Abstract
Introduction
Coronavirus disease 2019 (COVID-19) is strongly linked to dysregulation of various molecular, cellular, and physiological processes that change abundance of different biomolecules including metabolites that may be ultimately used as biomarkers for disease progression and severity. It is important at early stage to readily distinguish those patients that are likely to progress to moderate and severe stages.
Objectives
This study aimed to investigate the utility of saliva and plasma metabolomic profiles as a potential parameter for risk stratifying COVID-19 patients.
Method
LC–MS/MS-based untargeted metabolomics were used to profile the changes in saliva and plasma metabolomic profiles of COVID-19 patients with different severities.
Results
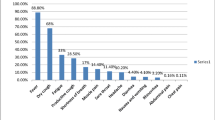
Saliva and plasma metabolites were screened in 62 COVID-19 patients and 18 non-infected controls. The COVID-19 group included 16 severe, 15 moderate, 16 mild, and 15 asymptomatic cases. Thirty-six differential metabolites were detected in COVID-19 versus control comparisons. SARS-CoV-2 induced metabolic derangement differed with infection severity. The metabolic changes were identified in saliva and plasma, however, saliva showed higher intensity of metabolic changes. Levels of saliva metabolites such as sphingosine and kynurenine were significantly different between COVID-19 infected and non-infected individuals; while linoleic acid and Alpha-ketoisovaleric acid were specifically increased in severe compared to non-severe patients. As expected, the two prognostic biomarkers of C-reactive protein and D-dimer were negatively correlated with sphingosine and 5-Aminolevulinic acid, and positively correlated with l-Tryptophan and l-Kynurenine.
Conclusion
Saliva disease-specific and severity-specific metabolite could be employed as potential COVID-19 diagnostic and prognostic biomarkers.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The global outbreak coronavirus disease 2019 (COVID-19) viral pneumonia caused by severe acute respiratory syndrome corona virus 2 (SARS-CoV-2), which originated in Wuhan, China, in December 2019 has become a worldwide pandemic claiming several thousands of lives worldwide. According to last update 4th of October 2021 of John Hopkins- Coronavirus Resource Center (https://coronavirus.jhu.edu/map.html), this virus has infected more than 234,987,662 people worldwide and 4,803,300 confirmed deaths. Despite the fact that more than 80% of COVID-19 patients have mild symptoms, such as anosmia (loss of smell) and ageusia (loss of taste) and in some cases accompanied with other more moderate symptoms including fever, exhaustion, cough, and shortness of breath (Hui et al., 2020; Yang et al., 2.3 Statistical analysis Metabolite levels were normalized using the Bioconductor package limma-voom and presented as log2 (Ritchie et al., 2015a). Metabolites normality was confirmed using Shapiro–Wilk test and Kolmogorov–Smirnov test. Log-transformed normalized intensities were used in LIMMA analyses to identify differential changes in metabolites between diseased and control groups. We used the default Benjamini–Horchberg correction for multiple testing (Dudoit et al., 2002; Smyth Gordon, 2004). One-way analysis of variance (ANOVA) and post hoc Tukey multiple comparison analyses were applied to compare between more than two independent variables. Correlation of sphingosine, 5-Aminolevulinic acid, l-Tryptophan, and l-Kynurenine saliva concentrations of COVID-19 patients and serum markers of COVID-19 severity such as D-dimer and C-reactive protein was evaluated using Pearson’s coefficient test with two-tailed p-value < 0.05 considered significant. Principal component analysis (PCA) was implemented using PCAGO R Shiny framework which is available at github.com/hoelzer-lab/pcagom. jHeatmap was used to create the heatmaps which is available at http://jheatmap.github.io/jheatmap/ (Deu-Pons et al., 2014). Metabolite Set Enrichment Analysis (MSEA) were carried using MetaboAnalystR3 package which is available at github.com/xia-lab/MetaboAnalystR (Pang et al., 2020; Liao et al., 2020; Vardhana & Wolchok, 2020).Thereby it is expected that metabolite reprogramming observed in severe COVID-19 disease to correlate to this immune dysregulation. Following this assumption, **ao and colleague studied the plasma metabolite programming during COVID-19 infection (**ao et al., 2021).They revealed strong correlation between metabolite dysregulation and hyperinflammation found in severe COVID-19 (**ao et al., 2021). Further, they confirmed this association by modulating viral induced inflammation using metabolite supplements (**ao et al., 2021). In another study, kynurenic acid to Kynurenine ratio, specifically in males was positively associated with age, inflammatory marker levels and negatively associated with T cell immune response during COVID-19 (Thomas et al., 2020). Kynurenine has a central role in modulating inflammation and immune response (Chen & Guillemin, 2009). Kynurenine shunt that moves the tryptophan degradation toward kynurenine and away from serotonin has been associated independently with depression, fatigue, muscle weakness, and other neurologic diseases (Mangoni, 1974; Németh et al., 2005). COVID-19 disease causes long-lasting symptoms months after recovery from acute infection that resembles kynurenine shunt presentations (Eroğlu et al., 2021; Huang et al., 2021). It is therefore possible for the metabolic programming to play a role in manifestation of both acute and long-term symptoms observed in COVID-19 (Huang et al., 2021; Ramakrishnan et al., 2021; Saheb Sharif-Askari et al., 2021). More studies are needed to profile the long-term metabolic change in those suffering from lingering long COVID symptoms. In addition to severe metabolic pathways, we have also profiled those that were specific to non-severe group which included sphingosine. Our differential metabolite analyses in both saliva and plasma samples have shown higher sphingosine levels in non-severe than severe COVID-19 (Fig. 3A, B and Fig. 6A). Interestingly, salivary sphingosine pattern revealed an inverse relationship between sphingosine levels and disease severity with significant increase of sphingosine levels in asymptomatic and its complete absence in severe disease. We detected sphingosine in salivary controls but not in plasma controls. Similar to our result, Janneh et al. (Janneh et al., 2021) and Shen et al. (Shen et al., 2020) associated the reduced plasma sphingosine levels to COVID-19 severity. In their findings plasma sphingosine levels differentiated symptomatic from non-symptomatic infection but it could not differentiate severe from non-severe disease (Janneh et al., 2021; Shen et al., 2020). Sphingosine present abundantly in healthy nasal, tracheal, and bronchial epithelial cells protecting the respiratory tracts from both bacterial and viral respiratory infection (Verhaegh et al., 2020). It prevent SARS-CoV-2 viral entry into host cells; sphingosine binds to ACE2 receptor and block the interaction between SARS-CoV-2 viral spike protein and host ACE2 receptor (Edwards et al., 2020). It is involved in synthesis and regulation of multivesicular bodies which modulate diverse cellular processes (Lang et al., 2020). Additional antiviral effect of sphingosine was shown against herpes simplex virus in macrophages where sphingosine binds, traps and degrades the virus within the multivesicular bodies (Lang et al., 2020). The bactericidal effect of this sphingolipid have been shown against different bacterial strains (Grassmé et al., 2017; Seitz et al., 2019). Verhegh and colleagues demonstrated that protonated amino group (NH3+) group of sphingosine binds to the—negatively charged lipid cardiolipin in Pseudomonas aeruginosa and Staphylococcus aureus plasma membranes which then induce rapid permeabilization and loss of bacterial metabolic activity (Verhaegh et al., 2020). Sphingosine is phosphorylated by sphingosine kinases to generate Sphingosine-1-phosphate (Liu et al., 2002). In our study we did not detect sphingosine-1-phosphate, however recently Marfia group linked the decreased serum level of sphingosine-1-phosphate with severe COVID-19 disease (Marfia et al., 2021). Sphingosine-1-phosphate is a pleotropic sphingolipid that acts as a major modulator of vascular and immune system (Törnquist et al., 2021). It displays dose-dependent effect on inflammation; at normal nanomolar physiologic concentration it acts as anti-inflammatory while at higher micromolar concentration it shows pro-inflammatory activity (Fettel et al., 2019; Whetzel et al., 2006). Airway smooth muscles treated with micromolar concentration of sphingosine-1-phosphate induced their proinflammatory effect which was inhibited upon treatment with corticosteroid, dexamethasone (Che et al., 2013). In small dose, Sphingosine-1-phosphate analogue, Fingolimod, showed reduction of inflammatory response and entered a clinical trial for treatment of cytokine storm during COVID-19; however the trial was stopped due to occurrence of lymphopenia adverse reaction (www.clinicaltrial.gov NCT04280588)(Marfia et al., 2021). Further studies are required to assess the potential of sphingolipid modulation towards control of SARS-CoV-2 viral infection as well as cytokine storm. Resembling sphingosine findings, 5-Aminolevulinic acid metabolite levels were detected in non-severe COVID-19, while this metabolite was not present in control and severe COVID-19 in both saliva and plasma results. This metabolite has antiviral effect and thereby their detection in the less severe infection suggest that it could have played a role in the control of infection severity. Ben-Hur and colleague showed that 5-Aminolevulinic Acid supplementation followed by photodynamic therapy was effective both in-vivo and in-vitro to inactivate intracellular herpes simplex virus (HSV) and human immunodeficiency virus (HIV) (Smetana et al., 1997). More recently, Japanese researchers have shown that pretreatment with this amino acid blocked the SARS-CoV-2 infection in both VeroE6 cells and human colon-derived Caco-2 cells in a time-dependent manner (Sakurai et al., 2021). Mechanistically, 5-ALA supplementation induced accumulation of protoporphyrin IX and heme inside host cells, that interfered with viral replication through targeting the G-quadruplex (G4) structure within the virus nonstructural protein 3 (Nsp3) (Sakurai et al., 2021). 5-Aminolevulinic Acid is bioavailable orally and could potentially be used for SARS-CoV-2 infection prevention (Sakurai et al., 2021). Beside sphingosine, other lipid mediators such as eicosanoids were previously shown to be upregulated during COVID-19 (Regidor et al., 2021). Eicosanoids were associated with lipid mediator storm during critical SARS-CoV-2 infection as well as post COVID-19 syndrome following mild disease (Archambault et al., 2021; Bohnacker et al., 2022). SARS-CoV-2 infection induced a long-term immune abnormalities in macrophage effector functions and eicosanoid metabolism which persisted up to 3–5 month following mild COVID-19 (Bohnacker et al., 2022). Future investigations are needed to study the role of other bioactive lipids during and after severe viral infections. LC–MS/MS analysis could be used for simultaneous detection of bioactive lipids such as eicosanoids in different patients’ samples of serum, bronchial alveolar lavage fluid (BALF), and sputum (Thakare et al., 2018). The current study identified the metabolites that are specific to severe and non-severe groups. Like sphingosine, metabolites presented in Fig. 3A, B such as N-Acetylputrescine could play a key role in development or resistance to severe viral infection and thereby further research is needed to understand the mechanism that regulate their abundance. Moreover, many of the dysregulated metabolites are expected to be directly or indirectly associated with clinical manifestation observed in acute phase and long-term sequel of COVID-19 and deserve further investigation. As expected, the two prognostic biomarkers of C-reactive protein and D-dimer were negatively correlated with sphingosine and 5-Aminolevulinic acid (Fig. 7A, B and E, F), and positively correlated with L-Tryptophan and l-Kynurenine (Fig. 7C, D and G, H). This study was conducted to profile the global metabolic dysregulation during acute COVID-19 disease, and to focus on those metabolites that are specific to non-severe and severe groups. Our study only provides a snapshot of SARS-CoV-2 induced metabolic reprogramming. Future longitudinal studies are required to better characterize the early, acute, and long-term change in COVID-19. Our study was limited by the sample size and larger cohort could be required to confirm the current findings. Although we have used age and gender matched cohort (Supplementary Table 1), metabolite change observed could be cofounded or not by other factors such as diet, disease, and medication. To control for the potential cofounder’s larger sample size will be needed. Another limitation was that deuterium-labeled internal standards were not used prior to sample extraction.
5 Conclusion
In conclusion, SARS-CoV-2 infection induce distinctive metabolic derangement in severe and non-severe COVID-19. Metabolic change could be detected in both salivary and plasma samples, however saliva collected from active side of infection presented with higher intensity of metabolic dysregulation. General COVID-19 metabolites (such as Linoleic acid and Alpha-ketoisovaleric in Fig. 3A, B) could be used for infection detection while metabolites distinguishing severe from non-severe stages (such as sphingosine and kynurenine) could be used as a prognostic marker. Further, metabolites such as l-Kynurenine, sphingosine and 5-aminolevulinic could play active role in modulating immune system inflammatory and antiviral response and could represent an effective therapeutic approach for the treatment of COVID-19 early and late immune dysregulations.
References
Al-Muhsen, S., Al-Numair, N. S., Saheb Sharif-Askari, N., Basamh, R., Alyounes, B., Jabaan, A., Saheb Sharif-Askari, F., Alosaimi, M. F., Alsohime, F., Halwani, R., & Al-Saud, H. (2022). Favipiravir effectiveness and safety in hospitalized moderate-severe COVID-19 patients: Observational prospective multicenter investigation in Saudi Arabia. Frontiers in Medicine, 9, 826247–826247.
Alaiya, A., Alshukairi, A., Shinwari, Z., Al-Fares, M., Alotaibi, J., AlOmaim, W., Alsharif, I., Bakheet, R., Alharbi, L., Allam, R., Asiri, A., Memish, Z., Alromaih, K., & Al-Mozaini, M. (2021). Alterations in the plasma proteome induced by SARS-CoV-2 and MERS-CoV reveal biomarkers for disease outcomes for COVID-19 patients. Journal of Inflammation Research, 14, 4313–4328.
Alamer, A., Alrashed, A. A., Alfaifi, M., Alosaimi, B., AlHassar, F., Almutairi, M., Howaidi, J., Almutairi, W., Mohzari, Y., Sulaiman, T., Al-Jedai, A., Alajami, H. N., Alkharji, F., Alsaeed, A., Alali, A. H., Baredhwan, A. A., Abraham, I., & Almulhim, A. S. (2021). Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: A retrospective study with propensity score matching sensitivity analysis. Current Medical Research and Opinion, 37, 1085–1097.
Archambault, A. S., Zaid, Y., Rakotoarivelo, V., Turcotte, C., Doré, É., Dubuc, I., Martin, C., Flamand, O., Amar, Y., Cheikh, A., Fares, H., El Hassani, A., Tijani, Y., Côté, A., Laviolette, M., Boilard, É., Flamand, L., & Flamand, N. (2021). High levels of eicosanoids and docosanoids in the lungs of intubated COVID-19 patients. The FASEB Journal, 35, e21666.
Blanco-Melo, D., Nilsson-Payant, B. E., Liu, W.-C., Uhl, S., Hoagland, D., Møller, R., Jordan, T. X., Oishi, K., Panis, M., Sachs, D., Wang, T. T., Schwartz, R. E., Lim, J. K., Albrecht, R. A., & tenOever, B. R. (2020). Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell, 181, 1036-1045.e9.
Bohnacker, S., Hartung, F., Henkel, F., Quaranta, A., Kolmert, J., Priller, A., Ud-Dean, M., Giglberger, J., Kugler, L. M., Pechtold, L., Yazici, S., Lechner, A., Erber, J., Protzer, U., Lingor, P., Knolle, P., Chaker, A. M., Schmidt-Weber, C. B., Wheelock, C. E., & Esser-von Bieren, J. (2022). Mild COVID-19 imprints a long-term inflammatory eicosanoid- and chemokine memory in monocyte-derived macrophages. Mucosal Immunology, 15, 515–524.
Cai, Y., Kim, D. J., Takahashi, T., Broadhurst, D. I., Yan, H., Ma, S., Rattray, N. J., Casanovas-Massana, A., Israelow, B., & Klein, J. (2021). Kynurenic acid may underlie sex-specific immune responses to COVID-19. Science Signaling, 14, eabf8483.
Che, W., Parmentier, J., Seidel, P., Manetsch, M., Ramsay, E. E., Alkhouri, H., Ge, Q., Armour, C. L., & Ammit, A. J. (2013). Corticosteroids inhibit sphingosine 1-phosphate–induced interleukin-6 secretion from human airway smooth muscle via mitogen-activated protein kinase phosphatase 1–mediated repression of mitogen and stress-activated protein kinase 1. American Journal of Respiratory Cell and Molecular Biology, 50, 358–368.
Chen, Y., & Guillemin, G. J. (2009). Kynurenine pathway metabolites in humans: Disease and healthy States. International Journal of Tryptophan, 2, 1–19.
dos Santos, C., Junior, G., Pereira, C. M., da Silva, K., Fidalgo, T., & Valente, A. P. (2020). Saliva NMR-based metabolomics in the war against COVID-19. Analytical Chemistry, 92, 15688–15692.
Deu-Pons, J., Schroeder, M. P., & Lopez-Bigas, N. (2014). jHeatmap: An interactive heatmap viewer for the web. Bioinformatics, 30, 1757–1758.
Dudoit, S., Yang, Y. H., Callow, M. J., & Speed, T. P. (2002). Statistical methods for identifying differentially expressed genes in replicated cDNA microarray experiments. Statistica Sinica, 15, 111–139.
Edwards, M. J., Becker, K. A., Gripp, B., Hoffmann, M., Keitsch, S., Wilker, B., Soddemann, M., Gulbins, A., Carpinteiro, E., Patel, S. H., Wilson, G. C., Pöhlmann, S., Walter, S., Fassbender, K., Ahmad, S. A., Carpinteiro, A., & Gulbins, E. (2020). Sphingosine prevents binding of SARS–CoV-2 spike to its cellular receptor ACE2. Journal of Biological Chemistry, 295, 15174–15182.
Eroğlu, İ, Eroğlu, B. Ç., & Güven, G. S. (2021). Altered tryptophan absorption and metabolism could underlie long-term symptoms in survivors of coronavirus disease 2019 (COVID-19). Nutrition (burbank, Los Angeles County, Calif.), 90, 111308–111308.
Fettel, J., Kühn, B., Guillen, N. A., Sürün, D., Peters, M., Bauer, R., Angioni, C., Geisslinger, G., Schnütgen, F., Heringdorf, D. M., Werz, O., Meybohm, P., Zacharowski, K., Steinhilber, D., Roos, J., & Maier, T. J. (2019). Sphingosine-1-phosphate (S1P) induces potent anti-inflammatory effects in vitro and in vivo by S1P receptor 4-mediated suppression of 5-lipoxygenase activity. The FASEB Journal, 33, 1711–1726.
Frampas, C.F., Longman, K., Spick, M.P., Lewis, H.M., Costa, C.D.S., Stewart, A., Dunn-Walters, D., Greener, D., Evetts, G.E., Skene, D., Trivedi, D., Pitt, A.R., Hollywood, K., Barran, P. and Bailey, M.J. (2021) Untargeted saliva metabolomics reveals COVID-19 severity. medRxiv, 2021.07.06.21260080.
Geyer, P. E., Arend, F. M., Doll, S., Louiset, M. L., Virreira Winter, S., Müller-Reif, J. B., Torun, F. M., Weigand, M., Eichhorn, P., Bruegel, M., Strauss, M. T., Holdt, L. M., Mann, M., & Teupser, D. (2021). High-resolution serum proteome trajectories in COVID-19 reveal patient-specific seroconversion. EMBO Molecular Medicine, 13, e14167.
Grassmé, H., Henry, B., Ziobro, R., Becker, K. A., Riethmüller, J., Gardner, A., Seitz, A. P., Steinmann, J., Lang, S., Ward, C., Schuchman, E. H., Caldwell, C. C., Kamler, M., Edwards, M. J., Brodlie, M., & Gulbins, E. (2017). β1-Integrin accumulates in cystic fibrosis luminal airway epithelial membranes and decreases sphingosine, promoting bacterial infections. Cell Host & Microbe, 21, 707-718.e8.
Guan, W. J., Ni, Z. Y., Hu, Y., Liang, W. H., Ou, C. Q., He, J. X., Liu, L., Shan, H., Lei, C. L., Hui, D. S. C., Du, B., Li, L. J., Zeng, G., Yuen, K. Y., Chen, R. C., Tang, C. L., Wang, T., Chen, P. Y., **ang, J., … Zhong, N. S. (2020). Clinical characteristics of coronavirus disease 2019 in China. New England Journal of Medicine, 382, 1708–1720.
Hoffmann, M., Kleine-Weber, H., Schroeder, S., Krüger, N., Herrler, T., Erichsen, S., Schiergens, T. S., Herrler, G., Wu, N.-H., Nitsche, A., Müller, M. A., Drosten, C., & Pöhlmann, S. (2020). SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell, 181, 271-280.e8.
Huang, C., Huang, L., Wang, Y., Li, X., Ren, L., Gu, X., Kang, L., Guo, L., Liu, M., Zhou, X., Luo, J., Huang, Z., Tu, S., Zhao, Y., Chen, L., Xu, D., Li, Y., Li, C., Peng, L., … Cao, B. (2021). 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet, 397, 220–232.
Hui, D. S., Azhar E.I., Madani, T.A., Ntoumi, F., Kock, R., Dar, O., Ippolito, G., McHugh, T.D., Memish, Z.A., Drosten, C., Zumla, A. & Petersen, E. (2020). The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - The latest 2019 novel coronavirus outbreak in Wuhan, China. International Journal of Infectious Diseases, 91, 264–266.
Hyvärinen, E., Savolainen, M., Mikkonen, J. J. W., & Kullaa, A. M. (2021). Salivary metabolomics for diagnosis and monitoring diseases: Challenges and possibilities. Metabolites, 11, 15–90.
Janneh, A. H., Kassir, M. F., Dwyer, C. J., Chakraborty, P., Pierce, J. S., Flume, P. A., Li, H., Nadig, S. N., Mehrotra, S., & Ogretmen, B. (2021). Alterations of lipid metabolism provide serologic biomarkers for the detection of asymptomatic versus symptomatic COVID-19 patients. Scientific Reports, 11, 14232.
Kimhofer, T., Lodge, S., Whiley, L., Gray, N., Loo, R. L., Lawler, N. G., Nitschke, P., Bong, S.-H., Morrison, D. L., Begum, S., Richards, T., Yeap, B. B., Smith, C., Smith, K. G. C., Holmes, E., & Nicholson, J. K. (2020). Integrative modeling of quantitative plasma lipoprotein, metabolic, and amino acid data reveals a multiorgan pathological signature of SARS-CoV-2 infection. Journal of Proteome Research, 19, 4442–4454.
Lang, J., Bohn, P., Bhat, H., Jastrow, H., Walkenfort, B., Cansiz, F., Fink, J., Bauer, M., Olszewski, D., Ramos-Nascimento, A., Duhan, V., Friedrich, S.-K., Becker, K. A., Krawczyk, A., Edwards, M. J., Burchert, A., Huber, M., Friebus-Kardash, J., Göthert, J. R., … Lang, K. S. (2020). Acid ceramidase of macrophages traps herpes simplex virus in multivesicular bodies and protects from severe disease. Nature Communications, 11, 1338.
Lee, J. S., Park, S., Jeong, H. W., Ahn, J. Y., Choi, S. J., Lee, H., Choi, B., Nam, S. K., Sa, M., Kwon, J.-S., Jeong, S. J., Lee, H. K., Park, S. H., Park, S.-H., Choi, J. Y., Kim, S.-H., Jung, I., & Shin, E.-C. (2020). Immunophenoty** of COVID-19 and influenza highlights the role of type I interferons in development of severe COVID-19. Science Immunology, 5, eabd1554.
Liao, M., Liu, Y., Yuan, J., Wen, Y., Xu, G., Zhao, J., Cheng, L., Li, J., Wang, X., Wang, F., Liu, L., Amit, I., Zhang, S., & Zhang, Z. (2020). Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nature Medicine, 26, 842–844.
Lima, D. P., Diniz, D. G., Moimaz, S. A. S., Sumida, D. H., & Okamoto, A. C. (2010). Saliva: Reflection of the body. International Journal of Infectious Diseases, 14, e184–e188.
Liu, H., Chakravarty, D., Maceyka, M., Milstien, S., & Spiegel, S. (2002). Sphingosine kinases: A novel family of lipid kinases. Progress in Nucleic Acid Research and Molecular Biology, 71, 493–511.
López-Hernández, Y., Monárrez-Espino, J., Oostdam, A.-S.H.-V., Delgado, J.E.C., Zhang, L., Zheng, J., Valdez, J.J.O., Mandal, R., González, F.d.L.O., Moreno, J.C.B., Trejo-Medinilla, F.M., López, J.A., Moreno, J.A.E. & Wishart, D.S. (2021). Targeted metabolomics identifies high performing diagnostic and prognostic biomarkers for COVID-19. Scientific Reports, 11, 14732.
Ma, Q., Liu, J., Liu, Q., Kang, L., Liu, R., **g, W., Wu, Y., & Liu, M. (2021). Global percentage of asymptomatic SARS-CoV-2 infections among the tested population and individuals with confirmed COVID-19 diagnosis: A systematic review and meta-analysis. JAMA Network Open, 4, e2137257–e2137257.
Mangoni, A. (1974). The “kynurenine shunt” and depression. Advances in Biochemical Psychopharmacology, 11, 293–298.
Marfia, G., Navone, S., Guarnaccia, L., Campanella, R., Mondoni, M., Locatelli, M., Barassi, A., Fontana, L., Palumbo, F., Garzia, E., Ciniglio Appiani, G., Chiumello, D., Miozzo, M., Centanni, S., & Riboni, L. (2021). Decreased serum level of sphingosine-1-phosphate: A novel predictor of clinical severity in COVID-19. EMBO Molecular Medicine, 13, e13424.
Masuda, R., Lodge, S., Nitschke, P., Spraul, M., Schaefer, H., Bong, S.-H., Kimhofer, T., Hall, D., Loo, R. L., Bizkarguenaga, M., Bruzzone, C., Gil-Redondo, R., Embade, N., Mato, J. M., Holmes, E., Wist, J., Millet, O., & Nicholson, J. K. (2021). Integrative modeling of plasma metabolic and lipoprotein biomarkers of SARS-CoV-2 infection in Spanish and Australian COVID-19 patient cohorts. Journal of Proteome Research, 20, 4139–4152.
Németh, H., Toldi, J., & Vécsei, L. (2005). Role of kynurenines in the central and peripheral nervous systems. Current Neurovascular Research, 2, 249–260.
NIH COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available at https://www.covid19treatmentguidelines.nih.gov/. Accessed 9 Sep 2021.
Pang, Z., Chong, J., Li, S., & **a, J. (2020). MetaboAnalystR 3.0: Toward an optimized workflow for global metabolomics. Metabolites, 10, 15–82.
Patel, H., Ashton, N. J., Dobson, R. J. B., Andersson, L.-M., Yilmaz, A., Blennow, K., Gisslen, M., & Zetterberg, H. (2021). Proteomic blood profiling in mild, severe and critical COVID-19 patients. Scientific Reports, 11, 6357.
Qin, C., Zhou, L., Hu, Z., Zhang, S., Yang, S., Tao, Y., **e, C., Ma, K., Shang, K., Wang, W., & Tian, D. S. (2020). Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clinical Infectious Diseases, 71, 762–768.
Ramakrishnan, R. K., Kashour, T., Hamid, Q., Halwani, R., & Tleyjeh, I. M. (2021). Unraveling the mystery surrounding post-acute sequelae of COVID-19. Frontiers in Immunology, 12, 20–28.
Regidor, P. A., De La Rosa, X., Santos, F. G., Rizo, J. M., Gracia Banzo, R., & Silva, R. S. (2021). Acute severe SARS COVID-19 patients produce pro-resolving lipids mediators and eicosanoids. European Review for Medical and Pharmacological Sciences, 25, 6782–6796.
Richardson, S., Hirsch, J. S., Narasimhan, M., Crawford, J. M., McGinn, T., Davidson, K. W., Barnaby, D. P., Becker, L. B., Chelico, J. D., Cohen, S. L., Cookingham, J., Coppa, K., Diefenbach, M. A., Dominello, A. J., Duer-Hefele, J., Falzon, L., Gitlin, J., Hajizadeh, N., Harvin, T. G., … Zanos, T. P. (2020). Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized With COVID-19 in the New York City area. JAMA, 323, 2052–2059.
Ritchie, M. E., Phipson, B., Wu, D., Hu, Y., Law, C. W., Shi, W., & Smyth, G. K. (2015a). Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Research, 43, e47.
Ritchie, M. E., Phipson, B., Wu, D. I., Hu, Y., Law, C. W., Shi, W., & Smyth, G. K. (2015b). limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Research, 43, e47–e47.
Saheb Sharif-Askari, N., Saheb Sharif-Askari, F., Ahmed, S. B. M., Hannawi, S., Hamoudi, R., Hamid, Q., & Halwani, R. (2021). Enhanced expression of autoantigens during SARS-CoV-2 viral infection. Frontiers in Immunology, 12, 15–98.
Sakurai, Y., Ngwe Tun, M. M., Kurosaki, Y., Sakura, T., Inaoka, D. K., Fu**e, K., Kita, K., Morita, K., & Yasuda, J. (2021). 5-amino levulinic acid inhibits SARS-CoV-2 infection in vitro. Biochemical and Biophysical Research Communications, 545, 203–207.
Sapkota, D., Søland, T. M., Galtung, H. K., Sand, L. P., Giannecchini, S., To, K. K. W., Mendes-Correa, M. C., Giglio, D., Hasséus, B., & Braz-Silva, P. H. (2020). COVID-19 salivary signature: diagnostic and research opportunities. Journal of Clinical Pathology, 74(6), 344–349.
Sapkota, D., Søland, T. M., Galtung, H. K., Sand, L. P., Giannecchini, S., To, K. K. W., Mendes-Correa, M. C., Giglio, D., Hasséus, B., & Braz-Silva, P. H. (2021). COVID-19 salivary signature: Diagnostic and research opportunities. Journal of Clinical Pathology, 74, 344.
Seitz, A. P., Schumacher, F., Baker, J., Soddemann, M., Wilker, B., Caldwell, C. C., Gobble, R. M., Kamler, M., Becker, K. A., Beck, S., Kleuser, B., Edwards, M. J., & Gulbins, E. (2019). Sphingosine-coating of plastic surfaces prevents ventilator-associated pneumonia. Journal of Molecular Medicine (berlin, Germany), 97, 1195–1211.
Shen, B., Yi, X., Sun, Y., Bi, X., Du, J., Zhang, C., Quan, S., Zhang, F., Sun, R., Qian, L., Ge, W., Liu, W., Liang, S., Chen, H., Zhang, Y., Li, J., Xu, J., He, Z., Chen, B., … Guo, T. (2020). Proteomic and metabolomic characterization of COVID-19 patient sera. Cell, 182, 59-72.e15.
Siddiqi, H. K., & Mehra, M. R. (2020). COVID-19 illness in native and immunosuppressed states: A clinical-therapeutic staging proposal. The Journal of Heart and Lung Transplantation, 39, 405–407.
Smetana, Z., Malik, Z., Orenstein, A., Mendelson, E., & Ben-Hur, E. (1997). Treatment of viral infections with 5-aminolevulinic acid and light. Lasers in Surgery and Medicine, 21, 351–358.
Smyth Gordon, K. (2004). Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Statistical Applications in Genetics and Molecular Biology, 3, 1–25.
Statsenko, Y., Al Zahmi, F., Habuza, T., Gorkom, K.N.-V., & Zaki, N. (2021). Prediction of COVID-19 severity using laboratory findings on admission: Informative values, thresholds ML Model Performance. British Medical Journal Open, 11, e044500.
Thakare, R., Chhonker, Y. S., Gautam, N., Nelson, A., Casaburi, R., Criner, G., Dransfield, M. T., Make, B., Schmid, K. K., Rennard, S. I., & Alnouti, Y. (2018). Simultaneous LC-MS/MS analysis of eicosanoids and related metabolites in human serum, sputum and BALF. Biomed Chromatography, 32, 15.
Thomas, T., Stefanoni, D., Reisz, J. A., Nemkov, T., Bertolone, L., Francis, R. O., Hudson, K. E., Zimring, J. C., Hansen, K. C., Hod, E. A., Spitalnik, S. L., & D’Alessandro, A. (2020). COVID-19 infection alters kynurenine and fatty acid metabolism, correlating with IL-6 levels and renal status. JCI Insight, 5, 15.
To, K. K. W., Yip, C. C. Y., Lai, C. Y. W., Wong, C. K. H., Ho, D. T. Y., Pang, P. K. P., Ng, A. C. K., Leung, K. H., Poon, R. W. S., Chan, K. H., Cheng, V. C. C., Hung, I. F. N., & Yuen, K. Y. (2019). Saliva as a diagnostic specimen for testing respiratory virus by a point-of-care molecular assay: A diagnostic validity study. Clinical Microbiology and Infection, 25, 372–378.
Törnquist, K., Asghar, M. Y., Srinivasan, V., Korhonen, L., & Lindholm, D. (2021). Sphingolipids as Modulators of SARS-CoV-2 Infection. Frontiers in Cell and Developmental Biology, 9, 1574.
Vardhana, S. A., & Wolchok, J. D. (2020). The many faces of the anti-COVID immune response. Journal of Experimental Medicine, 217, 158.
Verhaegh, R., Becker, K. A., Edwards, M. J., & Gulbins, E. (2020). Sphingosine kills bacteria by binding to cardiolipin. Journal of Biological Chemistry, 295, 7686–7696.
Whetzel, A. M., Bolick, D. T., Srinivasan, S., Macdonald, T. L., Morris, M. A., Ley, K., & Hedrick, C. C. (2006). Sphingosine-1 Phosphate Prevents Monocyte/Endothelial Interactions in Type 1 Diabetic NOD Mice Through Activation of the S1P1 Receptor. Circulation Research, 99, 731–739.
www.cdc.gov Available from: https://www.cdc.gov/coronavirus/2019-ncov/lab/guidelines-clinical-specimens.html. Accessed date 9 Sep 2020.
Wyllie, A. L., Fournier, J., Casanovas-Massana, A., Campbell, M., Tokuyama, M., Vijayakumar, P., Warren, J. L., Geng, B., Muenker, M. C., Moore, A. J., Vogels, C. B. F., Petrone, M. E., Ott, I. M., Lu, P., Venkataraman, A., Lu-Culligan, A., Klein, J., Earnest, R., Simonov, M., … Ko, A. I. (2020). Saliva or nasopharyngeal swab specimens for detection of SARS-CoV-2. New England Journal of Medicine, 383, 1283–1286.
**a, J., & Wishart, D. S. (2010). MSEA: A web-based tool to identify biologically meaningful patterns in quantitative metabolomic data. Nucleic Acids Research, 38, W71–W77.
**ao, N., Nie, M., Pang, H., Wang, B., Hu, J., Meng, X., Li, K., Ran, X., Long, Q., Deng, H., Chen, N., Li, S., Tang, N., Huang, A., & Hu, Z. (2021). Integrated cytokine and metabolite analysis reveals immunometabolic reprogramming in COVID-19 patients with therapeutic implications. Nature Communications, 12, 1618.
Xu, H., Zhong, L., Deng, J., Peng, J., Dan, H., Zeng, X., Li, T., & Chen, Q. (2020). High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. International Journal of Oral Science, 12, 8.
Yan, L., Zhang, H.-T., Goncalves, J., **ao, Y., Wang, M., Guo, Y., Sun, C., Tang, X., **g, L., Zhang, M., Huang, X., **ao, Y., Cao, H., Chen, Y., Ren, T., Wang, F., **ao, Y., Huang, S., Tan, X., … Yuan, Y. (2020). An interpretable mortality prediction model for COVID-19 patients. Nature Machine Intelligence, 2, 283–288.
Yang, X., Yu, Y., Xu, J., Shu, H., **a, J., Liu, H., Wu, Y., Zhang, L., Yu, Z., Fang, M., Yu, T., Wang, Y., Pan, S., Zou, X., Yuan, S., & Shang, Y. (2020). Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. The Lancet Respiratory Medicine, 8, 475–481.
Yoon, J., Yun, S. G., Nam, J., Choi, S. H., & Lim, C. S. (2017). The use of saliva specimens for detection of influenza A and B viruses by rapid influenza diagnostic tests. Journal of Virological Methods, 243, 15–19.
Zhang, C.-Z., Cheng, X.-Q., Li, J.-Y., Zhang, P., Yi, P., Xu, X., & Zhou, X.-D. (2016). Saliva in the diagnosis of diseases. International Journal of Oral Science, 8, 133–137.
Zheng, H. Y., Zhang, M., Yang, C. X., Zhang, N., Wang, X. C., Yang, X. P., Dong, X. Q., & Zheng, Y. T. (2020). Elevated exhaustion levels and reduced functional diversity of T cells in peripheral blood may predict severe progression in COVID-19 patients. Cellular & Molecular Immunology, 17, 541–543.
Zhou, Y., Fu, B., Zheng, X., Wang, D., Zhao, C., Qi, Y., Sun, R., Tian, Z., Xu, X., & Wei, H. (2020). Pathogenic T-cells and inflammatory monocytes incite inflammatory storms in severe COVID-19 patients. National Science Review, 7, 998–1002.
Funding
The authors extend their appreciation to the Deanship of Scientific Research, king Saud University for funding through Vice Deanship of Scientific Research Chairs; Research Chair of Prince Abdullah Ben Khalid Celiac Disease research chair; Riyadh, Kingdom of Saudi Arabia.
Author information
Authors and Affiliations
Contributions
RH, MHS, NCS, NSA designed the research. HAHA, LS, RS, BM collected the data and patient’s sample. NSA, FSA, NCS, HAM QH, MHS and RH performed the research. NSA, FSA, NCS and RH analyzed and interpreted the data and wrote the manuscript. All authors commented on the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was approved by Dubai Scientific Research Ethics Committee (DSREC). Written, informed consents were obtained from all study participants prior to inclusion.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Narjes Saheb Sharif-Askari, Nelson Cruz Soares—equal first co-authorship.
Supplementary Information
Below is the link to the electronic supplementary material.
11306_2022_1936_MOESM1_ESM.tiff
Metabolite Set Enrichment Analysis for top 34 differential metabolites derived from COVID-19 and non-infected controls comparisons. Top enriched pathways included tryptophan metabolism, purine metabolism, phosphonate and phosphinate metabolism and citrate cycle (TCA) pathways. Supplementary file1 (TIFF 37968 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saheb Sharif-Askari, N., Soares, N.C., Mohamed, H.A. et al. Saliva metabolomic profile of COVID-19 patients associates with disease severity. Metabolomics 18, 81 (2022). https://doi.org/10.1007/s11306-022-01936-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-022-01936-1