Abstract
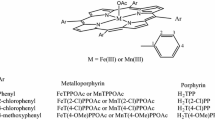
The catalytic efficiency of modified tetraphenylporphyrins with various metals (Mn, Fe, Co, Ni and Zn) and different electron-donating substituents (–OCH3, –OH, –N(CH3)2, –SO3Na) at the phenyl para-positions using hydrogen peroxide (H2O2, 30 wt%) as the oxidant has been researched in the epoxidation of unsaturated fatty acid methyl esters (FAMEs) under mild conditions. In the present system, the co-catalyst effect of inorganic salts had a crucial role in enhancing catalytic performance. According to the yields, the catalytic activities of metalloporphyrins were in the following order: Mn(TPP)Cl ≈ Fe(TPP)Cl > CoTPP > NiTPP > ZnTPP, and Mn(TPP)Cl > Mn(TMOPP)Cl ≈ Mn(THPP)Cl > Mn(TDMPP)Cl > Mn(TPPS4)Cl. Moreover, metalloporprhyrins after modification by electron-donating substituents displayed promoted thermolysis temperatures. Remarkably, the formation of intermediate porMnV=O has also been indicated, which was verified by both a kinetic study of FAMEs epoxidation and UV–Vis spectroscopy.








Similar content being viewed by others
References
Ortiz de Montellano PR (2010) Hydrocarbon hydroxylation by cytochrome P450 enzymes. Chem Rev 110:932–948
Perez MJ, Cederbaum AI (2002) Antioxidant and pro-oxidant effects of a manganese porphyrin complex against CYP2E1-dependent toxicity. Free Radical Bio Med 33:111–127
Dolphin D, Traylor TG, **e LY (1997) Polyhaloporphyrins: unusual ligands for metals and metal-catalyzed oxidations. Accounts Chem Res 30:251–259
Adler AD (1970) On the preparation of metalloporphyrins. J Inorg Nucl Chem 32:2443–2445
Groves JT, Nemo TE (1983) Epoxidation reactions catalyzed by iron porphyrins. Oxygen transfer from iodosylbenzene. J Am Chem Soc 105:5786–5791
Machado GS, Ucoski GM, Lima OJd, Ciuffi KJ, Wypych F, Nakagaki S (2013) Cationic and anionic metalloporphyrins simultaneously immobilized onto raw halloysite nanoscrolls catalyze oxidation reactions. Appl Catal A Gen 460–461:124–131
Ucoski GM, Nunes FS, DeFreitas-Silva G, Idemori YM, Nakagaki S (2013) Metalloporphyrins immobilized on silica-coated Fe3O4 nanoparticles: magnetically recoverable catalysts for the oxidation of organic substrates. Appl Catal A Gen 459:121–130
Wang Y, Jiang P, Zhang W, Zheng J (2013) 15-(4-Carboxyphenyl)-10,15,20-triphenylporphyrin manganese(III) chloride grafted on magnetic polyglycidyl methacrylate as biomimetic catalyst and their catalytic activity. Appl Surf Sci 270:531–538
Balasubramanian PN, Sinha A, Bruice TC (1987) A kinetic study of the epoxidation of 2,3-dimethyl-2-butene by tert-butyl hydroperoxide catalyzed by imidazole ligated (meso-tetraphenylporphinato)manganese(III). J Am Chem Soc 109:1456–1462
W-j Zhang, P-p Jiang, P-b Zhang, Liu P (2012) Immobilization of tetraphenylporphyrin manganese (III) chloride in HMS modified by Zr, Cu, and Zn oxides and their catalytic activity. Catal Lett 142:1512–1519
Reginato G, Di Bari L, Salvadori P, Guilard R (2000) Chiral atropisomeric metalloporphyrins in the enantioselective styrene epoxidation. Eur J Org Chem 2000:1165–1171
Morris GA, Nguyen ST, Hupp JT (2001) Enhanced activity of enantioselective (salen)Mn(III) epoxidation catalysts through supramolecular complexation. J Mol Catal A Chem 174:15–20
Ellis S, Kozhevnikov IV (2002) Homogeneous oxidation of methyl isobutyrate with oxygen catalysed by metal complexes: polyoxometalates versus metalloporphyrins and metallophthalocyanines. J Mol Catal A Chem 187:227–235
Guo C-C, Liu Q, Wang X-T, Hu H-Y (2005) Selective liquid phase oxidation of toluene with air. Appl Catal A Gen 282:55–59
Ventrice P, Ventrice D, Russo E, De Sarro G (2013) Phthalates: european regulation, chemistry, pharmacokinetic and related toxicity. Environ Toxicol Phar 36:88–96
Zhou J, Cai ZH, **ng KZ (2011) Potential mechanisms of phthalate ester embryotoxicity in the abalone Haliotis diversicolor supertexta. Environ Pollut 159:1114–1122
Biermann U, Friedt W, Lang S, Luhs W, Machmuller G, Metzger JO, Klaas MR, Schafer HJ, Schneider MP (2000) New syntheses with oils and fats as renewable raw materials for the chemical industry. Angew Chem Int Edit 39:2206–2224
Li Y, Zhuang M, Li T, Shi N (2009) Neurobehavioral toxicity study of dibutyl phthalate on rats following in utero and lactational exposure. J Appl Toxicol 29:603–611
Du G, Tekin A, Hammond EG, Wood LK (2004) Catalytic epoxidation of methyl linoleate. J Am Oil Chem Soc 81:477–480
Adler AD, Longo FR, Finarelli JD, Goldmacher J, Assour J, Korsakoff L (1967) A simplified synthesis for meso-tetraphenylporphine. J Org Chem 32:476
Hansen CB, Hoogers GJ, Drenth W (1993) Anchored manganese and ruthenium porphyrins as catalysts in the decomposition of cyclohexyl hydroperoxide. J Mol Catal 79:153–163
Guo C–C, Chu M-F, Liu Q, Liu Y, Guo D-C, Liu X-Q (2003) Effective catalysis of simple metalloporphyrins for cyclohexane oxidation with air in the absence of additives and solvents. Appl Catal A Gen 246:303–309
Wang MZ, Zhou CY, Wong MK, Che CM (2010) Ruthenium-catalyzed alkylation of indoles with tertiary amines by oxidation of a sp3 C–H bond and Lewis acid catalysis. Chem-Eur J 16:5723–5735
Exner O (1988) Correlation analysis of chemical data. SNTL–Plenum Press, New York
Chen H, Li H, Lin J-M (2012) Determination of ammonia in water based on chemiluminescence resonance energy transfer between peroxymonocarbonate and branched NaYF4:yb3 +/Er3 + nanoparticles. Anal Chem 84:8871–8879
Zhang L, Lu B, Lu C (2013) Chemiluminescence sensing of aminothiols in biological fluids using peroxymonocarbonate-prepared networked gold nanoparticles. Analyst 138:850–855
Araghi M, Bokaei F (2013) Manganese(III) porphyrin supported on multi-wall carbon nanotubes: a highly efficient and reusable biomimetic catalyst for oxidative decarboxylation of α-arylcarboxylic acids and oxidation of alkanes with sodium periodate. Polyhedron 53:15–19
Srour H, Jalkh J, Le Maux P, Chevance S, Kobeissi M, Simonneaux G (2013) Asymmetric oxidation of sulfides by hydrogen peroxide catalyzed by chiral manganese porphyrins in water/methanol solution. J Mol Catal A Chem 370:75–79
Hajimohammadi M, Bahadoran F, Davarani SSH, Safari N (2010) Selective photocatalytic epoxidation of cyclooctene by molecular oxygen in the presence of porphyrin sensitizers. React Kinet Mech Cat 99:243–250
Madadi M, Rahimi R (2012) Zeolite-immobilized Mn(III), Fe(III) and Co(III) complexes with 5,10,15,20-tetra(4-methoxyphenyl)porphyrin as heterogeneous catalysts for the epoxidation of (R)-(+)-limonene: synthesis, characterization and catalytic activity. React Kinet Mech Cat 107:215–229
Hamidipour L, Farzaneh F (2013) Cobalt metal organic framework as an efficient heterogeneous catalyst for the oxidation of alkanes and alkenes. React Kinet Mech Cat 109:67–75
Gunter MJ, Turner P (1991) Metalloporphyrins as models for the cytochromes-P-450. Coordin Chem Rev 108:115–161
Traylor TG (1991) Kinetics and mechanism studies in biomimetic chemistry–metalloenzyme model systems. Pure Appl Chem 63:265–274
Cai S, Wang L (2011) Epoxidation of unsaturated fatty acid methyl esters in the presence of SO3H-functional brφnsted acidic ionic liquid as catalyst. Chinese J Chem Eng 19:57–63
Saifuddin N, Zhan LW, Ning KX (2011) Heat-modeling of microwave assisted epoxidation of palm acid oil. Am J Applied Sci 8:217–229
Merlau ML, Grande WJ, Nguyen ST, Hupp JT (2000) Enhanced activity of manganese(III) porphyrin epoxidation catalysts through supramolecular complexation. J Mol Catal A Chem 156:79–84
Song WJ, Seo MS, George SD, Ohta T, Song R, Kang MJ, Tosha T, Kitagawa T, Solomon EI, Nam W (2007) Synthesis, characterization, and reactivities of manganese(V)-oxo porphyrin complexes. J Am Chem Soc 129:1268–1277
Acknowledgments
This work was supported financially by the National “Twelfth Five-Year” Plan for Science & Technology (2012BAD32B03), the National Natural Science Foundation of China (20903048) and the Innovation Foundation in Jiangsu Province of China (BY2013015-10).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, W., Jiang, P., Wu, J. et al. Catalytic epoxidation of fatty acid methyl esters by modified metalloporphyrins with variable metals and electron-donating substituents. Reac Kinet Mech Cat 112, 147–158 (2014). https://doi.org/10.1007/s11144-014-0676-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-014-0676-4