Abstract
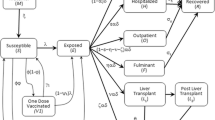
Hepatitis B (HepB) is one of the most common infectious diseases affecting over two billion people worldwide. About one third of all HepB cases are in China. In recent years, China made significant efforts to implement a nationwide HepB vaccination program and reduced the number of unvaccinated infants from 30 to 10%. However, many individuals still remain unprotected, particularly those born before 2003. Consequently, a catch-up retroactive vaccination is an important and potentially cost-effective way to reduce HepB prevalence. In this paper, we analyze a game theoretical model of HepB dynamics that incorporates government-provided vaccination at birth coupled with voluntary retroactive vaccinations. Given the uncertainty about the long-term efficacy of the HepB vaccinations, we study several scenarios. When the waning rate is relatively high, we show that this retroactive vaccination should be a necessary component of any HepB eradication effort. When the vaccine offers long-lasting protection, the voluntary retroactive vaccination brings the disease incidence to sufficiently low levels. Also, we find that the optimal vaccination rates are almost independent of the vaccination coverage at birth. Moreover, it is in an individual’s self-interest to vaccinate (and potentially re-vaccinate) at a rate just slightly above the vaccine waning rate.




Similar content being viewed by others
References
Abdeldayem HM, Salama I, Soliman S, Gameel K, Gabal AA, El KE, Helmy A (2008) Patients seeking liver transplant turn to China: outcomes of 15 Egyptian patients who went to China for a deceased-donor liver transplant. Exp Clin Transpl Off J Middle East Soc Organ Transpl 6(3):194–198
Acosta-Alonzo C, Erovenko IV, Lancaster A, Oh H, Rychtář J, Taylor D (2020) High endemic levels of typhoid fever in rural areas of Ghana may stem from optimal voluntary vaccination behavior. Preprint
Alter MJ (2003) Epidemiology and prevention of hepatitis B. Semin Liver Dis 23:039–046
Anonychuk AM, Tricco AC, Bauch CT, Gilca V, Duval B, John-Baptiste A, Woo G, Krahn M et al (2008) Cost-effectiveness analyses of hepatitis A vaccine. Pharmacoeconomics 26(1):17–32
Arriola L, Hyman JM (2009) Sensitivity analysis for uncertainty quantification in mathematical models. In: Mathematical and statistical estimation approaches in epidemiology. Springer, pp 195–247
Bancroft WH, Snitbhan R, McNair Scott R, Tingpalapong M, Watson WT, Tanticharoenyos P, Karwacki JJ, Srimarut S (1977) Transmission of hepatitis B virus to gibbons by exposure to human saliva containing hepatitis B surface antigen. J Infect Dis 135(1):79–85
Bankuru SV, Kossol S, Hou W, Mahmoudi P, Rychtář J, Taylor D (2020) A game-theoretic model of Monkeypox to assess vaccination strategies. Peerj. https://doi.org/10.7717/peerj.9272
Barker L, Murray R (1972) Relationship of virus dose to incubation time of clinical hepatitis and time of appearance of hepatitis-associated antigen. Am J Med Sci 263(1):27–33
Bauch CT, Earn DJ (2004) Vaccination and the theory of games. Proc Natl Acad Sci 101(36):13391–13394
Bauch C, Anonychuk A, Pham B, Gilca V, Duval B, Krahn M (2007a) Cost-utility of universal hepatitis A vaccination in Canada. Vaccine 25(51):8536–8548
Bauch C, Rao ASS, Pham B, Krahn M, Gilca V, Duval B, Chen M, Tricco A (2007b) A dynamic model for assessing universal hepatitis A vaccination in Canada. Vaccine 25(10):1719–1726
Bosch FX, Ribes J, Borràs J (1999) Epidemiology of primary liver cancer. Semin Liver Dis 19(3):271–285
Brettin A, Rossi-Goldthorpe R, Weishaar K, Erovenko IV (2018) Ebola could be eradicated through voluntary vaccination. R Soc Open Sci 5(1):171591
Broom M, Rychtár J, Spears-Gill T (2016) The game-theoretical model of using insecticide-treated bed-nets to fight malaria. Appl Math 7(09):852–860
Bruce MG, Bruden D, Hurlburt D, Zanis C, Thompson G, Rea L, Toomey M, Townshend-Bulson L, Rudolph K, Bulkow L et al (2016) Antibody levels and protection after hepatitis B vaccine: results of a 30-year follow-up study and response to a booster dose. J Infect Dis 214(1):16–22
CDC (2007) Progress in hepatitis B prevention through universal infant vaccination-China, 1997–2006. Morb Mortal Week Rep 56(18):441
Chang SL, Piraveenan M, Pattison P, Prokopenko M (2020) Game theoretic modelling of infectious disease dynamics and intervention methods: a review. J Biol Dyn 14(1):57–89
Chen S, Li J, Wang D, Fung H, Wong L-Y, Zhao L (2018) The hepatitis B epidemic in China should receive more attention. Lancet 391(10130):1572
Cheng E, Gambhirrao N, Patel R, Zhowandai A, Rychtář J, Taylor D (2020) A game-theoretical analysis of Poliomyelitis vaccination. J Theor Biol. https://doi.org/10.1016/j.jtbi.2020.110298
Crawford K, Lancaster A, Oh H, Rychtář J (2015) A voluntary use of insecticide-treated cattle can eliminate African slee** sickness. Lett Biomath 2(1):91–101
Dorsett C, Oh H, Paulemond ML, Rychtář J (2016) Optimal repellent usage to combat Dengue fever. Bull Math Biol 78(5):916–922
EASL (2012) European Association for the Study of the Liver. Clinical practice guidelines: management of chronic hepatitis B virus infection. J Hepatol 57(1):167–185
Edmunds W, Medley G, Nokes D (1996a) The transmission dynamics and control of hepatitis B virus in the Gambia. Stat Med 15(20):2215–2233
Edmunds W, Medley G, Nokes D (1996b) Vaccination against hepatitis B virus in highly endemic areas: waning vaccine-induced immunity and the need for booster doses. Trans R Soc Trop Med Hyg 90(4):436–440
Edmunds W, Medley G, Nokes D, O’callaghan C, Whittle H, Hall A (1996c) Epidemiological patterns of hepatitis B virus (HBV) in highly endemic areas. Epidemiol Infect 117(2):313–325
Hahné S, Ramsay M, Balogun K, Edmunds WJ, Mortimer P (2004) Incidence and routes of transmission of hepatitis B virus in England and Wales, 1995–2000: implications for immunisation policy. J Clin Virol 29(4):211–220
Hou J, Liu Z, Gu F (2005) Epidemiology and prevention of hepatitis B virus infection. Int J Med Sci 2(1):50
Hutton DW, So SK, Brandeau ML (2010) Cost-effectiveness of nationwide hepatitis B catch-up vaccination among children and adolescents in China. Hepatology 51(2):405–414
Hyams KC (1995) Risks of chronicity following acute hepatitis B virus infection: a review. Clin Infect Dis 20(4):992–1000
Ibuka Y, Li M, Vietri J, Chapman GB, Galvani AP (2014) Free-riding behavior in vaccination decisions: an experimental study. PLoS ONE 9(1):e87164. https://doi.org/10.1371/journal.pone.0087164
Inskip H, Hall A, Chotard J, Loik F, Whittle H (1991) Hepatitis B vaccine in the Gambian expanded programme on immunization: factors influencing antibody response. Int J Epidemiol 20(3):764–769
Kamyad AV, Akbari R, Heydari AA, Heydari A (2014) Mathematical modeling of transmission dynamics and optimal control of vaccination and treatment for hepatitis B virus. Comput Math Methods Med. https://doi.org/10.1155/2014/475451
Klein SRM, Foster AO, Feagins DA, Rowell JT, Erovenko IV (2019) Optimal voluntary and mandatory insect repellent usage and emigration strategies to control the chikungunya outbreak on Reunion Island. Preprint
Kobe J, Pritchard N, Short Z, Erovenko IV, Rychtář J, Rowell JT (2018) A game-theoretic model of cholera with optimal personal protection strategies. Bull Math Biol 80(10):2580–2599
Kretzschmar M, Mangen M-J, van de Laar M, de Wit A (2009) Model based analysis of hepatitis B vaccination strategies in the Netherlands. Vaccine 27(8):1254–1260
Lavanchy D (2004) Hepatitis B virus epidemiology, disease burden, treatment, and current and emerging prevention and control measures. J Viral Hepatitis 11(2):97–107
Leuridan E, Van Damme P (2011) Hepatitis B and the need for a booster dose. Clin Infect Dis 53(1):68–75
Liang P, Zu J, Zhuang G (2018) A literature review of mathematical models of hepatitis B virus transmission applied to immunization strategies from 1994 to 2015. J Epidemiol 28(5):221–229. https://doi.org/10.2188/jea.JE20160203
Lok AS, McMahon BJ (2009) Chronic hepatitis B: update 2009. Hepatology 50(3):661–662
Martinez A, Machado J, Sanchez E, Erovenko I (2019) Optimal vaccination strategies to reduce endemic levels of meningitis in Africa. Preprint
Maskin E (1999) Nash equilibrium and welfare optimality. Rev Econ Stud 66(1):23–38
Medley GF, Lindop NA, Edmunds WJ, Nokes DJ (2001) Hepatitis-B virus endemicity: heterogeneity, catastrophic dynamics and control. Nat Med 7(5):619
Molina C (2016) The good, the finite, and the infinite. Ph.D. thesis, McMaster University
Neilan RLM, Schaefer E, Gaff H, Fister KR, Lenhart S (2010) Modeling optimal intervention strategies for cholera. Bull Math Biol 72(8):2004–2018
Nommensen F, Go S, MacLaren D (1989) Half-life of HBs antibody after hepatitis B vaccination: an aid to timing of booster vaccination. Lancet 334(8667):847–849
O’Leary C, Hong Z, Zhang F, Dawood M, Smart G, Kaita K, Wu J (2010) A mathematical model to study the effect of hepatitis B virus vaccine and antivirus treatment among the Canadian Inuit population. Eur J Clin Microbiol Infect Dis 29(1):63
Overby L, Ling C, Decker R, Mushahwar I, Chau K (1982) Serodiagnostic profiles of viral hepatitis. In: Szmuzzess W, Alter MJ, Maynard JE (eds) Viral Hepat. Franklin Institute Press, Philadelphia
Pang J, Cui J-A, Zhou X (2010) Dynamical behavior of a hepatitis B virus transmission model with vaccination. J Theor Biol 265(4):572–578
Scheckelhoff K, Ejaz A, Erovenko IV (2019) A game-theoretic model of optimal clean equipment usage to prevent hepatitis C among injecting drug users. Preprint
Scott RM, Snitbhan R, Bancroft WH, Alter HJ, Tingpalapong M (1980) Experimental transmission of hepatitis B virus by semen and saliva. J Infect Dis 142(1):67–71
Serpell L, Green J (2006) Parental decision-making in childhood vaccination. Vaccine 24(19):4041–4046
Stevens CE, Neurath RA, Beasley RP, Szmuness W (1979) HBeAg and anti-HBe detection by radioimmunoassay: correlation with vertical transmission of hepatitis B virus in Taiwan. J Med Virol 3(3):237–241
Sykes D, Rychtář J (2015) A game-theoretic approach to valuating Toxoplasmosis vaccination strategies. Theor Popul Biol 105:33–38
Thornley S, Bullen C, Roberts M (2008) Hepatitis B in a high prevalence new zealand population: a mathematical model applied to infection control policy. J Theor Biol 254(3):599–603
Toy M, Hutton DW, So SK (2015) Cost-effectiveness and cost thresholds of generic and brand drugs in a national chronic hepatitis B treatment program in China. PLoS ONE 10(11):e0139876
United Nations (2019) World population prospects. https://population.un.org/wpp/. Accessed 12 May 2019
Van Damme P (2016) Long-term protection after hepatitis B vaccine. J Infect Dis 214(1):1–3
Wang F, Shen L, Cui F, Zhang S, Zheng H, Zhang Y, Liang X, Wang F, Bi S (2015) The long-term efficacy, 13–23 years, of a plasma-derived hepatitis B vaccine in highly endemic areas in China. Vaccine 33(23):2704–2709
Wang H, Men P, **ao Y, Gao P, Lv M, Yuan Q, Chen W, Bai S, Wu J (2019) Hepatitis B infection in the general population of China: a systematic review and meta-analysis. BMC Infect Dis 19(1):811
WHO (2019) Hepatitis B: World Health Organization fact sheet 204. http://www.who.int/mediacentre/factsheets/fs204/en/. Accessed 24 Dec 2019
Williams J, Nokes D, Medley G, Anderson R (1996) The transmission dynamics of hepatitis B in the UK: a mathematical model for evaluating costs and effectiveness of immunization programmes. Epidemiol Infect 116(1):71–89
World Bank (2019) Life expectancy at birth. https://data.worldbank.org/indicator/SP.DYN.LE00.IN?locations=CN. Accessed 18 Dec 2019
Zhang S, Zhou Y (2012) The analysis and application of an HBV model. Appl Math Model 36(3):1302–1312
Zhao S, Xu Z, Lu Y (2000) A mathematical model of hepatitis B virus transmission and its application for vaccination strategy in China. Int J Epidemiol 29(4):744–752
Zhu D, Wang J, Wangen KR (2014) Hepatitis B vaccination coverage rates among adults in rural China: are economic barriers relevant? Vaccine 32(49):6705–6710
Zhu D, Guo N, Wang J, Nicholas S, Wang Z, Zhang G, Shi L, Wangen KR (2018) Socioeconomic inequality in hepatitis B vaccination of rural adults in China. Hum Vaccines Immunother 14(2):464–470
Zou L, Ruan S, Zhang W (2010a) An age-structured model for the transmission dynamics of hepatitis B. SIAM J Appl Math 70(8):3121–3139
Zou L, Zhang W, Ruan S (2010b) Modeling the transmission dynamics and control of hepatitis B virus in China. J Theor Biol 262(2):330–338
Acknowledgements
AC, SM, MN, and VP worked on this manuscript as part of the course MATH/BIOL 380—Introduction to mathematical biology. They wish to acknowledge the help and support of their classmates. The authors would also like to thank the anonymous reviewers for their comments and suggestions that helped to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A: Equilibria of the Dynamics
Appendix A: Equilibria of the Dynamics
We have to solve the following system of (algebraic) equations.
1.1 Disease Free Equilibrium
In the disease free equilibrium when \(0=L_0=I_0=C_0=R_0\), the system (23)–(28) reduces to
Adding (29) and (30) together yields
By (30),
and thus
Now, we will determine the number of secondary infections caused by a single infected individual. The individual stays infected for a period \(\frac{1}{\mu _0+\gamma _1}\). During that period, at rate \(\beta S_0\), they infect susceptible individuals who become latently infected. Of those, only a fraction of \(\frac{\sigma }{\sigma +\mu _0}\) becomes acutely infected. Moreover, an infected individual can become a carrier with probability \(\frac{q\gamma _1}{\mu _0+\gamma _1}\). The individual stays in the carrier compartment for a period \(\frac{1}{\mu _0+\mu _1+\gamma _2}\). During that period, at rate \(\beta \epsilon S_0\), they infect susceptible individuals who then become latently infected. The carrier also makes other carriers at rate \(\mu \omega v\). Consequently, if l is a number of latently infected individuals caused by a single carrier, we get
and thus
Putting it all together, we get, as in Zou et al. (2010b)
The disease free equilibrium is stable when \(R_0<1\) (Zou et al. 2010b, Theorem 1).
1.2 Endemic Equilibrium
From (28), (26), and (27) we get
From (25),
which, after dividing by \(I^*\ne 0\), yields
From (24),
and finally, by (23)
which yields
Rights and permissions
About this article
Cite this article
Chouhan, A., Maiwand, S., Ngo, M. et al. Game-Theoretical Model of Retroactive Hepatitis B Vaccination in China. Bull Math Biol 82, 80 (2020). https://doi.org/10.1007/s11538-020-00748-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-020-00748-5