Abstract
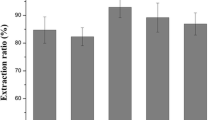
The tendency to consume the plant-based protein is gradually increasing with the increase of the world population. Although the leaf proteins are the sustainable protein sources, the protein isolation from them is difficult due to plants structure. The objective of this study was to evaluate the mallow leaf as an alternative protein source, to increase the protein efficiency with enzyme assisted extraction and to determine their functional properties for industrial food production. The optimum conditions for the enzymes were found as 5.6 pH value, 52.5 ˚C, 75 min and 7% enzyme concentration for Pectinex UF, 7 pH value, 48 ˚C, 75.5 min and 7% enzyme concentration for Pectinex Ultra SP-L by response surface methodology. The effect of precipitation methods, isoelectric (IP), ammonium sulfate (ASP) and isoelectric-ammonium sulfate (IASP), on efficiency and functional properties of plant proteins was investigated. The protein contents of IP, ASP and IASP were 40, 65.5 and 61.7%, respectively. Despite the lowest protein content, IP (97% at pH 7.5) has the highest solubility than ASP (95% at pH 7.5) and IASP (94.4% at pH 8.5). The highest foaming capacity at pH 6 was determined as 133.3, 90.9 and 80%, respectively, for IP, ASP and IASP. Also, IP has the highest emulsion activity and stability than ASP and IASP. All these evaluations, mallow leaf can be used for protein isolation, but its economic dimension should be investigated through more comprehensive studies.






Similar content being viewed by others
References
M. Shahbandeh, Global plant protein market value 2022–2027. https://www.statista.com/statistics/658619/global-plant-protein-market-value/ Accessed 03.01.2023
A. Akyüz, S. Ersus, Food Chem. (2021) https://doi.org/10.1016/j.foodchem.2020.127673
M. Vergara-Barberán, M. Lerma-García, J. Herrero-Martínez, E.F. Simó-Alfonso, Food Chem. (2015) https://doi.org/10.1016/j.foodchem.2014.07.116
Q. Cui, X. Ni, L. Zeng, Z. Tu, J. Li, K. Sun, X. Chen, X. Li, Hortic. Plant. J. (2017) https://doi.org/10.1016/j.hpj.2017.06.003
D.Q. Zhang, T.H. Mu, H.N. Sun, J.W. Chen, M. Zhang, Int. J. Food Prop. (2017) https://doi.org/10.1080/10942912.2016.1230873
P.H. Davis, Flora of Turkey and East Aegean Islands, vol. 2 (Edinburgh University Press, 1966), pp. 179–242
Association of Official Analytical Chemists (AOAC), Official method of analysis, 14th edn., Washington, DC, USA, (1990)
S.J. Makwana, B.A. Jadeja, J. Pharmacogn. Phytochem. 5, pp. 209–2014 (2016)
R. Sánchez-Vioque, A. Clemente, J. Vioque, J. Bautista, F. Millán, Food Chem. (1999) https://doi.org/10.1016/S0308-8146(98)00133-2
Y. Lv, L. Cai, M. Yang, X. Liu, N. Hui, J. Li, Int. J. Food Prop. (2017) https://doi.org/10.1080/10942912.2017.1354019
S. Gonzalez-Perez, Doctoral dissertation, Ph.D. thesis, Wageningen University, Wageningen, The Netherlands (2003)
R. Toews, N. Wang, Food Res. Int. (2013) https://doi.org/10.1016/j.foodres.2012.12.009
P. Kaur, S.K. Singh, V. Garg, M. Gulati, Y. Vaidya, Powder Technol. (2015) https://doi.org/10.1016/j.powtec.2015.06.034
N. **apong, M. Suphantharika, P. Jamnong, J. Food Eng. (2008) https://doi.org/10.1016/j.jfoodeng.2007.04.032
C.F. Chau, P.C. Cheung, Y.S. Wong, J. Agric. Food Chem. (1997) https://doi.org/10.1021/jf970047c
R. Tabaraki, Z. Yosefi, G.H.A. Asadi, JRAS. 8, 59–68 (2012)
L. Barros, A.M. Carvalho, I.C. Ferreira, Food Chem. Toxicol. (2010) https://doi.org/10.1016/j.fct.2010.03.012
E. Molina-Alcaide, D.R. Yáñez-Ruiz, Anim. Feed Sci. Technol. (2008) https://doi.org/10.1016/j.anifeedsci.2007.09.021
J.C. Gasparetto, C.A.F. Martins, S.S. Hayashi, M.F. Otuky, R. Pontarolo, J. Pharm. Pharmacol. (2012) https://doi.org/10.1111/j.2042-7158.2011.01383.x
M. Wichtl, Herbal Drugs and Phytopharmaceuticals: A Handbook for Practice on a Scientific Basis, 3rd edn. (Medpharm GmbH Scientific Publishers, 2004)
M.S. Hadnadev, T. Hadnađev-Dapčević, M.M. Pojić, B.M. Šarić, A.C. Mišan, P.T. Jovanov, M.B. Sakač, Food Feed Res. (2017) https://doi.org/10.5937/FFR1701011H
L.L. Tymchak, Clinical Chemistry: Principles, Techniques, and Correlations (Lippincott Williams & Wilkins, 2013), pp. 223–262
F. Sosulski, K. Sosulski, A. Mccurdy, Acta Aliment. 14, 41–49 (1988)
Y.A. Adebowale, I.A. Adeyemi, A.A. Oshodi, K. Niranjan, Food Chem. (2007) https://doi.org/10.1016/j.foodchem.2006.11.050
J.C. Wang, J.E. Kinsella, J. Food Sci. (1976) https://doi.org/10.1111/j.1365-2621.1976.tb00602.x
P. Schuck, Advances in Dairy Ingredients (Wiley-Blackwell, 2013), pp. 1–29
P.J. Kanu, Z. Kerui, Z.H. Ming, Q. Haifeng, J.B. Kanu, Z. Kexue, Asian J. Biochem. (2007) https://doi.org/10.3923/ajb.2007.289.301
R.E. Aluko, R.Y. Yada, Food Chem. (1995) https://doi.org/10.1016/0308-8146(95)93931-G
J.C. Onweluzo, Z.A. Obanu, K.C. Onuoha, J. Food Sci. Technol. 31, 301–304 (1994)
A. Mahajan, S. Dua, J. Food Sci. Tech. Mys 32, 162–165 (1995)
Funding
This work was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) under Grant 119O310.
Author information
Authors and Affiliations
Contributions
Seda ERSUS: Conceptualization, Supervision, Methodology, Writing-Review & Editing, Funding acquisition, Resources, Validation. Ayça AKYÜZ: Conceptualization, Methodology, Software, Project administration, Visualization, Formal analysis, Investigation.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ersus, S., Akyüz, A. Enzyme assisted extraction of protein from mallow leaf (Malva sylvestris L.) for production of alternative protein concentrate. Food Measure 17, 3283–3294 (2023). https://doi.org/10.1007/s11694-023-01874-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-023-01874-z