatives in petrochemical wastewater using full factorial design of experiment. Int J Ind Chem 9:295–303. https://doi.org/10.1007/S40090-018-0159-Y/FIGURES/6
Shokri A, Bayat A, Mahanpoor K (2019) Employing fenton-like process for the remediation of petrochemical wastewater through Box-Behnken design method. Desalin Water Treat 166:135–143. https://doi.org/10.5004/dwt.2019.24634
Silveira JE, Cardoso TO, Barreto-Rodrigues M et al (2017) Electro activation of persulfate using iron sheet as low-cost electrode: the role of the operating conditions. Environ Technol 39:1208–1216. https://doi.org/10.1080/09593330.2017.1323960
Varank G, Guvenc SY, Demir A et al (2020a) Modeling and optimizing electro-persulfate processes using Fe and Al electrodes for paper industry wastewater treatment. Water Sci Technol 81:345–357. https://doi.org/10.2166/WST.2020.115
Varank G, Yazici Guvenc S, Demir A (2020b) Electro-activated peroxymonosulfate and peroxydisulfate oxidation of leachate nanofiltration concentrate: multiple-response optimization. Int J Environ Sci Technol 17:2707–2720. https://doi.org/10.1007/s13762-020-02651-x
Varank G, Yazici Guvenc S, Dincer K, Demir A (2020c) Concentrated leachate treatment by electro-fenton and electro-persulfate processes using central composite design. Int J Environ Res 14:439–461. https://doi.org/10.1007/S41742-020-00269-Y
Wang J, Wang S (2018) Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chem Eng J 334:1502–1517. https://doi.org/10.1016/J.CEJ.2017.11.059
Wang X, Wang L, Li J et al (2014) Degradation of Acid Orange 7 by persulfate activated with zero valent iron in the presence of ultrasonic irradiation. Sep Purif Technol 122:41–46. https://doi.org/10.1016/J.SEPPUR.2013.10.037
Wei X, Gao N, Li C et al (2016) Zero-valent iron (ZVI) activation of persulfate (PS) for oxidation of bentazon in water. Chem Eng J 285:660–670. https://doi.org/10.1016/J.CEJ.2015.08.120
Xue W, Cui Y, Liu Z, Yang S, Li J (2020) Guo, X (2020) Treatment of landfill leachate nanofiltration concentrate after ultrafiltration by electrochemically assisted heat activation of peroxydisulfate. Sep Purif Technol 231:115928. https://doi.org/10.1016/j.seppur.2019.115928
Xu XR, Li XZ (2010) Degradation of azo dye Orange G in aqueous solutions by persulfate with ferrous ion. Sep Purif Technol 72:105–111. https://doi.org/10.1016/J.SEPPUR.2010.01.012
Yang W, Liu G, Chen Y, Miao D, Wei Q, Li H, Ma L, Zhou K, Liu L, Yu Z (2020) Persulfate enhanced electrochemical oxidation of highly toxic cyanide-containing organic wastewater using boron-doped diamond anode. Chemosphere 252:126499. https://doi.org/10.1016/j.chemosphere.2020.126499
Yazici Guvenc S, Varank G, Cebi A, Ozkaya B (2021) Electro-activated persulfate oxidation of biodiesel wastewater following acidification phase: optimization of process parameters using Box-Behnken design. Water, Air, Soil Pollut 232:1–19. https://doi.org/10.1007/S11270-020-04962-8
Zhi D, Lin Y, Jiang L et al (2020) Remediation of persistent organic pollutants in aqueous systems by electrochemical activation of persulfates: a review. J Environ Manage 260:110–125. https://doi.org/10.1016/J.JENVMAN.2020.110125
Zhou L, Zheng W, Ji Y et al (2013) Ferrous-activated persulfate oxidation of arsenic(III) and diuron in aquatic system. J Hazard Mater 263:422–430. https://doi.org/10.1016/J.JHAZMAT.2013.09.056
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest in this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shokri, A., Soleimani, F. & Sanavi Fard, M. The removal of COD in industrial wastewater by electro-persulfate process using central composite design. Chem. Pap. 76, 7133–7145 (2022). https://doi.org/10.1007/s11696-022-02370-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02370-2