Abstract
Background and aims
Several lines of evidence indicate that arbuscular mycorrhizal (AM) and ectomycorrhizal (ECM) associations can have different effects on soil nutrient dynamics. Some lineages of ECM fungi can extract N from organic matter, with varying impacts on decomposers, soil carbon pools, mineral N availability, and plants that lack ECM. However, these effects are not always observed, and it is not clear how they are mediated by environmental factors.
Methods
We used Plant Root Simulator probes to compare soil availability of a wide range of nutrients beneath replicated 30-yr old plantations of Chilean Nothofagus (ECM) and Cupressaceae (Austrocedrus, Fitzroya: AM) on a lowland temperate site. Probes were buried for two 8-week periods in early spring and late summer. We also compared understorey composition beneath plantations, to test for evidence of different successional trajectories beneath Nothofagus and Cupressaceae.
Results
Soil organic carbon, total N and total phosphorus did not differ significantly between Nothofagus and Cupressaceae stands. Redundancy analysis revealed significant effects of both plantation type (Nothofagus vs. Cupressaceae) and season on overall mineral nutrient availability. Mineral N availability did not differ significantly between Nothofagus and Cupressaceae plots, but pH and calcium availability were significantly lower beneath Nothofagus. Manganese (Mn) was much more available beneath Nothofagus stands, which might reflect abundant Mn-peroxidase, a key enzyme involved in breakdown of lignin by ECM fungi. Understorey composition varied considerably between individual plantations, but did not differ significantly between Nothofagus and Cupressaceae plantations.
Conclusions
Despite an overall effect on the stoichiometry of nutrient availability, we found little evidence of modification of the local N cycle by ECM fungi, or of divergent regeneration patterns beneath AM and ECM plantations. This might reflect the relatively N-rich character of this site, and/or mycorrhizal effects being counteracted by leaf trait differences between Chilean Cupressaceae and Nothofagus species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autotrophic plants have evolved a variety of different nutrient acquisition syndromes, and the relative viability of these syndromes appears to vary along environmental gradients. Relationships with climate have been documented, with ectomycorrhizal (ECM) plants usually dominant in climates that are unconducive to decomposition during much of year, and arbuscular mycorrhizas (AM) reported to be more prevalent in benign climates (Steidinger et al. 2019). There are also frequent reports of ECM association with soils that are poor in nitrogen (N) (Read 1991; Tsai et al. 2014; Rowlingson et al. 2022), or poor in nutrients in general (Wardle 1991; Niiyama et al. 1999; Corrales et al. 2018).
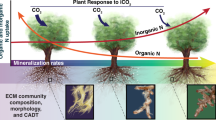
Although environment clearly influences fitness of different nutrient acquisition syndromes, these syndromes themselves can also alter local nutrient cycling. Several lines of evidence indicate AM and ECM associations can have different effects on soil nutrient dynamics. Some lineages of ECM fungi are able to extract nitrogen (N) and possibly other nutrients from soil organic matter (Read and Perez-Moreno 2003; Pellitier and Zak 2018; Romero-Olivares et al. 2021). This competition for N with saprophytes may slow decomposition (Gadgil and Gadgil 1971), increase the size of soil carbon C pools (Orwin et al. 2011; Averill et al. 2014), depress nutrient mineralization rates, and thus reduce availability of N (and potentially other nutrients) to plants that lack ECM associations (Corrales et al. 2016; Lin et al. 2017, 2018). This short-circuiting of the nitrogen cycle may thus set up a positive feedback mechanism favouring continued local dominance by ECM plants, resulting in sharp ecotones between AM and ECM-dominant vegetation in the absence of obvious discontinuities in the physical environment, and raising the possibility of mycorrhiza-mediated alternative stable states (Corrales et al. 2016; Lu and Hedin 2019; Lusk and Atatoa Carr 2023).
ECM effects on soil C and N dynamics are not uniformly observed, and it is not clear how these effects vary with environment or with plant taxonomy (Fernandez and Kennedy 2016). Data obtained by comparing soil chemistry under patches of AM and ECM trees in natural vegetation can be difficult to interpret, because of the possibility of sorting of AM and ECM species along pre-existing fine-scale environmental gradients (Nadelhoffer et al. 1983; Boerner and Koslowsky 1989; Hall et al. 2020). Comparisons of adjacent plantations of known history provide more incisive tests, and have frequently found lower N availability under ECM plantations than AM plantations (Lin et al. 2017). The ECM tree species included in the studies reviewed by Lin et al. (2017) belong predominantly to two northern-hemisphere families, Pinaceae and Fagaceae.
In the humid temperate forests of the southern hemisphere, Nothofagus (Nothofagaceae) is the only ECM genus that occurs widely in mature stands (Wardle 1991; Godoy and Marín 2019). Although the effects of Nothofagus on soil C and N dynamics have not been examined to date, its tendency to form monodominant stands (Wardle 1984) is reminiscent of the behaviour of some ECM tropical trees that appear to elevate soil C:N ratios and depress N mineralization (Corrales et al. 2016; Hall et al. 2020). Here we compare soil nutrient availability beneath replicated ~ 30-year-old stands of Chilean native Nothofagus and Cupressaceae (AM) planted on a relatively homogenous lowland site in south-central Chile. We asked (1) if ECM plantations of Nothofagus dominance has resulted in higher soil total C and C:N, and lower nutrient availability than beneath the adjacent AM plantations of Cupressaceae; and (2) if understorey floristic composition indicates different successional trajectories in the ECM and AM stands, foreshadowing the development of alternative stable states.
Materials and methods
Study site
The plantations we studied were established between 1989 and 1995 by Prof. Claudio Donoso (Universidad Austral de Chile) at Senderos del Bosque (39.867 S, 73.157 W) on the outskirts of Valdivia in south-central Chile. A suite of small plantations of Chilean native tree species were established at between 60 and 100 m asl on a west-facing slope. Slopes were mostly in the 15–20° range. The red clay soils at the site belong to Los Ulmos soil series (CIREN 1999), range in depth from 50 to 100 cm, and are developed over Permian-Triassic schists. Valdivia has an oceanic temperate climate, with a mean annual temperature of 11.7 °C and a mean annual precipitation of 1,750 mm (Dirección Meteorológica de Chile 2001). Seasonal thermal amplitude is low: the mean daily high of the warmest month (February) is 23.8° C, and the mean daily low of the coldest month (July) is 4.0 °C. Seasonal variation in precipitation is much more pronounced, with > 60% falling during the winter months of May to August. Summer is the driest season, both January and February averaging < 50 mm. Snowfalls are rare.
The vegetation present on the site at the time plantations were established had been severely degraded by a long history of wood-cutting, leaving a low cover (2–3 m tall) of the bamboo Chusquea quila (Poaceae) and shrubs such as Ugni molinae (Myrtaceae) and Gaultheria spp. (Ericaceae), with scattered emergent trees up to 10 m tall, mostly Laurelia sempervirens (Atherospermaceae) and Gevuina avellana (Proteaceae). In less disturbed native forest stands on similar sites near Valdivia, the main canopy species are the evergreens Aextoxicon punctatum (Aextoxicaceae), Eucryphia cordifolia (Cunoniaceae), L. sempervirens, Luma apiculata (Myrtaceae) and Podocarpus saligna (Podocarpaceae), with emergent deciduous Nothofagus obliqua (Nothofagaceae) and evergreen N. dombeyi.
Establishment of plantations
A mosaic of 0.3 to 0.5 ha plantations of a range of Chilean native timber species was established on the site. Individual plantations were sited opportunistically on gentle spurs, avoiding gullies. These included four Nothofagus spp. stands (ECM) and three Cupressaceae stands (AM) that we later used for our study (Table 1; Fig. 1). The planted Nothofagus species were N. dombeyi and N. obliqua, and the Cupressaceae were Austrocedrus chilensis and Fitzroya cupressoides. Small gaps were opened in the shrub cover to enable planting of seedlings on a 2 m x 2 m grid, equivalent to a density of 2,500 plants ha−1, in accordance with standard Chilean forestry practice (Donoso et al. 1998). Bare-rooted two-year-old seedlings were planted; seedlings of Nothofagus were two years old and typically 60–80 cm tall, and those of Cupressaceae were three years old and 30–40 cm tall. Six of the plantations were monospecific, the seventh being a mixture of two Nothofagus species (Table 1). Plantations were not thinned, pruned, or weeded; and no fertilizer was applied.
Spatial arrangement of plantations of Cupressaceae (Austrocedrus chilensis, Fitzroya cupressoides) and Nothofagus (N. dombeyi, N. obliqua) at Senderos del Bosque, Chile. See Table 1 for explanations of plantation names
Although non-target tree species had become abundant in some plantations by the time of sampling, they did not compromise our intended comparison of AM- versus ECM-dominant stands. Growth of residual L. sempervirens, and new recruitment of fast-growing species such as Embothrium coccineum (Proteaceae), resulted in considerable contributions of non-target species to the basal area of some plantations (Table 1). Most notably, basal area of the planted species (F. cupressoides) on plot FITCUP2 was exceeded by that of L. sempervirens. However, ectomycorrhizal trees (Nothofagus) contributed negligible basal area to the three Cupressaceae plantations (Table 1). AM tree species contributed > 85% to the basal areas of all three of these plantations, non-mycorrhizal Proteaceae comprising the remainder. Similarly, ECM species contributed at least 68% of the basal area of all four Nothofagus plantations (Table 1). AM Chusquea bamboos were present in the understories of all stands; although we did not quantify their abundance, visual estimates suggested up to c. 20% cover of Chusquea spp. in the Cupressaceae stands, and somewhat less in the Nothofagus stands.
Soil analyses
In 2021 we obtained soil samples from beneath each of the seven plantations, to assess similarity of total soil nutrient capital and pH. Given that we did not have initial soil data from 30 years previously, we assume that soils were overall similar among stands with AM trees and with ECM trees before plantations. The long axis of most plantations ran approximately east to west; soil was sampled on three north-south transects randomly spaced between 20 and 40 m apart along this east-west axis. At four randomly spaced points on each transect, a 20 mm diameter soil augur was used to extract the uppermost 120 mm of the mineral soil. Soil from these 12 points was homogenized and submitted as a single sample from each plantation.
Soils were dried at 60° C for 72 h, and sent for analysis at Hill Laboratories, Hamilton, New Zealand. pH was measured potentiometrically in a 1:2 (v/v) soil:water slurry. Organic C and total N were estimated by Dumas combustion. Total P was estimated by nitric/hydrochloric digestion, followed by inductively coupled plasma-optical emission spectroscopy.
Mineral nutrient availability
Plant Root Simulator (PRS®) probes (Western Ag Innovations Inc., Saskatoon, Canada) were used to measure ion availability in the mineral soil beneath each of the seven plantations. The probes consist of 10 cm2 ion exchange resin membranes held in plastic supports that are inserted vertically into the soil. Anion probes have a positively-charged membrane to attract and adsorb all negatively-charged anions, and cation probes a negatively charged membrane to adsorb all positively-charged ions.
PRS® probes were buried for two eight-week periods. The first burial period (15/08/21 to 14/10/21) was intended to represent early growing season conditions when mineralization rates are likely to be temperature-limited. The second burial period (14/01/22 to 11/03/22) was intended to represent late growing season conditions when mineralization rates are likely to be limited primarily by water availability, as the summer months of January and February are also the driest months in Valdivia.
A pair of PRS® probes (anion + cation) was buried at each of the same sampling points where soil samples were taken. This meant four pairs of probes were buried along each of three transects in each plantation during each burial period. Probes were inserted into slots cut in the soil with a garden knife, leaving the c. 1 cm of the plastic support protruding from the soil surface. Soil was tamped closely around the probe, to ensure soil contact with the ion exchange membrane. As the ion exchange membrane begins 6 cm below the top of the plastic support, most or all of the membrane was in contact with the mineral soil, rather than the surface organic horizons. Brightly-coloured flagging was attached to the top of each probe, to facilitate retrieval eight weeks later.
We successfully retrieved 330 (98.2%) of 336 buried probes. After retrieval, probes were cleaned with a nailbrush and ultrapure water, and submitted to WesternAg Innovations for analysis. The three pairs of probes inserted on each transect were treated as a single sample: they were submitted in the same bag to the laboratory, where they were eluted together. This meant four estimates of the availability of each ion were obtained from each plantation during each burial period.
Understorey composition
Understorey composition was surveyed on the same plots used for measurement of stand basal area (Table 1). We recorded frequencies of arborescent species ≥ 100 cm tall and < 5.0 cm diameter, excluding Chusquea bamboos.
Statistics
All statistical analysis was performed in R (R core team 2022). The overall effect of dominant taxon (Nothofagus vs. Cupressaceae) and season (spring vs. summer) on mineral nutrient availability was visualised using principal components analysis (function prcomp with scale = TRUE), and tested using redundancy analysis (rda in package vegan, Oksanen et al. 2019). As redundancy analysis did not account for non-independence of transects within stands, we took the geometric mean value of the pooled probes for each of three transects within each stand at each season for this analysis. As environmental differences associated with the spatial arrangement of our stands could also potentially influence nutrient availability, we also tested the effect of spatial distance using RDA. As we found no significant effect of spatial distance (P = 0.134), this variable was not explored further in our analyses or interpretations.
After finding overall significant effects of taxon and season in redundancy analysis, we tested the significance of their effects on availability of each nutrient. A linear mixed effect model with a random term was used to account for the three replicate samples within each combination of taxon and season. “Taxon” was treated as a categorical variable (Nothofagus vs. Cupressaceae). We used model simplification, using likelihood ratio tests to determine the significance of each term (function anova on lme models) for each soil nutrient. As the residuals of many of the soil measurements were non-normally distributed, we rank-transformed all variables before tests.
We used Permutational multivariate analysis of variance (PERMANOVA, vegan package) to test for differences in understorey composition between Nothofagus and Cupressaceae plantations. Non-metric multidimensional scaling (NMDS, vegan package) was used to provide a visual representation of variation in understorey composition.
Results
Soil analyses
Plantations of Cupressaceae and Nothofagus did not differ significantly in soil C, N or P content (Table 2). According to the criteria described by Blakemore et al. (1987) for similar south-temperate ecosystems in New Zealand, total N levels beneath the plantations were moderate to high; although C:N ratios were also rather high. Total P levels were low to moderate according to the same criteria. Average soil pH beneath Nothofagus stands (5.2) was significantly lower than that measured beneath Cupressaceae stands (5.7).
Mineral nutrient availability measured with PRS® probes
We removed cadmium from our dataset, as availability of this element was generally below the method detection limit. The remaining ions measured by the probes were ammonium (NH4+), nitrate (NO3−), calcium (Ca2+), magnesium (Mg2+), potassium (K+), phosphate (PO43−), iron (Fe2+/Fe3+), manganese (Mn2+), copper (Cu2+), zinc (Zn2+), boron (B3+), sulphate (SO42−), lead (Pb2+) and aluminium (Al3+).
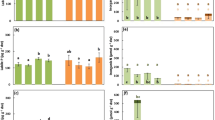
Redundancy analysis showed significant effects of taxon (Cupressaceae vs. Nothofagus: F1,11 = 2.56, P = 0.046) and season (F1,11 = 4.70, P = 0.005) on mineral nutrient availability, the two constrained axes explaining 39.8% of variance. This was reflected in the PCA analysis (Fig. 2), which showed a strong separation by season. One of the four Nothofagus stands (NOTSPP) clustered with Cupressaceae stands in the ordination, while the other three Nothofagus stands clustered together. Neither N nor P availability contributed strongly to either of the first two principal components (Table 3). Two variables contributed strongly to the second component: Ca2+ (0.550) and Mn2+ (-0.506).
PCA ordination of nutrient availability data obtained from soil beneath seven plantations at Senderos del Bosque, Chile. PRS® probes were used to measure availability of the following mineral nutrients over two 8-week periods during the 2021-22 growing season: NH4+, NO3−, Ca, Mg, K, P, Fe, Mn, Cu, Zn, B, S, Pb and Al. PC1 represents 37.4 and PC2 17.3% of the variance, all other axes explain < 10% of variance
In univariate analysis, season was retained as a significant predictor in the best models for all soil variables except Mn and B (Table 4). Dominant taxon was only retained in the best model for three soil variables: Ca, Mn, and as part of a significant interaction with season for NH4+. Ca availability was higher beneath Cupressaceae stands, and Mn was higher beneath Nothofagus stands (Fig. 3). Availability of both Ca and Mn was higher in spring than in summer (Fig. 3), as was true of most other ions (Table S1).
Understorey composition
Understorey composition differed rather idiosyncratically across individual plantations (Table S2). Despite a lack of overlap between convex hulls fitted to the NMDS ordination (Fig. 4), PERMANOVA did not show a significant difference between average understorey composition of Nothofagus and Cupressaceae plantations (R2 = 0.24, F = 1.57, P = 0.183). The commonest understorey species were (in descending order) Luma apiculata, Embothrium coccineum (Proteaceae), Eucryphia cordifolia and Gaultheria mucronata; these four species accounting for > 54% of all saplings on the seven plots (Table S2).
Discussion
We found that mineral nutrient availability beneath plantations of Chilean Cupressaceae and Nothofagus had become stoichiometrically distinct within 30 years (Fig. 2). Lower Ca availability in the soil A horizon beneath Nothofagus stands might reflect leaching of base cations as a result of acidification (Binkley and Valentine 1991), though it is unclear why similar losses of K and Mg were not apparent (Table 2). Similarly, a study of 50-yr old plantations reported significantly higher Ca availability beneath AM Fraxinus than beneath ECM Picea (Binkley and Valentine 1991), and a comparison of 34-yr old plantations of four conifer species found Ca availability to be highest beneath the only AM species, Thuja plicata (Prescott et al. 2000). Alternatively, differences in Ca availability beneath Cupressaceae and Nothofagus stands could be actively driven by differences in Ca cycling. Kiilsgaard et al. (1987) found that Ca concentrations in leaf litter of AM North American conifers (Cupressaceae and Taxodiaceae) were two- to three-fold higher than those of ECM Pinaceae.
Our finding of higher Mn availability beneath Nothofagus stands than beneath AM-dominant stands (Fig. 3) may reflect Mn peroxidase activity associated with ECM fungi. Mn peroxidase is one of the main ligninolytic enzymes produced by Basidiomycetes (Hofrichter 2002), and some ECM lineages including Cortinarius have retained genes coding for Mn peroxidase production (Chen et al. 2001; Pellitier and Zak 2018). Mn peroxidase oxidizes the manganese (II) ions (Mn2+) in wood and soil organic matter to highly reactive Mn3+, which is stabilized by fungal chelators such as oxalic acid. Chelated Mn3 + in turn attacks phenolic lignin structures (Hofrichter 2002). Mn peroxidase may be one of the primary drivers of the organic N economy that often develops in ECM forests (Lindahl and Tunlid 2015; Sterkenburg et al. 2018; Pellitier and Zak 2021).
Despite overall differences in the stoichiometry of soil nutrient availability, and probable evidence of elevated Mn peroxidase activity beneath Nothofagus stands, we found no clear evidence of differences in soil C and N dynamics beneath AM and ECM stands. There was no evidence of a depression of mineral N availability beneath Nothofagus stands (Table 3; Fig. 3), or of elevated C:N ratios (Table 2). Furthermore, understorey composition showed little evidence of divergent successional trajectories beneath Nothofagus and Cupressaceae plantations (Fig. 4, Table S2). Ericaceous shrubs are common in the understories of Nothofagus forest on low-N sites New Zealand (Wardle 1984; Rowlingson et al. 2022), and at high elevations and high latitudes in Chile (Cuevas 2000; Romanyà et al. 2005; Fuentes-Ramirez et al. 2020). This may reflect the ability of ericoid mycorrhizal fungi to access organic N sources (Read and Perez-Moreno 2003), in common with some ECM fungi. Although one species of Ericaceae (Gaultheria mucronata) was well-represented in understories at Senderos, more individuals of this species were recorded beneath AM Cupressaceae stands than beneath ECM Nothofagus (Table S2). Plantation understorey composition at Senderos thus did not appear to portend development of alternative stable states beneath AM vs. ECM plantations.
The limited evidence of an organic N economy at Senderos del Bosque might reflect the relatively N-rich character of the soil there. Modelling by Orwin et al. (2011) predicted nutrient immobilisation due to ECM mining of nutrients from soil organic matter on sites with low fertility, but not on sites with higher initial nutrient levels – a prediction upheld by subsequent empirical work (Mayer et al. 2023). Under this scenario, we would expect that Nothofagus will be transient on N-rich soils, undergoing successional replacement by more shade-tolerant trees. There is some evidence that ECM fungal lineages with low peroxidase activity (e.g., Cenococcum) impose lower carbon costs on their host plants than lineages that are more capable of accessing organic N sources using peroxidases (e.g. Cortinarius, Ramaria) (Hupperts and Lilleskov 2022). Host plants may therefore allocate carbon preferentially to fungi that mine N from soil organic matter only when N is in short supply. Although the elevated levels of Mn beneath Nothofagus stands at Senderos del Bosque are consistent with the presence of ECM fungi capable of enzymatic liberation of N from soil organic matter, lineages well-endowed with this capacity may not be the dominant component of local ECM communities.
A second possibility is that organic nutrient use by ectomycorrhizal fungi requires a more extended period to effect significant N immobilization, and hence influence stand dynamics. Trenching experiments suggest that switches from organic to mineral N economies can occur within 12 months in some situations (Gadgil and Gadgil 1971, 1975), but less is known about switches in the opposite direction. Comparisons of small, ~ 90 year-old forestry plots in the Morton Arboretum in Illinois found that topsoil C:N ratios were significantly higher beneath ECM trees than beneath AM species (Midgley and Sims 2020); however, ammonium levels and N mineralization were higher beneath ECM trees, counter to what would be expected if ECM fungi were competing for N with saprophytes. It may be relevant that the soils studied by Midgley and Sims (2020) had relatively high total N (0.44–0.54%) and fairly low C:N ratios (12.9–14.9). Like our study site at Senderos del Bosque, the Morton Herbarium might thus represent another situation where development of a strong organic N economy is unlikely (Orwin et al. 2011).
A third possibility is that leaf trait differences between the target species masked the impact of ECM fungi on C and N cycling. We did not measure leaf nutrient concentrations at our study sites, but sympatric comparisons elsewhere in Chile show higher leaf N in Nothofagus species than in Austrocedrus or Fitzroya (Lusk and Matus 2000; Lusk et al. 2003). Any N-depletion of organic matter by ECM fungi may therefore have been offset by higher initial litter N concentrations in the Nothofagus plantations, resulting in similar overall decomposition and mineralization rates in the two stand types. This third possibility seems the most promising explanation, as it reconciles the evidence of organic matter degradation by ECM fungi (elevated Mn availability beneath Nothofagus plantations) with the similarity of soil total C and N levels beneath the two stand types (Table 2).
The lower pH beneath Nothofagus stands (Table 2) may reflect a general tendency of ectomycorrhizal associations to acidify soils. Comparative studies of plantations have reported that pH is generally lower beneath ectomycorrhizal Pinaceae and Fagaceae than under neighbouring stands of arbuscular mycorrhizal Acer, Fraxinus and Thuja (Binkley and Valentine 1991; Prescott et al. 2000; Augusto et al. 2002), although France et al. (1989) reported the opposite pattern on a site subject to acid deposition from upwind industries. The generality of the effect of mycorrhizal associations on soil pH was confirmed by a comparative study of 30 tree species in an arboretum, reporting averages of 6.3 vs. 5.6 beneath AM and ECM species, respectively (Midgley and Sims 2020).
In conclusion, we found little evidence that the establishment of AM and ECM stands at Senderos del Bosque will give rise to alternative stable states. Although elevated Mn levels beneath Nothofagus plantations were consistent with organic matter degradation by ECM fungi (Fig. 3), the similarity of N availability and organic C beneath Nothofagus and Cupressaceae plantations may indicate that any impact on decomposition and mineralization rates was offset by countervailing leaf trait differences between the dominant tree species. The understories of the ~ 30-yr old Nothofagus plantations at Senderos del Bosque are populated largely by more shade-tolerant AM and non-mycorrhizal tree species (Table 1) that would eventually replace the Nothofagus overstorey if it were left unharvested – as is seen in the normal course of successional development in lowland south-central Chile (Veblen et al. 1996). An organic N economy is likely to have greater influence at higher elevations and higher latitudes in Chile, where ericaceous shrubs often grow in the understories of Nothofagus pumilio stands, on soils with higher C:N ratios (Cuevas 2000; Romanyà et al. 2005; Fuentes-Ramirez et al. 2020).
References
Augusto L, Ranger J, Binkley D, Rothe A (2002) Impact of several common tree species of European temperate forests on soil fertility. Ann Sci 59:233–253
Averill C, Turner BL, Finzi AC (2014) Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nature 505:543. https://doi.org/10.1038/nature12901
Binkley D, Valentine D (1991) Fifty-year biogeochemical effects of green ash, white pine, and Norway spruce in a replicated experiment. For Ecol Manag 40:13–25. https://doi.org/10.1016/0378-1127(91)90088-D
Blakemore L, Searle P, Daly B (1987) Methods for chemical analysis of soils. New Zealand Soil Bureau Scientific Report 10A. Soil Bureau, Department of Scientific and Industrial Research, Lower Hutt, New Zealand
Boerner REJ, Koslowsky SD (1989) Microsite variations in soil chemistry and nitrogen mineralization in a beech-maple forest. Soil Biol Biochem 21:795–801. https://doi.org/10.1016/0038-0717(89)90173-9
Chen DM, Andrew FST, Burke RM, John WGC (2001) Identification of genes for Lignin peroxidases and Manganese peroxidases in Ectomycorrhizal Fungi. New Phytol 152:151–158
CIREN (1999) Estudio agrológico De La Provincia De Valdivia X-Región. Descripción De Suelos. Material y Símbolos. Centro de Información de Recursos Naturales, Santiago
Corrales A, Henkel TW, Smith ME (2018) Ectomycorrhizal associations in the tropics – biogeography, diversity patterns and ecosystem roles. New Phytol 220:1076–1091. https://doi.org/10.1111/nph.15151
Corrales A, Mangan SA, Turner BL, Dalling JW (2016) An ectomycorrhizal nitrogen economy facilitates monodominance in a neotropical forest. Ecol Lett 19:383–392. https://doi.org/10.1111/ele.12570
Cuevas JG (2000) Tree recruitment at the Nothofagus Pumilio alpine timberline in Tierra Del Fuego. Chile J Ecol 88:840–855. https://doi.org/10.1046/j.1365-2745.2000.00497.x
Dirección Meteorológica de Chile (2001) Estadística Climatología, Tomo II. General de Aeronáutica Civil, Santiago, Chile
Donoso PJ, González ME, Escobar B, Basso I, Otero L (1998) Viverización Y plantación De Raulí, Roble Y Coihue en Chile. In: Donoso C, Lara A (eds) Silvicultura De Los Bosques Nativos De Chile. Editorial Universitaria Santiago
Fernandez CW, Kennedy PG (2016) Revisiting the ‘Gadgil effect’: do interguild fungal interactions control carbon cycling in forest soils? New Phytol 209:1382–1394. https://doi.org/10.1111/nph.13648
France EA, Binkley D, Valentine D (1989) Soil chemistry changes after 27 years under four tree species in southern Ontario. Can J for Res 19:1648–1650. https://doi.org/10.1139/x89-251
Fuentes-Ramirez A, Salas-Eljatib C, González ME, Urrutia-Estrada J, Arroyo-Vargas P, Santibañez P (2020) Initial response of understorey vegetation and tree regeneration to a mixed-severity fire in old-growth Araucaria–Nothofagus forests. Appl Veg Sci 23:210–222. https://doi.org/10.1111/avsc.12479
Gadgil RL, Gadgil P (1971) Mycorrhiza and litter decomposition. Nature 233:133
Gadgil PD, Gadgil RL (1975) Suppression of litter decomposition by mycorrhizal roots of Pinus radiata. New Z J Forestry Service 5:33–41
Godoy R, Marín C (2019) Mycorrhizal studies in temperate rainforests of southern Chile. In: ML Pagano M (ed) Mycorrhizal Fungi in South America. Springer Nature, Basingstoke
Hall JS, Harris DJ, Saltonstall K, Medjibe VP, Ashton MS, Turner BL (2020) Resource acquisition strategies facilitate Gilbertiodendron dewevrei monodominance in African lowland forests. J Ecol 108:433–448
Hofrichter M (2002) Review: lignin conversion by manganese peroxidase (MnP). Enzym Microb Technol 30:454–466. https://doi.org/10.1016/S0141-0229(01)00528-2
Hupperts SF, Lilleskov EA (2022) Predictors of taxonomic and functional composition of black spruce seedling ectomycorrhizal fungal communities along peatland drainage gradients. Mycorrhiza 32:67–81. https://doi.org/10.1007/s00572-021-01060-3
Kiilsgaard CW, Greene SE, Stafford SG (1987) Nutrient concentrations in litterfall from some western conifers with special reference to calcium. Plant Soil 102:223–227. https://doi.org/10.1007/BF02370707
Lin G, Guo D, Li L, Ma C, Zeng D-H (2018) Contrasting effects of ectomycorrhizal and arbuscular mycorrhizal tropical tree species on soil nitrogen cycling: the potential mechanisms and corresponding adaptive strategies. Oikos 127:518–530. https://doi.org/10.1111/oik.04751
Lin G, McCormack ML, Ma C, Guo D (2017) Similar below-ground carbon cycling dynamics but contrasting modes of nitrogen cycling between arbuscular mycorrhizal and ectomycorrhizal forests. New Phytol 213:1440–1451. https://doi.org/10.1111/nph.14206
Lindahl BD, Tunlid A (2015) Ectomycorrhizal fungi–potential organic matter decomposers, yet not saprotrophs. New Phytol 205:1443–1447
Lu M, Hedin LO (2019) Global plant–symbiont organization and emergence of biogeochemical cycles resolved by evolution-based trait modelling. Nat Ecol Evol 3:239–250. https://doi.org/10.1038/s41559-018-0759-0
Lusk CH, Atatoa Carr M (2023) Canopy structure and understorey light availability in Nothofagus and podocarp-broadleaf stands in a New Zealand forest. N Z J Bot. https://doi.org/10.1080/0028825X.2023.2240752
Lusk CH, Matus F (2000) Juvenile tree growth rates and species sorting on fine-scale soil fertility gradients in a Chilean temperate rain forest. J Biogeogr 27:1011–1020
Lusk CH, Wright I, Reich PB (2003) Photosynthetic differences contribute to competitive advantage of evergreen angiosperm trees over evergreen conifers in productive habitats. New Phytol 160:329–336
Mayer M, Matthews B, Sandén H, Katzensteiner K, Hagedorn F, Gorfer M, Berger H, Berger TW, Godbold DL, Rewald B (2023) Soil fertility determines whether ectomycorrhizal fungi accelerate or decelerate decomposition in a temperate forest. New Phytol. https://doi.org/10.1111/nph.18930. ((in press))
Midgley MG, Sims RS (2020) Mycorrhizal association better predicts tree effects on soil than leaf habit. Front Forests Global Change 3:74
Nadelhoffer KJ, Aber JD, Melillo JM (1983) Leaf-litter production and soil organic matter dynamics along a nitrogen-availability gradient in Southern Wisconsin (U.S.A). Can J for Res 13:12–21. https://doi.org/10.1139/x83-003
Niiyama K, Rahman KA, Iida S, Kimura K, Azizi R, Appanah S (1999) Spatial patterns of common tree species relating to topography, canopy gaps and understorey vegetation in a hill dipterocarp forest at Semangkok Forest Reserve, Peninsular Malaysia. J Trop for Sci 731–745
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara R, Simpson GL, Solymos P, Stevens MHH, Wagner H (2019) Package ‘vegan’. Community Ecology Package. 2.5-4 edn. https://cran.r-project.org, https://github.com/vegandevs/vegan
Orwin KH, Kirschbaum MUF, St John MG, Dickie IA (2011) Organic nutrient uptake by mycorrhizal fungi enhances ecosystem carbon storage: a model-based assessment. Ecol Lett 14:493–502. https://doi.org/10.1111/j.1461-0248.2011.01611.x
Pellitier PT, Zak DR (2018) Ectomycorrhizal fungi and the enzymatic liberation of nitrogen from soil organic matter: why evolutionary history matters. New Phytol 217:68–73. https://doi.org/10.1111/nph.14598
Pellitier PT, Zak DR (2021) Ectomycorrhizal fungal decay traits along a soil nitrogen gradient. New Phytol 232:2152–2164. https://doi.org/10.1111/nph.17734
Prescott C, Vesterdal L, Pratt J, Venner K, Montigny Ld, Trofymow J (2000) Nutrient concentrations and nitrogen mineralization in forest floors of single species conifer plantations in coastal british Columbia. Can J for Res 30:1341–1352
R Core Team (2022) R: A Language and Environment for Statistical Computing, Vienna, Austria., Available at: https://www.R-project.org/
Read DJ (1991) Mycorrhizas in ecosystems. Experientia 47:376–391. https://doi.org/10.1007/BF01972080
Read DJ, Perez-Moreno J (2003) Mycorrhizas and nutrient cycling in ecosystems – a journey towards relevance? New Phytol 157:475–492. https://doi.org/10.1046/j.1469-8137.2003.00704.x
Rodriguez R, Marticorena C, Alarcón D, Baeza C, Cavieres L, Finot VL, Fuentes N, Kiessling A, Mihoc M, Pauchard A (2018) Catálogo De las plantas vasculares de Chile. Gayana Botánica 75:1–430
Romanyà J, Fons J, Sauras-Yera T, Gutiérrez E, Vallejo VR (2005) Soil–plant relationships and tree distribution in old growth Nothofagus betuloides and Nothofagus pumilio forests of Tierra Del Fuego. Geoderma 124:169–180. https://doi.org/10.1016/j.geoderma.2004.04.011
Romero-Olivares AL, Morrison EW, Pringle A, Frey SD (2021) Linking genes to traits in fungi. Microb Ecol 82:145–155. https://doi.org/10.1007/s00248-021-01687-x
Rowlingson A, June SR, Lusk CH (2022) Dynamics of southern beech (Nothofagaceae) stands in the lowland North Island of New Zealand. N Z J Bot 60:227–254. https://doi.org/10.1080/0028825X.2021.1998143
Steidinger BS, Crowther TW, Liang J, Van Nuland ME, Werner GDA, Reich PB, Nabuurs G, de Miguel S, Zhou M, Picard N, Herault B, Zhao X, Zhang C, Routh D, Peay KG, Abegg M, Adou Yao CY, Alberti G, Almeyda Zambrano A, Alvarez-Davila E, Alvarez-Loayza P, Alves LF, Ammer C, Antón-Fernández C, Araujo-Murakami A, Arroyo L, Avitabile V, Aymard G, Baker T, Bałazy R, Banki O, Barroso J, Bastian M, Bastin J-F, Birigazzi L, Birnbaum P, Bitariho R, Boeckx P, Bongers F, Bouriaud O, Brancalion Pedro HS, Brandl S, Brearley FQ, Brienen R, Broadbent E, Bruelheide H, Bussotti F, Cazzolla Gatti R, Cesar R, Cesljar G, Chazdon R, Chen HYH, Chisholm C, Cienciala E, Clark CJ, Clark D, Colletta G, Condit R, Coomes D, Cornejo Valverde F, Corral-Rivas JJ, Crim P, Cumming J, Dayanandan S, de Gasper AL, Decuyper M, Derroire G, DeVries B, Djordjevic I, Iêda A, Dourdain A, Obiang NLE, Enquist B, Eyre T, Fandohan AB, Fayle TM, Feldpausch TR, Finér L, Fischer M, Fletcher C, Fridman J, Frizzera L, Gamarra JGP, Gianelle D, Glick HB, Harris D, Hector A, Hemp A, Hengeveld G, Herbohn J, Herold M, Hillers A, Honorio Coronado EN, Huber M, Hui C, Cho H, Ibanez T, Jung I, Imai N, Jagodzinski AM, Jaroszewicz B, Johannsen V, Joly CA, Jucker T, Karminov V, Kartawinata K, Kearsley E, Kenfack D, Kennard D, Kepfer-Rojas S, Keppel G, Khan ML, Killeen T, Kim HS, Kitayama K, Köhl M, Korjus H, Kraxner F, Laarmann D, Lang M, Lewis S, Lu H, Lukina N, Maitner B, Malhi Y, Marcon E, Marimon Beatriz S, Marimon-Junior BH, Marshall AR, Martin E, Martynenko O, Meave JA, Melo-Cruz O, Mendoza C, Merow C, Monteagudo Mendoza A, Moreno V, Mukul SA, Mundhenk P, Nava-Miranda MG, Neill D, Neldner V, Nevenic R, Ngugi M, Niklaus P, Oleksyn J, Ontikov P, Ortiz-Malavasi E, Pan Y, Paquette A, Parada-Gutierrez A, Parfenova E, Park M, Parren M, Parthasarathy N, Peri PL, Pfautsch S, Phillips O, Piedade MT, Piotto D, Pitman NCA, Polo I, Poorter L, Poulsen AD, Poulsen JR, Pretzsch H, Ramirez Arevalo F, Restrepo-Correa Z, Rodeghiero M, Rolim S, Roopsind A, Rovero F, Rutishauser E, Saikia P, Saner P, Schall P, Schelhaas M-J, Schepaschenko D, Scherer-Lorenzen M, Schmid B, Schöngart J, Searle E, Seben V, Serra-Diaz JM, Salas-Eljatib C, Sheil D, Shvidenko A, Silva-Espejo J, Silveira M, Singh J, Sist P, Slik F, Sonké B, Souza AF, Stereńczak K, Svenning J-C, Svoboda M, Targhetta N, Tchebakova N, Ht Steege, Thomas R, Tikhonova E, Umunay P, Usoltsev V, Valladares F, van der Plas F, Van Do T, Vasquez Martinez R, Verbeeck H, Viana H, Vieira S, von Gadow K, Wang H-F, Watson J, Westerlund B, Wiser S, Wittmann F, Wortel V, Zagt R, Zawila-Niedzwiecki T, Zhu Z-X, Zo-Bi IC, Consortium G (2019) Climatic controls of decomposition drive the global biogeography of forest-tree symbioses. Nature 569:404–408. https://doi.org/10.1038/s41586-019-1128-0
Sterkenburg E, Clemmensen KE, Ekblad A, Finlay RD, Lindahl BD (2018) Contrasting effects of ectomycorrhizal fungi on early and late stage decomposition in a boreal forest. ISME J 12:2187–2197. https://doi.org/10.1038/s41396-018-0181-2
Tsai J, David MB, Darmody RG (2014) Twenty-three-year changes in upland and bottomland forest soils of central Illinois. Soil Sci 179:95–102. https://doi.org/10.1097/ss.0000000000000043
Veblen TT, Donoso C, Kitzberger T, Rebertus AJ (1996) Ecology of southern Chilean and Argentinean Nothofagus forests. In: Veblen T, Hill R, Read J (eds) The Ecology and Biogeography of Nothofagus forests. Yale University Press, New Haven
Wardle JA (1984) The New Zealand beeches: Ecology, Utilisation and Management. New Zealand Forest Service, Christchurch
Wardle P (1991) Vegetation of New Zealand. Cambridge University Press, Cambridge
Acknowledgements
We thank Nicolás Pacheco and Tomás Riquelme-Buitano for field assistance.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions This research was funded by the Royal Society of New Zealand, through Marsden grant 20-UOW-041.
Author information
Authors and Affiliations
Contributions
The study was conceived by CHL and IAD. The study site was selected by PJD and CHL. Fieldwork was led by RG and PJD. The data were analysed by IAD and CHL. The manuscript was written mostly by CHL, and edited and commented on by all other authors.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Ivika Ostonen.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lusk, C.H., Godoy, R., Donoso, P.J. et al. Soil nutrient availability and understorey composition beneath plantations of ecto- and arbuscular mycorrhizal Chilean native trees. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06563-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06563-4