Abstract
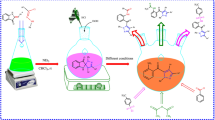
The alkaloid carbazole is one of the relevant structural motifs in drug discovery. Indeed, carbazoles have been largely investigated for their various biological properties. In this article, in order to ensure the possibility of carrying out the carbazole N-alkylation synthesis reaction at the synthesis laboratory level and to gain access to a new active biomolecule, we present the theoretical study of strategies for the synthesis of an N-alkylcarbazole using the DFT method at B3LYP, with the basis set 6-31G(d,p). In fact, this research has enabled us to establish a simple, economic, and efficient synthesis strategy for this nitrogenous heterocycle with an excellent yield, by N-alkylation between carbazole and propargyl bromide under different experimental conditions. We have developed an environmentally respectful synthesis process, a gentle packaging protocol and a practical procedure for the preparation of this polycyclic aromatic compound. The structure of the synthesized compound was determined by spectroscopic methods: 1H-NMR, 13C-NMR, and MS data. The theoretical results corroborate with experiences. The study of the substitution effect on the optical properties of reagents and products shows that N-alkylation has an influence on the optical properties.





Similar content being viewed by others
Availability of data and materials
All data sets were generated and analyzed in the current study.
References
Greger H (2017) Phytocarbazoles: alkaloids with great structural diversity and pronounced biological activities. Phytochem Rev 16(6):1095–1153
Schmidt AW, Reddy KR, Knölker H-J (2012) Occurrence, biogenesis, and synthesis of biologically active carbazole alkaloids. Chem Rev 112(6):3193–3328
Caruso A et al (2019) Carbazole derivatives as antiviral agents: an overview. Molecules 24.10:1912
Salih N, Salimon J, Yousif E (2016) Synthesis and antimicrobial activities of 9H-carbazole derivatives. Arab J Chem 9:S781–S786
Knölker H-J, Reddy KR (2002) Isolation and synthesis of biologically active carbazole alkaloids. Chem Rev 102(11):4303–4428
Rüegg UT, Gillian B (1989) Staurosporine, K-252 and UCN-01: potent but nonspecific inhibitors of protein kinases. Trends Pharmacol Sci 10(6):218–220
Montoia, Andreia et al (2014) “Antiplasmodial activity of synthetic ellipticine derivatives and an isolated analog.” Bioorganic Med Chem Lett 24.12:2631–2634
Patel, Om PS et al (2016) “Naturally occurring carbazole alkaloids from Murraya koenigii as potential antidiabetic agents.” J Nat Prod 79.5:1276–1284
Hagiwara, Hitomi et al (2000) “A novel total synthesis of antibiotic carbazole alkaloid carbazomycin G.” Tetrahedron 56.32:5807–5811
Achamlale S, Mabrouk H, Elachqar A, El Hallaoui A, El Hajji S, Bellan J, Mazières MR, Wolf JG, Pierrot M (2007) Phosphorus, sulfur, and silicon and the related elements 182:357. https://doi.org/10.1080/10426500600919959
Mabrouk El, Arrousse N, Korchi A, Lachgar M, Oubair A, Elachqar A, Jabha M, Lachkar M, El hajjaji F, Rais Z, Taleb M (2020) Chem Res Chinese Universities (in press). https://doi.org/10.1007/s40242-020-0173-4
Mabrouk E et al (2010) One-pot regioselective synthesis of n-benzoyl 2-amino-3, 4-dihydro-3-oxo-2h-1, 4-benzothiazines. Orient J Chem 26(4):1249
Mabrouk E, Elachqar A, El Hallaoui A, El Hajji S, Martinez J, Rolland V (2013) Arab J Chem 6:93. https://doi.org/10.1016/j.arabjc.2010.09.023
Arrousse N, Mabrouk El, Hammouti B, El hajjaji F, Rais Z, Taleb M (2020) Mediterr J Chem 10(5):477. https://doi.org/10.13171/mjc10502005151417feh
Arrousse N, Mabrouk El, Ismail Alaoui K, El hajjaji F, Rais Z, Taleb M (2020) SN Applied Sciences 2(5). https://doi.org/10.1007/s42452-020-2782-4
Arrousse N, Salim R, AL Houari G, EL hajjaji F, Zarrouk A, Raisa Z, Taleb M, Dheeraj Singh Chauhan and Quraish MA (2020) J Chem Sci 132:112 . https://doi.org/10.1007/s12039-020-01818-w
Arrousse N, Salim R, Kaddouri Y, Zarrouk A, Zahri D, El Hajjaji F, Touzani R, Taleb M, Jodeh S (2020) Arab J Chem 13(7):5949
Arrousse N, Mabrouk El, Hammouti B, El hajjaji F, Rais Z, Taleb M (2020) Int. J. Corros. Scale. Inhib 9(2):661. https://doi.org/10.17675/2305-6894-2020-9-2-18
Zhu D, Shi S, Qian R (1986) Polymerization of 3-(9-carbazolyl) propyne and 1, 6-bis (9-carbazolyl)-2, 4-hexadiyne with simultaneous charge transfer intercalation in molten iodine. Die Makromolekulare Chemie, Rapid Communications 7(5):313–317
Nakano M, Masuda T, Higashimura T (1995) Homo-and copolymerizations of 3-(N-carbazolyl)-1-propyne and its homologues by Mo and W catalysts. Polym Bull 34:191–197
Feringa BL, Browne WR (2011) Molecular Switches, Wiley VCH, Weinheim, Germany 2:792. ISBN 978–3–527–63441–5
Jabha et al (2021) “The effect of substitution and polymerization of 2, 7-Divinylcarbazole-benzo-bis-thiadiazole on optoelectronic properties: a DFT study.” Orbital: Electron J Chem 291–300
El Alaoui, Abdellah et al (2022) “Theoretically studying the optoelectronic properties of oligomers based on 2.7-divinyl-cabazole.” Eclética Química 47.1:40–54
Mohamed J (2018) “Study optoelectronic and geometric properties of new compounds based on carbazole-thiophene bridged for solar cells.” Orbital: Electron J Chem 552–560
Domingo LR, Pérez P (2011) The nucleophilicity N index in organic chemistry. Org Biomol Chem 9(20):7168–7175
El Yaktini A, Lachiri A, El Faydy M, Benhiba F, Zarrok H, El Azzouzi M, Zertoubi M, Azzi M, Lakhrissi B, Zarrouk A (2018) Int J Corros Scale Inhib 7(4):609. https://doi.org/10.17675/2305-6894-2018-7-4-9
Gazquez JL, Mendez F (1994) The hard and soft acids and bases principle: an atoms in molecules viewpoint. J Phys Chem 98:4591–4593
Bogdal D, Pielichowski J, Jaskot K (1997) New synthesis method of N-alkylation of carbazole under microwave irradiation in dry media. Synth Commun 27(9):1553–1560
Houssine, Mabrouk El et al (2010) “Synthesis of new racemic α, α-diaminocarboxylic ester derivatives.” Molecules 15.12:9354–9363
Mabrouk E, Elachqar A, Hallaoui El, Molbank A (2013) M792. https://doi.org/10.3390/M792
Oubair A, Fihi R, Chebabe D (2017) Int J Innov Appl Stud 20(4):1221
Oubair A, Fihi R, Mazouz H (2016) Bulletin of the Royal Society of Sciences of Liège 85:119
Acknowledgements
This work is financially supported by the National Center for Scientific and Technical Research (CNRST), Morocco (PROTARS D13 / 03).
Author information
Authors and Affiliations
Contributions
Conceptualization: Mabrouk, E. M. Jabha. Data curation: El Mesky, M, Zgueni, H., Youssefi Y. Formal Analysis: Mabrouk, E.; Amri, N. Funding acquisition: Not applicable. Investigation: Mabrouk, E.; Amri, N, Chebabe, D. Methodology: Mabrouk, E.; Jabha, M. Project administration: Oubair, A; Azrour, M. Resources: Mabrouk, E.; El Mesky, M. Software: Not applicable. Supervision: Mabrouk, E.; Jabha, M. Validation: Mabrouk, E.; Amri, N. Visualization: Mabrouk, E.; Azdouz, M. Writing—original draft: Mabrouk, E.; Azdouz, M. Writing—review and editing: Mabrouk, E.; Oubair, A; Znini M.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jabha, M., Mesky, M.E., Zgueni, H. et al. The intelligence way of economical synthesis strategies of an N-alkylcarbazole. Struct Chem 35, 605–614 (2024). https://doi.org/10.1007/s11224-023-02214-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02214-z