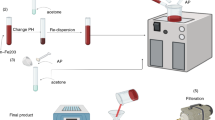
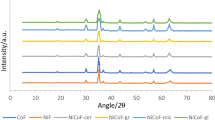
Abstract
While ferric oxide is frequently used as a catalyst, aluminum, with combustion enthalpy 32000 J g−1, is a versatile highly energy-dense material. This binary mixture can achieve a synergistic impact with a strong thermite reaction. Additionally, nanoscale particles can offer better interfacial surface area, catalytic effect, and a high heat release rate. This study reports on the facile fabrication of colloidal ferric oxide nanoparticles (Fe2O3 NPs) of 5-nm average size; aluminum nanoplates of 100 nm were employed. Colloidal nanothermites (Fe2O3/Al) were synthesized and integrated into ammonium perchlorate (APC) via co-precipitation. Uniform dispersion of nanothermite particles into APC was confirmed via elemental map** using an EDAX detector. The nanothermite mixture's potentials (as a catalyst and highly energy-dense material) was described by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA). Thermite NPs offered a decrease in APC endothermic phase change by 48%, increasing the total heat released by 68%. While APC demonstrated a decomposition enthalpy of 836 J/g, for the APC nanocomposite it was 1405 J/g. Aluminum particles increased decomposition enthalpy, and Fe2O3 NPs acted as an effective catalyst to decrease the required activation energy. The kinetic decomposition study of APC nanocomposite was investigated via isothermal heating using TGA. Nanothermite particles offered a reduction in APC activation energy by 11% and 14% using Kissinger's and Kissinger–Akahira–Sunose (KAS) models, respectively. Herein, this is the first report on catalytic activity assessment of colloidal nanothermite particles on APC decomposition.
Graphic Abstract










Similar content being viewed by others
Availability of Data and Material
Not applicable.
References
J. Conkling, and C. Mocella, Chemistry of Pyrotechnics Basic Principles and Theory (London: CRC, 2012).
S. Elbasuney, M. Gobara, and M. Yehia, J. Inorg. Organomet. Polym. Mater. 29, 721 (2019).
S. Elbasuney, M. Gobara, M.G. Zaky, M. Radwan, A. Maraden, S. Ismael, E. Elsaka, M.A. Elkodous, and G.S. El-Sayyad, J. Mater. Sci. Mater. Electron. 31, 8212 (2020).
W.-Q. Pang, L.T. DeLuca, X.-Z. Fan, O.G. Glotov, K. Wang, Z. Qin, and F.-Q. Zhao, Combust. Flam. 220, 157 (2020).
S. Jain, G. Gupta, D.R. Kshirsagar, V.H. Khire, and B. Kandasubramanian, Def. Technol. 15, 313 (2019).
P.A. Figueiredo, and F.M. Brójo, Energy Proc. 136, 202 (2017).
R. Meyer, J. Kohler, and A. Homburg, Explosives, 6th ed., (Weinheim: Wiley, 2007).
A. Manash, and P. Kumar, Def. Technol. 15, 227 (2019).
C.A.M. Dillier, E.D. Petersen, and E.L. Petersen, Proc. Combust. Inst. 38, 4409 (2020).
D.-Y. Tang, Z.-M. Fan, G. Yang, Y. Wang, P.-J. Liu, and Q.-L. Yan, Combust. Flam. 219, 33 (2020).
R. Sangtyani, H.S. Saha, A. Kumar, A. Kumar, M. Gupta, and P.V. Chavan, Combust. Flam. 209, 357 (2019).
A.S. Budhwar, A. Gautam, P.V. More, C.S. Pant, S. Banerjee, and P.K. Khanna, Vacuum 156, 483 (2018).
S. Elbasuney, and M. Yehia, Def. Technol. 15, 868 (2019).
S. Elbasuney, and A. Fahd, Fuel 237, 1274 (2019).
S. Elbasuney, J. Inorg. Organomet. Polym. Mater. 28, 1793 (2018).
S. Elbasuney, A. Elsaidy, M. Kassem, H. Tantawy, R. Sadek, A. Fahd, and M. Gobara, J. Inorg. Organomet. Polym. Mater. 28, 2231 (2018).
S. Elbasuney, Powder Technol. 268, 158 (2014).
S. Elbasuney, Particular 22, 66 (2015).
S. Elbasuney, Powder Technol. 277, 63 (2015).
J.A. Vara, P.N. Dave, and V.R. Ram, Nano-struct. Nano-obj. 20, 100372 (2019).
N. Yadav, P.K. Srivastava, and M. Varma, Def. Technol. 2, 3–9 (2020). https://doi.org/10.1016/j.dt.2020.06.007 (in press).
S. Elbasuney, and M. Yehia, J. Inorg. Organomet. Polym. Mater. 30, 706 (2020).
S. Elbasuney, Appl. Surf. Sci. 409, 438 (2017).
S. Elbasuney, and M. Yehia, J. Inorg. Organomet. Polym. Mater. 29, 1349 (2019).
A. Khawam, and D.R. Flanagan, J. Pharm. Sci. 95, 472 (2006).
D. Trache, A. Abdelaziz, and B. Siouani, J. Therm. Anal. Calorim. 128, 335 (2017).
S. Vyazovkin, A.K. Burnham, J.M. Criado, L.A. Pérez-Maqueda, C. Popescu, and N. Sbirrazzuoli, Thermochim. Acta 520, 1 (2011).
D. Trache, K. Khimeche, A. Mezroua, and M. Benziane, J. Therm. Anal. Calorim. 124, 1485 (2016).
S. Elbasuney, A. Hamed, S. Ismael, M. Mokhtar, and M. Gobara, J. Inorg. Organomet. Polym. Mater. 30, 3980 (2020).
E.L. Dreizin, Prog. Energy Combust. Sci. 35, 141 (2009).
S. Elbasuney, G.S. El-Sayyad, M. Yehia, and S.K.A. Aal, J. Mater. Sci.: Mater. Electron. 31, 20805 (2020).
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
All the authors approve the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elbasuney, S., Hamed, A., Yehia, M. et al. Colloidal Nanothermite Particles: Advanced Nanocatalyst and Energy Dense Material for Ammonium Perchlorates. J. Electron. Mater. 50, 6128–6134 (2021). https://doi.org/10.1007/s11664-021-09141-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-021-09141-x