Abstract
Some previous studies had linked air pollutants and greenness to the risk of death from tuberculosis (TB). Only a few studies had examined the effect of particulate matter (PM2.5) on the mortality of TB, and few studies had assessed the impact and interaction of multiple air pollutants and greenness on the mortality of newly treated TB patients. The study included 29,519 newly treated TB patients from three cities in Anhui province. We collected meteorological data and five pollutants data from The National Meteorological Science Center and air quality monitoring stations. Greenness data were generated by remote sensing inversion of medium-resolution satellite images. We geocoded each patient based on the residential address to calculate the average exposure to air pollutants and the average greenness exposure for each patient during treatment. The Cox proportional risk regression model was used to evaluate the effects of air pollutants and greenness on mortality in newly treated tuberculosis patients. Our results found that the higher the concentration of air pollutants in the living environment of newly treated TB patients, the greater the risk of death: HR 1.135 (95% CI: 1.123–1.147) and HR 1.333 (95% CI: 1.296–1.370) per 10 μg/m3 of PM2.5 and SO2, respectively. Greenness reduced the mortality among newly treated TB patients: HR for NDVI exposure 0.936 (95% CI: 0.925–0.947), HR for NDVI_250m exposure 0.927 (95% CI: 0.916–0.938), and HR for NDVI_500m exposure 0.919 (95% CI: 0.908–0.931). Stratifying the cohort by median greenness exposure, HRs for air pollutants were lower in the high greenness exposure group. Mortality in newly treated TB patients is influenced by air pollutants and greenness. Higher green exposure can mitigate the effects of air pollution. Improving air quality may help reduce mortality among newly treated TB patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tuberculosis (TB) is a chronic infectious disease caused by mycobacterium tuberculosis (MTB), which is one of the oldest diseases in the world. Mycobacterium tuberculosis can invade various organs of the human body, but mainly invades the lung. Globally, TB is the 13th leading cause of death and the second leading infectious killer after COVID-19 (ahead of HIV/AIDS). In 2020, 1.5 million people died from TB (including 214,000 people living with HIV). In 2020, an estimated 10 million people worldwide are living with TB, including 5.6 million men, 3.3 million women, and 1.1 million children. TB is present in all countries and age groups. China has the second-highest number of TB patients in the world, accounting for 8.5% of the global total. The World Health Organization (WHO) estimates that an uncured active TB patient can infect 10 to 15 people in a year through close contact (WHO 2021).
Many previous studies have shown that air pollution plays a role in the development of TB (Kim 2014; Rivas-Santiago et al. 2015; Li et al. 2019). A meta-analysis has shown that long-term exposure to biomass smoke and ambient air pollution in adults is associated with an increased risk of respiratory infections such as chronic obstructive pulmonary disease and tuberculosis (Kc et al. 2018). Tobacco smoke consists of a large number of compounds that are also present in air pollutants. Many studies have shown that smoking tobacco can lead to an increased risk of contracting TB, develo** active TB, and even worse treatment outcomes such as increased mortality (Lin et al. 2007; Slama et al. 2007; Jee et al. 2009; Horne et al. 2012; Maciel et al. 2013). A series of time-series studies have shown a link between air pollution and TB risk with mixed results. The relationship between six common air pollutants and the risk of tuberculosis outpatient visits was examined in a time-series study in Fuyang, China. The results showed that exposure to fine particulate matter PM2.5 (particulate matter < 2.5 μm in aerodynamic diameter), fine particulate matter PM10 (particulate matter < 10 μm in aerodynamic diameter), ozone (O3), nitrogen dioxide (NO2), and carbon monoxide (CO) increased the risk of outpatient visits, and only sulfur dioxide (SO2) had a certain protective effect (Wang et al. 2022a, b). Another study (Huang et al. 2020) also found that exposure to air pollutants PM2.5, NO2, SO2, and O3 was associated with the risk of TB outpatient visits, and a subgroup analysis showed that seasonal variations may have an even greater impact on the risk. A link between air pollutants and TB has also been found in several multi-city studies. A multi-city study in China found that short-term exposure to SO2 reduced the risk of TB, and that exposure had a greater impact on farmers and workers (Wang et al. 2022a, b). A study about the impact of particulate air pollution on TB in seven major Korean cities found that one standard deviation increase in PM10 exposure over 6 years was associated with a 1.2 times higher TB notification rate (Kim et al. 2020). A Chinese study in Bei**g and Hong Kong found a positive correlation between outdoor PM2.5 exposure and seasonal changes in tuberculosis incidence (You et al. 2016). Although many studies have examined the relationship between air pollution and TB, most focused on the incidence of TB and the risk of TB hospital admission. Only a few studies have looked at the effect of ambient air pollution exposure on TB treatment outcomes. A cohort study of tuberculosis in China (Peng et al. 2017) found that long-term exposure to PM2.5 was significantly associated with tuberculosis mortality (aHR = 1.46, 95% CI: 1.15–1.85). Another TB cohort study found that traffic-related air pollution was associated with increased all-cause mortality among active TB patients being treated in California, USA (Blount et al. 2017). The effect of air pollution on TB mortality needs further study.
Environmental green exposure as a potential natural indicator is considered to have three general functions, which are reducing the harm from exposure to noise, high temperature, and air pollution; improving resilience; and promoting healthy activities (Markevych et al. 2017). Several studies have shown an association between exposure to green space and respiratory mortality (James et al. 2016; Crouse et al. 2017; Ji et al. 2019). Green exposure may contribute to the therapeutic rehabilitation of TB patients. A cohort study on the effect of green exposure on all-cause mortality in patients with multidrug-resistant tuberculosis (MDR-TB) treatment in Zhejiang, China, found that green exposure reduced mortality in patients with MDR-TB in areas with lower nighttime light (Ge et al. 2021a, b). A population-based cohort study in Zhejiang and Ningxia, China, finally found that PM2.5 exposure increased mortality among patients with multidrug-resistant tuberculosis, but patients could benefit from green exposure by reducing the effects of PM2.5 (Ge et al. 2021a, b). Although several studies have assessed the effects of greenness exposure, no studies have examined whether green space exposure affects treatment outcomes in newly treated TB patients.
As one of the most populous countries in the world, China is also a country with a high burden of TB, with a rising incidence and mortality of TB. Better treatment and management of newly treated TB patients to reduce secondary transmission and relapse can effectively reduce the TB burden. Based on a cohort of newly treated TB patients in Anhui province, we aimed to investigate the combined effects of air pollutants and greenness on newly treated TB patients.
Materials and methods
Study location
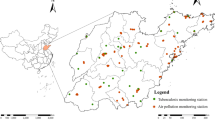
Anhui province is located in eastern China, between 114°54′–119°37′ east longitude and 29°41′–34°38′ north latitude. It covers an area of 140,100 km2. According to the latest census data, Anhui province has 61.3 million permanent residents, ranking among the top 10 in China. Anhui province spans the Yangtze River, Huai River, and **n’an River, with the Huai River basin covering 67,000 km2, the Yangtze River basin 66,000 km2, and the **n’an River basin 60,500 km2. The Yangtze River flows through central and southern Anhui with a total length of 416 km, while the Huai River flows through northern Anhui with a total length of 430 km. Anhui is located in the transition region of warm temperate zone, with the Huai River as the dividing line. The north of Anhui is warm temperate semi-humid monsoon climate, and the south is subtropical humid monsoon climate. The main characteristics are mild climate, plenty of sunshine, obvious monsoon, and four distinct seasons. The annual average temperature of the province is 14–16 °C, with a north–south difference of about 2 °C. The annual average sunshine is 1800–2500 h, average frost-free period 200–250 days, and average precipitation 800–1600 mm. This study takes the Huai River and the Yangtze River as the boundary, and selects Huainan, Hefei, and Huangshan as the research areas. Cement industry is one of the most important sources of industrial pollutant discharge in Anhui province. According to estimates, in 2018, SO2 and NO2 emissions from Anhui’s cement industry were 13,000 tons and 140,000 tons, respectively, accounting for 10.7% and 45.9% of the total emissions from major industries in the province, respectively. Nitrogen oxides are precursors to the formation of PM2.5 and ozone and have a direct impact on air quality. At the same time, automobile exhaust is one of the main sources of air pollutants. According to the report of the Anhui Statistical Yearbook 2022, Hefei has 61.13 million permanent residents and 59.39% of the urban population; Huainan has a permanent population of 3.04 million, with 61.91% of the urban population; the permanent population of Huangshan city is 1.332 million, and the urban population accounts for 59.25%. The number of days with air quality reaching or better than grade II is 86% in Hefei, 74.8% in Huainan, and 99.7 in Huangshan city, indicating that the air quality of Huangshan city is better. The number of medical and health institutions in the three cities is as follows: Hefei city 3543, Huainan city 1657, Huangshan city 1108.
Object of the study
The object of the study was a newly treated patient, which was first discovered and did not receive any anti-tuberculosis drug treatment, or after the discovery of irregular, unreasonable treatment, but the course of treatment does not exceed 1 month.
Study cohort
All newly treated TB patients reported from January 1, 2015, to December 31, 2020, in the three cities were eligible for inclusion in this study. These cases were reported by the Centers for Disease Control and Prevention in the three cities, and the data were recorded and collected by the provincial Tuberculosis Prevention and Control Institute. Immigrant patients were excluded from the study because they might return home for treatment and we could not collect information on where they lived and when they were treated. We also excluded patients with incomplete information, such as no residential address and incomplete treatment date records.
Environmental exposure assessment
Assessment of exposure to air pollutants
Our air pollutants’ concentration data came from the China Air Quality Online Monitoring and Analysis platform (https://www.aqistudy.cn/historydata/). We collected the data of the three cities from January 1, 2015, to December 31, 2020, including PM10, SO2, PM2.5, NO2 (μg/m3, 24 h), and O3 (μg/m3, 8 h). We geocoded the detailed address of each patient and assigned them exposure estimates based on data from contaminant sites.
Assessment of exposure to greenness
In this study, the normalized difference vegetation index (NDVI) was used as the evaluation index of green exposure. Geocoding was carried out through address information to obtain the latitude and longitude coordinates. The 2015–2020 NDVI monthly distribution data product of Anhui province is generated and processed by remote sensing inversion on the basis of medium-resolution satellite images, with a spatial resolution of 250 m. We combined cloud-free images from each summer to create an image covering the entire province of Anhui and selected a map of green space density in our study area (Fig. 1). The range of NDVI value is between − 1 and 1, and the negative value indicates that the land cover is water, cloud, etc., while 0 stands for bare earth, rock, etc. A positive value represents healthy vegetation, and a larger value represents a higher greenness (Klompmaker et al. 2019). We set the negative value of NDVI to 0. We calculated the mean monthly NDVI exposure values for patients during treatment and the mean monthly exposure allocated within the 250-m and 500-m buffer zones of residential addresses. The calculation formula of NDVI is as follows:
NIR is the pixel value of the infrared band, and R is the pixel value of the red band (Li et al. 2022).
All pollutant exposure matching, NDVI calculation, and geographical distribution processing were performed on ArcGIS software (version 10.8).
Individual and environmental covariates
We collected three types of potential covariates: basic individual characteristics, patient type and treatment management style, and environmental factors. Individual characteristics of patients include sex, age, and occupation. The patient type includes the source of patients, diagnostic classification of patients, whether patients are severe, and whether patients are detainees. The environmental factors include working environment, region, mean temperature (MT), and relative humidity (RH). Meteorological data (MT and RH) were obtained from the National Center for Meteorological Science (http://data.cma.cn/), and we estimated the mean temperature and mean relative humidity of the patients during treatment using geocoded residential addresses.
Statistical analysis
The number and proportion of all newly treated TB patients were stratified according to individual characteristics, source of patients, environmental factors, and so on. A simple descriptive statistical analysis was performed on environmental exposure variables including air pollutants, greenness exposure, and meteorological data. The correlations among air pollutant individual exposure concentrations and environmental exposure were described by the Spearman correlation coefficient and scatter plot.
We used Cox proportional risk regression models to assess the relationship between persistent exposure to air pollutants and green exposure and mortality. Covariates with p < 0.2 in the univariate Cox model were included in the multivariate Cox proportional risk regression model (Ge et al. 2021a, b). HRs for environmental exposure variables are reported as fixed increments of 10 μg/m3 for air pollutants and fixed increments of per unit for greenness exposure. We conducted three kinds of modeling for the multivariate Cox proportional risk regression model: Model 1, adjusted for the individual characteristics of the patients including sex, age, and occupation; Model 2, Model 1, plus patient type (source of patients, diagnostic classification of patients, whether patients are severe, and whether patients are detainees) and treatment management style; Model 3, Model 2, plus environmental factors including working environment, region, mean temperature, and relative humidity. We evaluate the models’ proportional risk assumptions by visually examining fractional residuals corresponding to the event time. Based on the main model (Model 3), we stratified the entire cohort according to the median greenness exposure to investigate whether the impact of air pollution exposure on the treatment outcome of newly treated TB patients varies with the change of greenness exposure.
Three sensitivity analyses were performed: (1) Stratified analyses of the entire cohort were performed to explore the correction effects of age, sex, and region in Model 3. (2) NO2 exposure was adjusted on the basis of Model 3, and the potential impact between exposures was assessed through the bi-exposure models. (3) Patients enrolled in the cohort after the outbreak of COVID-19 were excluded.
Finally, we used restricted cubic splines (RCS) to determine the exposure–response relationship between environmental exposure and mortality. All analyses were performed through the “survival” package in R software (version 4.0.0).
Results
We followed 29,519 newly treated TB patients between January 1, 2015, and December 31, 2020. There were 17,747 person-years used in the analysis, with a mean follow-up of 219 days, and 369 people died during treatment (1.25%).
Descriptive results
Table 1 and Table 2 provide the results of the simple descriptive statistical analysis of air pollutants, greenness exposures, meteorological indicators, and newly treated TB patients. The average patient exposure concentration of PM2.5 was 48.03 μg/m3 (12.61–103.35 μg/m3), PM10 was 76.85 μg/m3 (25.03–142.22 μg/m3), SO2 was 11.28 μg/m3 (4.05–37.88 μg/m3), O3 was 111.91 mg/m3 (40.43–173.54 μg/m3), and NO2 was 34.85 μg/m3 (7.65–80.75 μg/m3). The average concentrations of PM2.5 and PM10 were between the level 1 and level 2 daily concentration limits for ambient air pollutants in the national environmental quality standards. The average concentration of O3 is between the 8-h average level 1 concentration limit and level 2 concentration limit. The average concentrations of SO2 and NO2 were below the average daily level 1 concentration limit. The minimum and maximum NDVI were 0.011 and 0.836, respectively. The minimum value of NDVI_250m was 0.148, and the maximum was 0.872. The minimum and maximum NDVI_500m were 0.011 and 0.910, respectively. The minimum value of the mean temperature was 3.99 °C, and the maximum was 28.20 °C. The minimum value of the relative humidity was 65.84%, and the maximum was 86.92%.
A total of 29,519 newly treated TB patients were included in this cohort, including 20,937 males and 8582 females. 72.2% of patients were younger than 65 years old. 60.5% of patients worked indoors. 24,305 patients worked in labor-intensive jobs. A total of 9211 patients lived in urban areas and 20,308 in rural areas. Hefei had the highest number of patients, while Huangshan had the least. A minority (1.4%) of patients were detainees. 84.5% of patients were on transfer treatment. 92.3% of patients were III TB patients. A total of 540 patients were seriously ill with TB. Among all patients, 20,450 patients received entire supervision, 8965 patients received intensive supervision, and 97 patients were self-medicated. The results of the entire cohort stratified by green exposure are shown in Table 2. As shown in Fig. 2, we found significant correlations between the two particulate pollutants (|r|= 0.90) and between the three greenness exposure indicators.
The Spearman rank correlation coefficients and scatter plot between air pollutant individual exposure concentrations and environmental exposure. PM2.5, particulate matter < 2.5 μm in aerodynamic diameter; PM10, particulate matter < 10 μm in aerodynamic diameter; SO2, sulfur dioxide; O3, ozone; NO2, nitrogen dioxide; NDVI, normalized difference vegetation index; NDVI_250m, normalized difference vegetation index in the 250-m buffer; NDVI_500m, normalized difference vegetation index in the 500-m buffer; MT, mean temperature; RH, relative humidity. *rs > 0.7
Effects of air pollutants and greenness on mortality in newly treated TB patients
We used Cox proportional risk regression models and fitted three models to assess the impact of air pollutants and greenness exposure on mortality (see the “Materials and methods” section for details). Overall, an increase of 10 μg/m3 in the exposure concentration of various air pollutants had a positive and statistically significant effect on mortality. The results from Model 3 found that exposure to PM2.5 was associated with increased mortality (HR = 1.135, 95% CI: 1.123–1.147). The HR for PM10 exposure is 1.182 (95% CI: 1.173–1.147), the HR for O3 exposure is 1.034 (95% CI: 1.028–1.040), and the HR for SO2 exposure is 1.333 (95% CI: 1.296–1.370). We observed that greenness exposure reduced mortality. The results from Model 3 found that exposure to NDVI associated with a reduction in mortality (HR = 0.936, 95% CI: 0.925–0.947). The HR for NDVI_250m exposure is 0.927 (95% CI: 0.916–0.938), and the HR for NDVI_500m exposure is 0.919 (95% CI: 0.908–0.931). The results of each model are shown in Table 3.
Effects of air pollution on mortality in newly treated TB patients stratified by greenness exposure across the full cohort
We stratified the entire cohort according to greenness exposure and used Model 3 to assess whether the impact of exposure to various air pollutants on mortality was affected by different greenness. From Fig. 3 and Supplementary Table S1, we can see that the impact of air pollutants on mortality was low during high green exposure. For example, after stratification of the cohort by median NDVI, the HRs of PM2.5 exposure on mortality were 1.139 (95% CI: 1.122–1.155) and 1.121 (95% CI: 1.103–1.140) in the low exposure group and the high exposure group, respectively. After stratification of the cohort by median NDVI_250m, the HRs of SO2 exposure on mortality were 1.352 (95% CI: 1.300–1.406) and 1.308 (95% CI: 1.256–1.362) in the low exposure group and the high exposure group, respectively.
Hazard ratios and 95% CI of per 10 µg/m3 increase in air pollutant exposure associated with mortality among newly treated patients with tuberculosis in the full cohort after being stratified by greenness exposure. Cox models are adjusted for covariates: sex, age, occupation, source of patients, diagnostic classification of patients, whether patients are severe, whether patients are detainees, treatment management style, working environment, region, mean temperature, and relative humidity. PM2.5, particulate matter < 2.5 μm in aerodynamic diameter; PM10, particulate matter < 10 μm in aerodynamic diameter; SO2, sulfur dioxide; O3, ozone; NDVI, normalized difference vegetation index; NDVI_250m, normalized difference vegetation index in the 250-m buffer; NDVI_500m, normalized difference vegetation index in the 500-m buffer. aLow exposure is defined as from the minimum to the median. bHigh exposure is defined as from the median to maximum
Sensitivity analysis
Effects of air pollutants and greenness on mortality in newly treated TB patients in different sex, age, and region groups
We stratified the entire cohort by age, sex, and region, and then analyzed the independent effects of air pollutant exposure and greenness exposure on mortality in different subgroups. We found that exposure to air pollutants was more harmful for patients living in urban areas, elderly patients (age ≥ 65 years old), and men, with the exception of SO2. Living in areas with higher greenness exposure may be more protective for male patients, younger patients (age < 65 years old), and patients living in urban areas (Fig. 4; Supplementary Table S2).
Hazard ratios and 95% CI of per 10 µg/m3 increase in air pollutant exposure and per 0.1 µg/m3 increase in greenness exposure associated with mortality among newly treated patients with tuberculosis stratified by sex, age, and region. Cox models are adjusted for covariates: sex, age, occupation, source of patients, diagnostic classification of patients, whether patients are severe, whether patients are detainees, treatment management style, working environment, region, mean temperature, and relative humidity. PM2.5, particulate matter < 2.5 μm in aerodynamic diameter; PM10, particulate matter < 10 μm in aerodynamic diameter; SO2, sulfur dioxide; O3, ozone; NDVI, normalized difference vegetation index; NDVI_250m, normalized difference vegetation index in the 250-m buffer; NDVI_500m, normalized difference vegetation index in the 500-m buffer
Bi-exposure models
Based on Model 3, NO2 exposure was added to fit the bi-exposure model. We found that there was no significant change in the results before and after NO2 inclusion (Table 3; Supplementary Fig. S1 and Supplementary Table S3).
Excluding patients after the outbreak of COVID-19
In Supplementary Table S4, we found that the results of the three models did not change substantially after removing the patients from the cohort after the outbreak of COVID-19, indicating that the outbreak of COVID-19 had no significant impact on the results of the study.
Exposure–response curves
As shown in Supplementary Fig. S2 and Supplementary Fig. S3, the exposure–response curves suggested a non-linear association between environmental exposure (air pollutants and greenness) and mortality in newly treated TB patients. Greenness exposure was negatively associated with the risk of death in newly treated TB patients, while air pollutants were positively associated with the risk of death.
Discussion
To our knowledge, this is the first study on the relationship between environmental exposure (air pollutants and greenness) and mortality in newly treated TB patients in China. Our study found that air pollutant exposure and greenness exposure were associated with mortality during treatment in newly treated TB patients. Exposure to air pollutants increased mortality in newly treated TB patients, while greenness exposure was associated with reduced mortality in newly treated TB patients. Higher levels of greenness exposure may reduce the adverse effects of air pollutants on TB patients. Our results suggest that overall environmental exposure has a greater impact on male patients and urban patients.
After adjusting for confounding factors such as general individual characteristics, patient types, average temperature, and average relative humidity, we found that all four pollutants had a positive impact on mortality in newly treated TB patients. The results showed that the mortality increased by 13.5% for every 10 μg/m3 increase in PM2.5 exposure, 18.2% for every 10 μg/m3 increase in PM10 exposure, 3.4% for every 10 μg/m3 increase in O3 exposure, and 33.3% for every 10 μg/m3 increase in SO2 exposure.
Some previous studies had also shown a positive impact of air pollutants on TB mortality. A multi-city time-series study in Shandong, China, found that short-term exposure (≤ 30 days) to SO2, CO, and PM2.5 had a significant impact on TB mortality (Liu et al. 2021). A cohort study of drug-resistant TB in China found that every 10 μg/m3 increase in PM2.5 was associated with a 70.2% increase in mortality among drug-resistant patients (Ge et al. 2021a, b). In another tuberculosis cohort study in China, it was found that with each 2.06 μg/m3 increase in PM2.5 exposure, the total mortality of patients increased by 30%, and the tuberculosis mortality increased by 46%. Long-term exposure to PM2.5 had a positive effect on the mortality of patients with tuberculosis (Peng et al. 2017). Previous studies had explored how PM2.5 damages human systems and organs and accelerates the progression of TB (Nel 2005; D’Amato et al. 2010; Ibironke et al. 2019; Popovic et al. 2019). PM2.5 can damage the immune function of the human respiratory system, because PM2.5 can reduce the function of macrophages, cause stress response and oxidation, and reduce the immunity of the lungs. PM2.5 can also damage airway epithelial cells and reduce the body’s immunity to mycobacterium tuberculosis. PM2.5 can also carry some external impurities into the lungs, such as the alveoli and bronchi, which can damage the body’s immune function (Bauer et al. 2012; Laumbach and Kipen 2012; Bai et al. 2018; Cheng et al. 2019; Torres et al. 2019). Some studies suggest that PM10 has a similar damaging effect to PM2.5 (Kim et al. 2020; Pompilio and Di Bonaventura 2020; **ang et al. 2021). As for the harmful effect of SO2, some studies have shown that exposure to SO2 can reduce and hinder the production and release of tumor necrosis factor-α (TNF-α) in vivo. As TNF-α can inhibit mycobacterium tuberculosis in vivo by controlling the formation of granuloma, SO2 can hinder the resistance of the human body to tuberculosis branch rods (Knorst et al. 1996; Mohan et al. 2001). A laboratory study had shown that O3 can damage lung function, affect gas exchange between the lungs and the outside world, and increase airway inflammation (Smith et al. 2016).
This study found that greenness exposure reduced mortality in newly treated TB patients. We found that exposure to NDVI was associated with a reduction in mortality (HR = 0.936), and we calculated greenness exposures for the 250-m and 500-m buffers, the results still show the protective effect. The protective effect of greenness exposure on tuberculosis was consistent with previous studies. A cohort study in China (Ge et al. 2021a, b) found that greenness exposure within a 500-m buffer was stratified by quartile, with patients in the remaining quartile having a lower risk of death than those in the lowest quartile and HR values of less than 1. In the past, people were treated and prevented by breathing fresh air, and some patients were even sent to the mountains for fresh air. One study found that this method of breathing fresh air could reduce the risk of death from tuberculosis (McCarthy 2001). Spending more time in nature is good for human health, and greenness may help TB patients recover better, as living in greener areas may expose them to more fresh air. In addition, studies have shown that higher levels of greenness may be beneficial to mental health. A study conducted in Australia found that people who thought their neighborhoods were greener reported better mental health (Sugiyama et al. 2008). Some studies suggested that greenness can reduce stress and air pollution, which is good for human health. Green can absorb more particle pollutants in the air, and the surface of plants can also absorb some gaseous pollutants, effectively reducing the content of pollutants in the air (Chen et al. 2016; Markevych et al. 2017; Ji et al. 2020). Our study stratified the full cohort according to the median greenness exposure and found that greenness exposure significantly reduced the harmful effects of air pollution, which was consistent with previous studies. A cohort study of MDR-TB in China found that greater greenness exposure resulted in an average 0.188–0.194 reduction in HR associated with PM2.5.
Our data suggested that exposure to greenness and air pollutants had a greater impact on male patients and patients living in urban areas, a conclusion supported by previous studies. A Canadian cohort study found that greenness exposure was more protective against non-accidental death in men than in women (Crouse et al. 2017). A Chinese study also found a greater protective effect of greenness exposure for younger patients (< 60 years old) and patients living in urban areas (Ge et al. 2021a, b). A cohort study in China found that older people living in urban areas tended to be more likely to benefit from a greener environment (Ji et al. 2019). Compared with rural areas, pollutant concentrations were much higher in urban areas, which may be the reason why air pollutants had a greater impact on urban patients. The greater impact of environmental exposure on men may be due to their greater exposure to the outdoors than women and the fact that more men have unhealthy habits such as smoking and drinking. In addition, physiological differences in the airways between men and women may also play a role (Sopori et al. 1998). Our study also found that pollutant exposure had a greater impact on the elderly, which may be due to their decreased body defense ability and lower ability to resist the harm of pollutant exposure than the young (Zhang et al. 2019; Huang et al. 2020).
Our study had some strengths: First, our study was one of the few to evaluate the effects of both air pollutant exposure and greenness exposure on TB, and we were the first cohort study of newly treated TB patients Secondly, our study included all the newly treated TB patients in the three cities from 2015 to 2020, and the three cities were distributed in central Anhui, northern Anhui, and southern Anhui, respectively, with good regional heterogeneity, which made our study to have good representativeness. Finally, our study explored the effects of multiple pollutants. Compared with previous studies, our contents were more abundant and the bi-exposure model was fitted to make our research results more accurate. But our study still had some limitations. First, we used contaminant data from monitoring sites to match patients, which may affect the authenticity of exposure. Due to the very similar local meteorological conditions and geographical location of each city, the heterogeneity of individual exposure was very low, so there was little difference between the underestimated or overestimated value and the real exposure value. Second, we used NDVI and two buffer zones to evaluate greenness exposure, missing the information of vegetation type, so we could only do general greenness exposure, which could not reflect the type and quality of greenness exposure. Finally, smoking status, socioeconomic status, and exposure to indoor pollutants were not included in the study, although we adjusted for multiple covariates. Future studies should consider incorporating these confounding factors.
Conclusions
In this study, air pollutants were positively correlated with mortality in newly treated TB patients, while greenness exposure was negatively correlated with mortality in newly treated TB patients. The association between air pollutants and mortality in newly treated TB patients decreased with increased exposure to greenness space. Overall, air pollutants and greenness had a greater impact on male patients and urban patients. Greenness was more protective for younger patients, while air pollutants were more harmful for older people. Increasing green vegetation and reducing emissions of air pollutants may help reduce mortality among newly treated TB patients.
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Bai L, Su X, Zhao D, Zhang Y, Cheng Q, Zhang H, Wang S, **e M, Su H (2018) Exposure to traffic-related air pollution and acute bronchitis in children: season and age as modifiers. J Epidemiol Community Health 72(5):426–433. https://doi.org/10.1136/jech-2017-209948
Bauer RN, Diaz-Sanchez D, Jaspers I (2012) Effects of air pollutants on innate immunity: the role of Toll-like receptors and nucleotide-binding oligomerization domain-like receptors. J Allergy Clin Immunol. 129 (1), 14–24, 25–26. https://doi.org/10.1016/j.jaci.2011.11.004
Blount RJ, Pascopella L, Catanzaro DG, Barry PM, English PB, Segal MR, Flood J, Meltzer D, Jones B, Balmes J, Nahid P (2017) Traffic-related air pollution and all-cause mortality during tuberculosis treatment in California. Environ Health Persp 125(9):97026. https://doi.org/10.1289/ehp1699
Chen L, Liu C, Zou R, Yang M, Zhang Z (2016) Experimental examination of effectiveness of vegetation as bio-filter of particulate matters in the urban environment. Environ Pollut 208(Pt A):198–208. https://doi.org/10.1016/j.envpol.2015.09.006
Cheng H, Zhu F, Lei R, Shen C, Liu J, Yang M, Ding R, Cao J (2019) Associations of ambient PM2.5 and O3 with cardiovascular mortality: a time-series study in Hefei, China. Int J Biometeorol 63 (10), 1437–1447. https://doi.org/10.1007/s00484-019-01766-2.
Crouse DL, Pinault L, Balram A, Hystad P, Peters PA, Chen H, van Donkelaar A, Martin RV, Menard R, Robichaud A, Villeneuve PJ (2017) Urban greenness and mortality in Canada’s largest cities: a national cohort study. Lancet Planet Health 1(7):e289–e297. https://doi.org/10.1016/s2542-5196(17)30118-3
D’Amato G, Cecchi L, D’Amato M, Liccardi G (2010) Urban air pollution and climate change as environmental risk factors of respiratory allergy: an update. J Investig Allergol Clin Immunol. 20 (2), 95–102, 102
Ge E, Gao J, Ren Z, Liu X, Luo M, Zhong J, Fei F, Chen B, Wang X, Wei X, Peng Y (2021a) Greenness exposure and all-cause mortality during multi-drug resistant tuberculosis treatment: a population-based cohort study. Sci Total Environ 771:145422. https://doi.org/10.1016/j.scitotenv.2021.145422
Ge E, Gao J, Wei X, Ren Z, Wei J, Liu X, Wang X, Zhong J, Lu J, Tian X, Fei F, Chen B, Wang X, Peng Y, Luo M, Lei J (2021b) Effect modification of greenness on PM2.5 associated all-cause mortality in a multidrug-resistant tuberculosis cohort. Thorax. 2020–216819. https://doi.org/10.1136/thoraxjnl-2020-216819.
Horne DJ, Campo M, Ortiz JR, Oren E, Arentz M, Crothers K, Narita M (2012) Association between smoking and latent tuberculosis in the U.S. population: an analysis of the National Health and Nutrition Examination Survey. Plos One. 7 (11), e49050. https://doi.org/10.1371/journal.pone.0049050.
Huang K, Ding K, Yang XJ, Hu CY, Jiang W, Hua XG, Liu J, Cao JY, Zhang T, Kan XH, Zhang XJ (2020) Association between short-term exposure to ambient air pollutants and the risk of tuberculosis outpatient visits: a time-series study in Hefei. China Environ Res 184:109343. https://doi.org/10.1016/j.envres.2020.109343
Ibironke O, Carranza C, Sarkar S, Torres M, Choi H T, Nwoko J, Black K, Quintana-Belmares R, Osornio-Vargas A, Ohman-Strickland P, Schwander S (2019) Urban air pollution particulates suppress human T-cell responses to mycobacterium tuberculosis. Int J Environ Res Public Health. 16 (21). https://doi.org/10.3390/ijerph16214112.
James P, Hart JE, Banay RF, Laden F (2016) Exposure to greenness and mortality in a nationwide prospective cohort study of women. Environ Health Perspect 124(9):1344–1352. https://doi.org/10.1289/ehp.1510363
Jee SH, Golub JE, Jo J, Park IS, Ohrr H, Samet JM (2009) Smoking and risk of tuberculosis incidence, mortality, and recurrence in South Korean men and women. Am J Epidemiol 170(12):1478–1485. https://doi.org/10.1093/aje/kwp308
Ji JS, Zhu A, Bai C, Wu CD, Yan L, Tang S, Zeng Y, James P (2019) Residential greenness and mortality in oldest-old women and men in China: a longitudinal cohort study. Lancet Planet Health 3(1):e17–e25. https://doi.org/10.1016/s2542-5196(18)30264-x
Ji JS, Zhu A, Lv Y, Shi X (2020) Interaction between residential greenness and air pollution mortality: analysis of the Chinese Longitudinal Healthy Longevity Survey. Lancet Planet Health 4(3):e107–e115. https://doi.org/10.1016/s2542-5196(20)30027-9
Kc R, Shukla SD, Gautam SS, Hansbro PM, O’Toole RF (2018) The role of environmental exposure to non-cigarette smoke in lung disease. Clin Transl Med 7(1):39. https://doi.org/10.1186/s40169-018-0217-2
Kim H, Yu S, Choi H (2020) Effects of particulate air pollution on tuberculosis development in seven major cities of Korea from 2010 to 2016: methodological considerations involving long-term exposure and time lag. Epidemiol Health 42:e2020012. https://doi.org/10.4178/epih.e2020012
Kim J (2014) Is ambient air pollution another risk factor of tuberculosis? Korean J Intern Med 29(2):170–172. https://doi.org/10.3904/kjim.2014.29.2.170
Klompmaker JO, Janssen N, Bloemsma LD, Gehring U, Wijga AH, van den Brink C, Lebret E, Brunekreef B, Hoek G (2019) Associations of combined exposures to surrounding green, air pollution, and road traffic noise with cardiometabolic diseases. Environ Health Perspect 127(8):87003. https://doi.org/10.1289/ehp3857
Knorst MM, Kienast K, Muller-Quernheim J, Ferlinz R (1996) Effect of sulfur dioxide on cytokine production of human alveolar macrophages in vitro. Arch Environ Health 51(2):150–156. https://doi.org/10.1080/00039896.1996.9936009
Laumbach RJ, Kipen HM (2012) Respiratory health effects of air pollution: update on biomass smoke and traffic pollution. J Allergy Clin Immunol. 129 (1), 3–11, 12–13. https://doi.org/10.1016/j.jaci.2011.11.021
Li G, Zhang H, Hu M, He J, Yang W, Zhao H, Zhu Z, Zhu J, Huang F (2022) Associations of combined exposures to ambient temperature, air pollution, and green space with hypertension in rural areas of Anhui province, China: a cross-sectional study. Environ Res 204(Pt D):112370. https://doi.org/10.1016/j.envres.2021.112370
Li Z, Mao X, Liu Q, Song H, Ji Y, Xu D, Qiu B, Tian D, Wang J (2019) Long-term effect of exposure to ambient air pollution on the risk of active tuberculosis. Int J Infect Dis 87:177–184. https://doi.org/10.1016/j.ijid.2019.07.027
Lin HH, Ezzati M, Murray M (2007) Tobacco smoke, indoor air pollution and tuberculosis: a systematic review and meta-analysis. Plos Med 4(1):e20. https://doi.org/10.1371/journal.pmed.0040020
Liu Y, Zhao S, Li Y, Song W, Yu C, Gao L, Ran J, He D, Li H (2021) Effect of ambient air pollution on tuberculosis risks and mortality in Shandong, China: a multi-city modeling study of the short- and long-term effects of pollutants. Environ Sci Pollut R 28(22):27757–27768. https://doi.org/10.1007/s11356-021-12621-6
Maciel EL, Brioschi AP, Peres RL, Guidoni LM, Ribeiro FK, Hadad DJ, Vinhas SA, Zandonade E, Palaci M, Dietze R, Johnson JL (2013) Smoking and 2-month culture conversion during anti-tuberculosis treatment. Int J Tuberc Lung Dis 17(2):225–228. https://doi.org/10.5588/ijtld.12.0426
Markevych I, Schoierer J, Hartig T, Chudnovsky A, Hystad P, Dzhambov AM, de Vries S, Triguero-Mas M, Brauer M, Nieuwenhuijsen MJ, Lupp G, Richardson EA, Astell-Burt T, Dimitrova D, Feng X, Sadeh M, Standl M, Heinrich J, Fuertes E (2017) Exploring pathways linking greenspace to health: theoretical and methodological guidance. Environ Res 158:301–317. https://doi.org/10.1016/j.envres.2017.06.028
McCarthy OR (2001Aug) The key to the sanatoria. J R Soc Med 94(8):413–417. https://doi.org/10.1177/014107680109400813
Mohan VP, Scanga CA, Yu K, Scott HM, Tanaka KE, Tsang E, Tsai MM, Flynn JL, Chan J (2001) Effects of tumor necrosis factor alpha on host immune response in chronic persistent tuberculosis: possible role for limiting pathology. Infect Immun 69(3):1847–1855. https://doi.org/10.1128/iai.69.3.1847-1855.2001
Nel A (2005) Atmosphere. Air pollution-related illness: effects of particles. Science 308 (5723), 804–806. https://doi.org/10.1126/science.1108752
Peng Z, Liu C, Xu B, Kan H, Wang W (2017) Long-term exposure to ambient air pollution and mortality in a Chinese tuberculosis cohort. Sci Total Environ 580:1483–1488. https://doi.org/10.1016/j.scitotenv.2016.12.128
Pompilio A, Di Bonaventura G (2020) Ambient air pollution and respiratory bacterial infections, a troubling association: epidemiology, underlying mechanisms, and future challenges. Crit Rev Microbiol 46(5):600–630. https://doi.org/10.1080/1040841x.2020.1816894
Popovic I, Soares MR, Ge E, Marks GB, Dong GH, Wei X, Knibbs LD (2019) A systematic literature review and critical appraisal of epidemiological studies on outdoor air pollution and tuberculosis outcomes. Environ Res 170:33–45. https://doi.org/10.1016/j.envres.2018.12.011
Rivas-Santiago CE, Sarkar S, Cantarella PT, Osornio-Vargas A, Quintana-Belmares R, Meng Q, Kirn TJ, Ohman SP, Chow JC, Watson JG, Torres M, Schwander S (2015) Air pollution particulate matter alters antimycobacterial respiratory epithelium innate immunity. Infect Immun 83(6):2507–2517. https://doi.org/10.1128/iai.03018-14
Slama K, Chiang CY, Enarson DA, Hassmiller K, Fanning A, Gupta P, Ray C (2007) Tobacco and tuberculosis: a qualitative systematic review and meta-analysis. Int J Tuberc Lung Dis 11(10):1049–1061
Smith GS, Van Den Eeden SK, Garcia C, Shan J, Baxter R, Herring AH, Richardson DB, Van Rie A, Emch M, Gammon MD (2016) Air pollution and pulmonary tuberculosis: a nested case-control study among members of a Northern California health plan. Environ Health Perspect 124(6):761–768. https://doi.org/10.1289/ehp.1408166
Sopori ML, Kozak W, Savage SM, Geng Y, Kluger MJ (1998) Nicotine-induced modulation of T Cell function. Implications for inflammation and infection. Adv Exp Med Biol 437:279–289. https://doi.org/10.1007/978-1-4615-5347-2_31
Sugiyama T, Leslie E, Giles-Corti B, Owen N (2008) Associations of neighbourhood greenness with physical and mental health: do walking, social coherence and local social interaction explain the relationships? J Epidemiol Community Health 62(5):e9. https://doi.org/10.1136/jech.2007.064287
Torres M, Carranza C, Sarkar S, Gonzalez Y, Osornio VA, Black K, Meng Q, Quintana-Belmares R, Hernandez M, Angeles GJ, Paramo-Figueroa VH, Iniguez-Garcia MA, Flores JL, Zhang JJ, Gardner CR, Ohman-Strickland P, Schwander S (2019) Urban airborne particle exposure impairs human lung and blood mycobacterium tuberculosis immunity. Thorax 74(7):675–683. https://doi.org/10.1136/thoraxjnl-2018-212529
Wang XQ, Li YQ, Hu CY, Huang K, Ding K, Yang XJ, Cheng X, Zhang KD, Yu WJ, Wang J, Zhang YZ, Ding ZT, Zhang XJ, Kan XH (2022a) Short-term effect of ambient air pollutant change on the risk of tuberculosis outpatient visits: a time-series study in Fuyang. China Environ Sci Pollut Res Int 29(20):30656–30672. https://doi.org/10.1007/s11356-021-17323-7
Wang XQ, Zhao JW, Zhang KD, Yu WJ, Wang J, Li Y Q, Cheng X, Li ZH, Mao YC, Hu CY, Huang K, Ding K, Yang XJ, Chen SS, Zhang XJ, Kan XH (2022b) Short-term effect of sulfur dioxide (SO2) change on the risk of tuberculosis outpatient visits in 16 cities of Anhui Province, China: the first multi-city study to explore differences in occupational patients. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-022-19438-x
**ang K, Xu Z, Hu Y, He Y, Dan Y, Wu Q, Fang X, Pan H (2021) Association between ambient air pollution and tuberculosis risk: a systematic review and meta-analysis. Chemosphere 277:130342. https://doi.org/10.1016/j.chemosphere.2021.130342
You S, Tong YW, Neoh KG, Dai Y, Wang CH (2016) On the association between outdoor PM2.5 concentration and the seasonality of tuberculosis for Bei**g and Hong Kong. Environ Pollut 218:1170–1179. https://doi.org/10.1016/j.envpol.2016.08.071
Zhang Y, Ni H, Bai L, Cheng Q, Zhang H, Wang S, **e M, Zhao D, Su H (2019) The short-term association between air pollution and childhood asthma hospital admissions in urban areas of Hefei city in China: a time-series study. Environ Res 169:510–516. https://doi.org/10.1016/j.envres.2018.11.043
Acknowledgements
We would like to acknowledge the School of Public Health, Anhui Medical University, and Anhui Chest Hospital for their support to this project.
Funding
This study was supported by the National Natural Science Foundation of China (82073565), the National Key Project for Infectious Disease (2018ZX10722301-001–004), and the Major National Science and Technology Projects during the 12th Five-Year Plan period (2013ZX10003008-001–003).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. **n-Qiang Wang and **ao-Hong Kan collaboratively designed the study, both making substantial intellectual contribution. **n-Qiang Wang, Kang-Di Zhang, and Wen-Jie Yu analyzed the data and drafted the manuscript. **ao-Hong Kan and **u-Jun Zhang revised the manuscript. Jia-Wen Zhao, Cheng-Yang Hu, and Kai Huang contributed in collecting the data. **n-Qiang Wang, Kang-Di Zhang, and Wen-Jie Yu contributed equally. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Anhui Medical University Ethics Committee. All patient information included in the study was unidentified and anonymous.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, XQ., Zhang, KD., Yu, WJ. et al. Associations of exposures to air pollution and greenness with mortality in a newly treated tuberculosis cohort. Environ Sci Pollut Res 30, 34229–34242 (2023). https://doi.org/10.1007/s11356-022-24433-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24433-3