Abstract
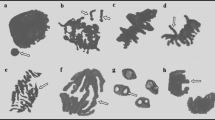
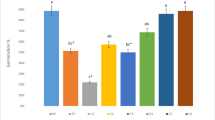
Fungicides are pesticides that are frequently used in agriculture because of their action against fungal diseases. However, the widespread application of pesticides around the world raises environmental and public health concerns, since these compounds are toxic and can pose risks to ecosystems and human health. The aim of this study was to evaluate the phytotoxic, cytogenotoxic, and biochemical effects of azoxystrobin and carbendazim on Lactuca sativa L. and their physiological effects on Phaseolus vulgaris L. by analyzing the cell cycle and chromosomal and nuclear alterations in L. sativa; the biochemical effects of azoxystrobin and carbendazim on Phaseolus vulgaris L. and their physiological effects on Phaseolus vulgaris L. by analyzing the cell cycle and chromosomal and nuclear alterations in L. sativa; the biochemical effects by analyzing the activity of antioxidant enzymes in L. sativa; and the physiological effects by analyzing chlorophyll content and chlorophyll a fluorescence in P. vulgaris. It was observed that both fungicides were phytotoxic and cytotoxic, reducing root growth and the mitotic index, cytogenotoxic, increasing the occurrence of chromosomal alterations, as well as inducing oxidative stress and an increase in chlorophyll fluorescence emission and altered energy absorption in the plants used as a test system. In view of this, studies such as the one presented here indicate that the use of pesticides, even in small quantities, can lead to damage to the metabolism of plant organisms.






Similar content being viewed by others
Data availability
The authors make data and materials available.
References
Agrolink (2024) Bula Elatus®. Disponível em: www.agrolink.com.br. Acesso em: 20 de maio de 2024
Aebi H (1984) Catalase in vitro. In: Methods in enzymology. Acad Press 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Andrade LF, Davide LC, Gedraite LS (2010) The effect of cyanide compounds, fluorides, aluminum, and inorganic oxides present in spent pot liner on germination and root tip cells of Lactuca sativa. Ecotoxicol Environ Saf 73(4):626–631. https://doi.org/10.1016/j.ecoenv.2009.12.012
Andrade-Vieira LF, Andrade-Vieira LF, Botelho CM, Laviola BG, Palmieri MJ, Praça-Fontes M (2014) Effects of Jatropha curcas oil in Lactuca sativa root tip bioassays. An Acad Bras Ciênc 86(1):373–382. https://doi.org/10.1590/0001-3765201420130041
Anke T (1995) The antifungal strobilurins and their possible ecological role. Can J Bot 73(1):940–945. https://doi.org/10.1139/b95-342
Anvisa. Agência nacional de vigilância sanitária. Programa de análise de resíduos de agrotóxicos em alimentos (PARA). 2019. Disponível em: https://www.gov.br/anvisa/pt-br/centraisdeconteudo/publicacoes/agrotoxicos/publicacoes/programa-de-analise-de-residuos-de-agrotoxicos-relatorio-2017-e-2018.pdf/view. Acesso em: 28 de janeiro de 2023
Anvisa. Agência nacional de vigilância sanitária. Reavaliação toxicológica do ingrediente ativo carbendazim. Agenda Regulatória. 2023. Disponível em: https://www.gov.br/anvisa/pt-br/assuntos/noticias-anvisa/2022/carbendazim-anvisa-concluiu-processo-de-reavaliacao-e-mantem-o-banimento/CarbendazimDICOL.pdf Acesso em: 20 de maio de 2024.
Aragão FB, Queiroz VT, Ferreira A, Costa AV, Pinheiro PF, Carrijo TT, Andrade-Vieira LF (2017) Phytotoxicity and cytotoxicity of Lepidaploa rufogrisea (Asteraceae) extracts in the plant model Lactuca sativa (Asteraceae). Rev Biol Tropical 65(2)
Aragão FB, Duarte ID, Fantinato DE, Galter IN, Silveira GL, Reis GB, Andrade-Vieira LF, Matsumoto ST (2021) Toxicogenetic of tebuconazole based fungicide through Lactuca sativa bioassays. Ecotoxicol Environ Saf 213. https://doi.org/10.1016/j.ecoenv.2021.111985
Bernardes PM, Andrade-Vieira LF, Aragão FB, Ferreira A, Ferreira MSF (2015) Toxicity of difenoconazole and tebuconazole in Allium cepa. Water Air Soil Pollut 226(7):1–11. https://doi.org/10.1007/s11270-015-2462-y
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cassol D, da Silva FSP, Falqueto AR, Bacarin MA (2008) An evaluation of non-destructive methods to estimate totalchlorophyll content. Photosynthetica 46(4):634
de Paula Alves SR, de Francisco ALO, de Carvalho TC (2017) Azospirillum brasilense and nitrogen: action on the physiological potential of wheat seeds. Brazilian Journal of Applied Technology for Agricultural Science/Revista Brasileira de Tecnologia Aplicada nas Ciências Agrárias 10(2)
Demant M, Greulich J, Glatthaar M, Haunschild J, Rein S (2012) Modelling of physically relevant features in photoluminescence images. Energy Procedia 27:247–252
Fatma F, Verma S, Kamal A, Srivastava A (2017) Phytotoxicity of pesticides mancozeb and chlorpyrifos: correlation with the antioxidative defence system in Allium cepa. Physiol Mol Biol Plants 24(1):115–123. https://doi.org/10.1007/s12298-017-0490-3
Fenta BA, Beebe SE, Kunert KJ (2020) Role of fixing nitrogen in common bean growth under water deficit conditions. Food Energy Secur 9(1):e183. https://doi.org/10.1002/fes3.183
Fernandes TCC, Mazzeo DEC, Marin-Morales A (2007) Mechanism of micronuclei formation in polyploidizated cells of Allium cepa exposed to trifluralin herbicide. Pestic Biochem Physiol 88(3):252–259. https://doi.org/10.1016/j.pestbp.2006.12.003
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48(12):909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
INCA (2021) Ambiente, trabalho e câncer: aspectos epidemiológicos, toxicológicos e regulatórios. Rio de Janeiro
Iwaniuk P, Lozowicka B (2022) Biochemical compounds and stress markers in lettuce upon exposure to pathogenic Botrytis cinerea and fungicides inhibiting oxidative phosphorylation. Planta 255(3):61. https://doi.org/10.1007/s00425-022-03838-x
Jiang J, Si Y, Yu R, Chen L, Zhao X (2018) Biological response of zebrafish after short-term exposure to azoxystrobin. Chemosphere 202:56–64. https://doi.org/10.1016/j.chemosphere.2018.03.055
Koehler AM, Shew HD (2017) Field efficacy and baseline sensitivities of fungicides used for the management of Septoria leaf spot of stevia. Crop Prot 109:95–101
Koshiba T (1993) Cytosolic ascorbate peroxidase in seedlings and leaves of maize (Zea mays). Plant Cell Physiol 34(5):713–721. https://doi.org/10.1093/oxfordjournals.pcp.a078474
Kuchy AH, Wani AA, Kamili AN (2016) Cytogenetic effects of three commercially formulated pesticides on somatic and germ cells of Allium cepa. Environ Sci Pollut Res 23:6895–6906. https://doi.org/10.1007/s11356-015-5912-6
Leme DM, Marin-Morales MA (2009) Allium cepa test in environmental monitoring: a review on its application. Mutation Research/reviews in Mutation Research 682(1):71–81. https://doi.org/10.1016/j.mrrev.2009.06.002
Liu, S., Fu, L., Wang, S., Chen, J., Jiang, J., Che, Z., ... & Chen, G. (2019). Carbendazim resistance of Fusarium graminearum from Henan wheat. Plant Disease, 103(10), 2536–2540. https://doi.org/10.1094/PDIS-02-19-0391-RE
Liu X, Wang X, Zhang F, Yao X, Qiao Z, Deng J, Jiang X (2022) Toxic effects of fludioxonil on the growth, photosynthetic activity, oxidative stress, cell morphology, apoptosis, and metabolism of Chlorella vulgaris. Sci Total Environ 838:156069. https://doi.org/10.1016/j.scitotenv.2022.156069
Mccord JM, Fridovich I (1969) Superoxide dismutase an enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244(22):6049–6055. https://doi.org/10.1016/S0021-9258(18)63504-5
Mello MLS, Vidal BC (1978) A reação de Feulgen. Ciência e Cultura 30:665–676
Meng F, Yang X, Riksen M, Xu M, Geissen V (2021) Growth response of common bean (Phaseolus vulgaris L.) to soil contaminated with microplastics. Sci Total Environ 755:142516. https://doi.org/10.1016/j.scitotenv.2020.142516
Mishra S, Srivastava S, Tripathi RD, Govindarajan R, Kuriakose SV, Prasad MNV (2006) Phytochelatin synthesis and response of antioxidants during cadmium stress in Bacopa monnieri L◊. Plant Physiol Biochem 44(1):25–37. https://doi.org/10.1016/j.plaphy.2006.01.007
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22(5):867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nithyameenakshi S, Jeyaramraja PR, Manian S (2006) Investigations on phytotoxicity of two new fungicides Azoxystrobin and Difenoconazole. Am J Plant Physiol 1:89–98
Rocha LF, Francisco BS, Dutra FB, Teração BS, de Almeida LS, Viveiros E, ... & da Silva JMS (2021) Allelopathic effects of rubber tree (Hevea brasiliensis (Willd. Ex A. Juss.) Müll. Arg.) on the germination and initial growth of lettuce (Lactuca sativa L.). Research, Society and Development, 10(14). https://doi.org/10.33448/rsd-v10i14.21712
Salam AEA, Hussein EH, El-Itriby HA, Anwar WA, Mansour SA (1993) The mutagenicity of Gramoxone (paraquat) on different eukaryotic systems. Mutat Res Genet Toxicol 319(2):89–101. https://doi.org/10.1016/0165-1218(93)90067-N
Silveira GL, Lima MGF, Dos Reis GB, Palmieri MJ, Andrade-Vieria LF (2017) Toxic effects of environmental pollutants: comparative investigation using Allium cepa L. and Lactuca sativa L. Chemosphere 178:359–367. https://doi.org/10.1016/j.chemosphere.2017.03.048
Singh RP, Sharad S, Kapur S (2004) Free radicals and oxidative stress in neurodegenerative diseases: relevance of dietary antioxidants. J Indian Acad Clin Med 5(3):218–225
Sousa SM, Silva PS, Viccini LF (2010) Cytogenotoxicity of Cymbopogon citratus (DC) Stapf (lemon grass) aqueous extracts in vegetal test systems. An Acad Bras Ciênc 82:305–311. https://doi.org/10.1590/S0001-37652010000200006
Souza AKR, Morassuti CY, de Deus WB (2018) Environmental pollution by heavy metals and the use of plants as bioindicators. Ata Biomedica Brasiliensia 9(3):95–106
Taiz L, Zeiger E (2017) Fisiologia e desenvolvimento vegetal, 6th edn. Artmed Editora, Rio de Janeiro
Truta E, Capraru G, Zamfirache MM, Asaftei M, Toma C, Olteanu Z, Ivanescu L (2010) Estimation of genotoxic potential of carbendazim in fenugreek. Studia Universitatis "Vasile Goldis" Arad. Seria Stiintele Vietii (Life Sciences Series) 20(2):39
Türkoğlu Ş (2012) Determination of genotoxic effects of chlorfenvinphos and fenbuconazole in Allium cepa root cells by mitotic activity, chromosome aberration, DNA content, and comet assay. Pestic Biochem Physiol 103(3)224–230. https://doi.org/10.1016/j.pestbp.2012.06.001
Verma S, Srivastava A (2018) Morphotoxicity and cytogenotoxicity of pendimethalin in the test plant Allium cepa L.-A biomarker based study. Chemosphere 206:248–254. https://doi.org/10.1016/j.chemosphere.2018.04.177
White CC, Viernes H, Krejsa CM, Botta D, Kavabagg DT (2003) Fluorescence-based microtiter plate assay for glutamate-cysteine ligase activity. Anal Biochem 318:175–180. https://doi.org/10.1016/S0003-2697(03)00143-X
Yao J, Sun D, Cen H, Xu H, Weng H, Yuan F, He Y (2018) Phenoty** of Arabidopsis drought stress response using kinetic chlorophyll fluorescence and multicolor fluorescence imaging. Front Plant Sci 9:603. https://doi.org/10.3389/fpls.2018.00603
Zhang LZ, Wei N, Wu QX, ** ML (2007) Anti-oxidant response of Cucumis sativus L. to fungicide carbendazim. Pestic Biochem Physiol 89(1):54–59. https://doi.org/10.1016/j.pestbp.2007.02.007
Zhang F, Lv X, Jia H, Huang C, Wei J, Ding Z, Wang J (2022) Toxicity of the novel fungicide oxathiapiprolin to Chlorella vulgaris: assessments at different levels of biological organization. Chemosphere 291:132752. https://doi.org/10.1016/j.chemosphere.2021.132752
Zhu YY, Liang XY, Li YJ, Duan YB, Zhou MG (2018) F240 of β2-tubulin explains why Fusarium graminearum is less sensitive to carbendazim than Botrytis cinerea. Phytopathology 108:352–361. https://doi.org/10.1094/PHYTO-09-17-0295-R
Funding
Research grant funded by the Coordination for the Improvement of Higher Education Personnel (CAPES).
Author information
Authors and Affiliations
Contributions
The author Kalia Dável Grecco performed the cytogenetic analysis and study design; the authors Kristian Rodolfo Santos, Mylena Boeque Lascola, and Sara Nascimento dos Santos contributed to the writing and revision of the article; the author Francielen Barroso Aragão contributed to the cytogenetic analyses and experimental setup; the authors Iasmini Nicoli Galter, Enzo Zini Moreira Silva, Iara da Costa Souza and Marisa Narciso Fernandes contributed with the biochemical analyses; the authors Juliana Lima Trindade and Diolina Moura Silva contributed with the physiological analyses, and the author Silvia Tamie Matsumoto provided all the support and guidance in the study.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
All authors have consented to participation.
Consent for publication
All authors have consented to publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Gangrong Shi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Grecco, K.D., Santos, K.R., Aragão, F.B. et al. Toxicogenetic, biochemical, and physiological effects of azoxystrobin and carbendazim fungicides over Lactuca sativa L. and Phaseolus vulgaris L. Environ Sci Pollut Res (2024). https://doi.org/10.1007/s11356-024-34013-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-024-34013-2