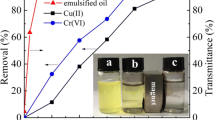
Abstract
Produced water has a considerable amount of oil that can harm the environment if dumped prior to treatment. Iron oxide (maghemite) nanoparticles have attracted much attention as potential adsorbents due to their unique properties, high adsorption capacity, super magnetization, and high surface area. In this study, maghemite 3 nm and 15–20 nm have been used as adsorbents. Oil has been successfully removed from oil-water emulsion using maghemite with the application of a magnetic field. The effects of the varying process parameters, including adsorbent dose, particle size, mixing time, API, pH, temperature, and oil concentration, on the percentage of oil removal (OR%) were studied. The highest OR% was achieved at 20 min, 100 mg/L oil concentration, 80 mg adsorbent dose, room temperature, 30 API, and pH 6; these data were taken as a reference to evaluate the other parameters. The highest ORs% were 94% and 83% for MNPs of 3 nm and 15–20 nm, respectively. Stability and reusability tests of the maghemite exhibited an effective oil recovery of up to 77% and 64% after 9 cycles. They displayed a maximum adsorption capacity of 3280 mg/g and 3235 mg/g for the 3 nm and 15–20 nm nanoparticles, respectively. The Langmuir isotherm model showed a better fit with experimental data compared to the Freundlich-isotherm model. Kinetic adsorption models fit effectively with the pseudo-second-order model. Thus, the results demonstrate maghemite nanoparticles’ efficiency in removing oil from oil-water emulsions. This innovative technique will reduce time and optimize the conventional treatment process.









Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Abrouki, Y., Mabrouki, J., Anouzla, A., Rifi, S. K., Zahiri, Y., Nehhal, S., et al. (2021). Optimization and modeling of a fixed-bed biosorption of textile dye using agricultural biomass from the Moroccan Sahara. Desalination and Water Treatment, 240, 144 https://www.academia.edu/78004341/Optimization_and_modeling_of_a_fixed_bed_biosorption_of_textile_dye_using_agricultural_biomass_from_the_Moroccan_Sahara. Accessed 30 June 2023
Alshamsi, H. A., & Hussein, B. S. (2018). Synthesis, characterization and photocatalysis of g-Fe2O3 nanoparticles for degradation of cibacron brilliant yellow 3G-P. Asian Journal of Chemistry, 30(2), 273–279. https://doi.org/10.14233/ajchem.2018.20888
Anjum, M., Miandad, R., Waqas, M., Gehany, F., & Barakat, M. A. (2019). Remediation of wastewater using various nano-materials. Arabian Journal of Chemistry, 12(8), 4897–4919. https://doi.org/10.1016/j.arabjc.2016.10.004
Aristizábal-Fontal, J. E., Cortés, F. B., & Franco, C. A. (2018). Viscosity reduction of extra heavy crude oil by magnetite nanoparticle-based ferrofluids. Adsorption Science & Technology, 36(1–2), 23–45. https://doi.org/10.1177/0263617417704309
Asadu, C. O., Ekwueme, B. N., Onu, C. E., Onah, T. O., Sunday Ike, I., & Ezema, C. A. (2022). Modelling and optimization of crude oil removal from surface water via organic acid functionalized biomass using machine learning approach. Arabian Journal of Chemistry, 15(9), 104025. https://doi.org/10.1016/j.arabjc.2022.104025
Ayawei, N., Ebelegi, A. N., & Wankasi, D. (2017). Modelling and interpretation of adsorption isotherms. Journal of Chemistry, 2017, 1–11. https://doi.org/10.1155/2017/3039817
Cuccia, V., Seles, S. R. N., & Ladeira, A. C. Q. (2019). Removal of cobalt from lubricant oil by the use of bentonite: Equilibrium, kinetic and adsorption preliminary studies. Brazilian Journal of Radiation Sciences, 7(2A). https://doi.org/10.15392/bjrs.v7i2A.564
Elmobarak, W. F., & Almomani, F. (2021). Application of magnetic nanoparticles for the removal of oil from oil-in-water emulsion: Regeneration/reuse of spent particles. Journal of Petroleum Science and Engineering, 203, 108591. https://doi.org/10.1016/j.petrol.2021.108591
Etale, A., Tutu, H., & Drake, D. C. (2016). Application of maghemite nanoparticles as sorbents for the removal of Cu(II), Mn(II) and U(VI) ions from aqueous solution in acid mine drainage conditions. Applied Water Science, 6(2), 187–197. https://doi.org/10.1007/s13201-014-0217-3
Eweida, B. Y., Omer, A. M., Tamer, T. M., Soliman, H. A.-E. M., Zaatot, A. A., & Mohy-Eldin, M. S. (2022). Kinetics, isotherms and thermodynamics of oil spills removal by novel amphiphilic chitosan-g-Octanal Schiff base polymer developed by click grafting technique. Polymer Bulletin. https://doi.org/10.1007/s00289-022-04260-9
Ewis, D., Benamor, A., Ba-Abbad, M. M., Nasser, M., El-Naas, M., & Qiblawey, H. (2020). Removal of oil content from oil-water emulsions using iron oxide/bentonite nano adsorbents. Journal of Water Process Engineering, 38, 101583. https://doi.org/10.1016/j.jwpe.2020.101583
Faisal, W., & Almomani, F. (2020). Application of Fe3O4 magnetite nanoparticles grafted in silica (SiO2) for oil recovery from oil in water emulsions. Chemosphere, 265, 129054. https://doi.org/10.1016/j.chemosphere.2020.129054
Fakhru’l-Razi, A., Pendashteh, A., Abdullah, L. C., Biak, D. R. A., Madaeni, S. S., & Abidin, Z. Z. (2009). Review of technologies for oil and gas produced water treatment. Journal of Hazardous Materials, 170(2–3), 530–551. https://doi.org/10.1016/j.jhazmat.2009.05.044
Hernández, L., Augusto, P. A., Castelo-Grande, T., & Barbosa, D. (2021). Regeneration and reuse of magnetic particles for contaminant degradation in water. Journal of Environmental Management, 285, 112155. https://doi.org/10.1016/j.jenvman.2021.112155
Hilma Siregar, S., Wijaya, K., Sri Kunarti, E., Syoufian, A., & Suyanta. (2019). Kinetics adsorption of heavy oil spills in rivers on magnetite-(CTAB-montmorillonite) adsorbent. IOP Conference Series: Materials Science and Engineering, 509, 012136. https://doi.org/10.1088/1757-899X/509/1/012136
Huang, S., Li, X., Jiao, Y., & Shi, J. (2015). Fabrication of a superhydrophobic, fire-resistant, and mechanical robust sponge upon polyphenol chemistry for efficiently absorbing oils/organic solvents. Industrial & Engineering Chemistry Research, 54(6), 1842–1848. https://doi.org/10.1021/ie504812p
Husain, A., & Al-Harthi, M. A. (2023). Chemical treatment of oilfield wastewater and the effect of temperature on treatment efficiency: A review. Journal of Petroleum Science and Engineering, 220, 111089. https://doi.org/10.1016/j.petrol.2022.111089
Igunnu, E. T., & Chen, G. Z. (2014). Produced water treatment technologies. International Journal of Low Carbon Technologies, 9(3), 157–177. https://doi.org/10.1093/ijlct/cts049
Jamshidiyan, M., Shirani, A. S., & Alahyarizadeh, G. (2017). Solvothermal synthesis and characterization of magnetic FeO nanoparticle by different sodium salt sources. Materials Science-Poland, 35(1), 50–57. https://doi.org/10.1515/msp-2017-0004
Jani, N. A., Haddad, L., Abdulhameed, A. S., Jawad, A. H., Alothman, Z. A., & Yaseen, Z. M. (2022). Modeling and optimization of the adsorptive removal of crystal violet dye by durian (Durio zibethinus) seeds powder: Insight into kinetic, isotherm, thermodynamic, and adsorption mechanism. Biomass Conversion and Biorefinery. https://doi.org/10.1007/s13399-022-03319-x
Jawad, A. H., Hameed, B. H., & Abdulhameed, A. S. (2023). Synthesis of biohybrid magnetic chitosan-polyvinyl alcohol/MgO nanocomposite blend for remazol brilliant blue R dye adsorption: solo and collective parametric optimization. Polymer Bulletin, 80(5), 4927–4947. https://doi.org/10.1007/s00289-022-04294-z
Jawad, A. H., Rangabhashiyam, S., Abdulhameed, A. S., Syed-Hassan, S. S. A., Alothman, Z. A., & Wilson, L. D. (2022a). Process optimization and adsorptive mechanism for reactive blue 19 dye by magnetic crosslinked chitosan/MgO/Fe3O4 biocomposite. Journal of Polymers and the Environment, 30(7), 2759–2773. https://doi.org/10.1007/s10924-022-02382-9
Jawad, A. H., Saber, S. E. M., Abdulhameed, A. S., Reghioua, A., Alothman, Z. A., & Wilson, L. D. (2022b). Mesoporous activated carbon from mangosteen (Garcinia mangostana) peels by H3PO4 assisted microwave: Optimization, characterization, and adsorption mechanism for methylene blue dye removal. Diamond and Related Materials, 129, 109389. https://doi.org/10.1016/j.diamond.2022.109389
Li, C., Li, J., Wang, N., Zhao, Q., & Wang, P. (2021). Status of the treatment of produced water containing polymer in oilfields: A review. Journal of Environmental Chemical Engineering, 9(4), 105303. https://doi.org/10.1016/j.jece.2021.105303
Li, J., Luo, M., Zhao, C.-J., Li, C.-Y., Wang, W., Zu, Y.-G., & Fu, Y.-J. (2013). Oil removal from water with yellow horn shell residues treated by ionic liquid. Bioresource Technology, 128, 673–678. https://doi.org/10.1016/j.biortech.2012.11.009
Liu, J., Wang, N., Zhang, H., & Baeyens, J. (2019). Adsorption of Congo red dye on FexCo3-xO4 nanoparticles. Journal of Environmental Management, 238, 473–483. https://doi.org/10.1016/j.jenvman.2019.03.009
Lodhia, J., Mandarano, G., Ferris, N., Eu, P., & Cowell, S. (2010). Development and use of iron oxide nanoparticles (part 1): Synthesis of iron oxide nanoparticles for MRI. Biomedical Imaging and Intervention Journal, 6(2). https://doi.org/10.2349/biij.6.2.e12
Lü, T., Qi, D., Zhang, D., Lü, Y., & Zhao, H. (2018). A facile method for emulsified oil-water separation by using polyethylenimine-coated magnetic nanoparticles. Journal of Nanoparticle Research, 20(4), 88. https://doi.org/10.1007/s11051-018-4193-7
Lyu, C., Zhou, X., Lu, X., Zhang, Y., Li, C., Zhou, Q., et al. (2021). The effect of particle size on the interpretation of pore structure of shale by N2 adsorption. Geofluids, 2021, 1–9. https://doi.org/10.1155/2021/8898142
Malhas, R., Al-Ibrahim, Y., Al-Meraj, A., Abdullah, H., & Alshatti, A. (2021). Application of magnetic separation for oil spill remediation and recovery in Kuwait sea water. Desalination and Water Treatment, 209, 114–120. https://doi.org/10.5004/dwt.2021.26498
Meroufel, B., Benali, O., Benyahia, M., Benmoussa, Y., & Zenasni, M. A. (2013). Adsorptive removal of anionic dye from aqueous solutions by Algerian kaolin: Characteristics, isotherm, kinetic and thermodynamic studies. Journal of Materials and Environmental Science, 4(3), 482.
Mittal, A., Roy, I., & Gandhi, S. (2022). Magnetic nanoparticles: An overview for biomedical applications. Magnetochemistry, 8(9), 107. https://doi.org/10.3390/magnetochemistry8090107
Okoro, H. K., Pandey, S., Ogunkunle, C. O., Ngila, C. J., Zvinowanda, C., Jimoh, I., Lawal, I. A., Orosun, M. M., Adeniyi, A. G., et al. (2022). Nanomaterial-based biosorbents: Adsorbent for efficient removal of selected organic pollutants from industrial wastewater. Emerging Contaminants, 8, 46–58. https://doi.org/10.1016/j.emcon.2021.12.005
Oktor, K., Yuzer, N. Y., Hasirci, G., & Hilmioglu, N. (2023). Optimization of removal of phosphate from water by adsorption using biopolymer chitosan beads. Water, Air, & Soil Pollution, 234(4), 271. https://doi.org/10.1007/s11270-023-06230-x
Olajire, A. A. (2020). Recent advances on the treatment technology of oil and gas produced water for sustainable energy industry-mechanistic aspects and process chemistry perspectives. Chemical Engineering Journal Advances, 4, 100049. https://doi.org/10.1016/j.ceja.2020.100049
Osorio, J., Ahmed, R., & Elgaddafi, R. (2022). Oilfield-produced water treatment using bare maghemite nanoparticles. Results in Engineering, 16, 100641. https://doi.org/10.1016/j.rineng.2022.100641
Patel, H., Shah, S., Ahmed, R., & Ucan, S. (2018). Effects of nanoparticles and temperature on heavy oil viscosity. Journal of Petroleum Science and Engineering, 167, 819–828. https://doi.org/10.1016/j.petrol.2018.04.069
Patowary, M., Ananthakrishnan, R., & Pathak, K. (2014). Superhydrophobic and oleophilic barium sulfate material for oil spill cleanups: Fabrication of surface modified sorbent by a one-step interaction approach. Journal of Environmental Chemical Engineering, 2(4), 2078–2084. https://doi.org/10.1016/j.jece.2014.09.007
Patowary, M., Ananthakrishnan, R., & Pathak, K. (2017). Effective oil removal from water by magnetically driven superhydrophobic and oleophilic magnetic titania nanotubes. Environmental Science and Pollution Research, 24(22), 18063–18072. https://doi.org/10.1007/s11356-017-9458-7
Qiao, K., Tian, W., Bai, J., Wang, L., Zhao, J., Du, Z., & Gong, X. (2019). Application of magnetic adsorbents based on iron oxide nanoparticles for oil spill remediation: A review. Journal of the Taiwan Institute of Chemical Engineers, 97, 227–236. https://doi.org/10.1016/j.jtice.2019.01.029
Raj, K. G., & Joy, P. A. (2015). Coconut shell based activated carbon–iron oxide magnetic nanocomposite for fast and efficient removal of oil spills. Journal of Environmental Chemical Engineering, 3(3), 2068–2075. https://doi.org/10.1016/j.jece.2015.04.028
Rajasulochana, P., & Preethy, V. (2016). Comparison on efficiency of various techniques in treatment of waste and sewage water – A comprehensive review. Resource-Efficient Technologies, 2(4), 175–184. https://doi.org/10.1016/j.reffit.2016.09.004
Robati, D. (2013). Pseudo-second-order kinetic equations for modeling adsorption systems for removal of lead ions using multi-walled carbon nanotube. Journal of Nanostructure in Chemistry, 3(1), 55. https://doi.org/10.1186/2193-8865-3-55
Saber, O., Aljaafari, A., Asiri, S., & Batoo, K. (2018). Designing magnetic layered double hydroxides and two-dimensional magnetic nano-nets of cobalt ferrite through a novel approach. Applied Sciences, 8(11), 2099. https://doi.org/10.3390/app8112099
Saruchi, & Kumar, V. (2019). Separation of crude oil from water using chitosan based hydrogel. Cellulose, 26(10), 6229–6239. https://doi.org/10.1007/s10570-019-02539-1
Shahzad, M. W., Ybyraiymkul, D., Burhan, M., & Ng, K. C. (2018). Renewable energy-driven desalination hybrids for sustainability. In M. Eyvaz & E. Yüksel (Eds.), Desalination and Water Treatment. InTech. https://doi.org/10.5772/intechopen.77019
Sobhanardakani, S., Jafari, A., Zandipak, R., & Meidanchi, A. (2018). Removal of heavy metal (Hg(II) and Cr(VI)) ions from aqueous solutions using Fe2O3@SiO2 thin films as a novel adsorbent. Process Safety and Environmental Protection, 120, 348–357. https://doi.org/10.1016/j.psep.2018.10.002
Soliman, E. M., Ahmed, S. A., & Fadl, A. A. (2020). Adsorptive removal of oil spill from sea water surface using magnetic wood sawdust as a novel nano-composite synthesized via microwave approach. Journal of Environmental Health Science and Engineering, 18(1), 79–90. https://doi.org/10.1007/s40201-019-00440-4
Tatarchuk, T., Soltys, L., & Macyk, W. (2023). Magnetic adsorbents for removal of pharmaceuticals: A review of adsorption properties. Journal of Molecular Liquids, 384, 122174. https://doi.org/10.1016/j.molliq.2023.122174
Theurer, J., Ajagbe, O., Osorio, J., Elgaddafi, R., Ahmed, R., Walters, K., & Abbott, B. (2020). Removal of residual oil from produced water using magnetic nanoparticles. SPE Journal, 25(05), 2482–2495. https://doi.org/10.2118/199466-PA
Vargas-Ortiz, J. R., Gonzalez, C., & Esquivel, K. (2022). Magnetic iron nanoparticles: Synthesis, surface enhancements, and biological challenges. Processes, 10(11), 2282. https://doi.org/10.3390/pr10112282
Vlaev, L., Petkov, P., Dimitrov, A., & Genieva, S. (2011). Cleanup of water polluted with crude oil or diesel fuel using rice husks ash. Journal of the Taiwan Institute of Chemical Engineers, 42(6), 957–964. https://doi.org/10.1016/j.jtice.2011.04.004
Wang, H., & Shadman, F. (2013). Effect of particle size on the adsorption and desorption properties of oxide nanoparticles. AICHE Journal, 59(5), 1502–1510. https://doi.org/10.1002/aic.13936
Yousef, R., Qiblawey, H., & El-Naas, M. H. (2020). Adsorption as a process for produced water treatment: A review. Processes, 8(12), 1657. https://doi.org/10.3390/pr8121657
Yu, L., Han, M., & He, F. (2017). A review of treating oily wastewater. Arabian Journal of Chemistry, 10, S1913–S1922. https://doi.org/10.1016/j.arabjc.2013.07.020
Zhu, Q., Tao, F., & Pan, Q. (2010). Fast and selective removal of oils from water surface via highly hydrophobic core−shell Fe2O3@C nanoparticles under magnetic field. ACS Applied Materials & Interfaces, 2(11), 3141–3146. https://doi.org/10.1021/am1006194
Acknowledgements
The authors thank Australian University for using their laboratory and facilities for the present research work.
Funding
The research study and the paper preparation was done without any external funding source.
Author information
Authors and Affiliations
Contributions
Conceptualization, writing original draft, review and editing: R. Malhas. Writing, review and editing: J. El Achkar. Methodology validation: B. Misbah. Final review: S. Al Radhwan. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Malhas, R., El Achkar, J.H., Misbah, B. et al. Optimizing Oil Removal from Oil-Water Emulsions Using Novel Iron Oxide Magnetic Nanoparticles. Water Air Soil Pollut 234, 564 (2023). https://doi.org/10.1007/s11270-023-06590-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06590-4