Abstract
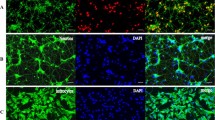
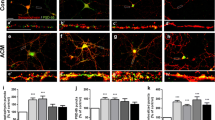
Seizure activity is linked to astrocyte activation as well as dysfunctional cortical neuron excitability produced from changes in calcium-activated potassium (KCa) channel function. Ciliary neurotrophic factor-treated astrocyte conditioned medium (CNTF-ACM) can be used to investigate the peripheral effects of activated astrocytes upon cortical neurons. However, CNTF-ACM’s effect upon KCa channel activity in cultured cortical neurons has not yet been investigated. Whole-cell patch clamp recordings were performed in rat cortical neurons to evaluate CNTF-ACM’s effects upon charybdotoxin-sensitive large-conductance KCa (BK) channel currents and apamin-sensitive small-conductance KCa (SK) channel current. Biotinylation and RT-PCR were applied to assess CNTF-ACM’s effects upon the protein and mRNA expression, respectively, of the SK channel subunits SK2 and SK3 and the BK channel subunits BKα1 and BKβ3. An anti-fibroblast growth factor-2 (FGF-2) monoclonal neutralizing antibody was used to assess the effects of the FGF-2 component of CNTF-ACM. CNTF-ACM significantly increased KCa channel current density, which was predominantly attributable to gains in BK channel activity (p < 0.05). CNTF-ACM produced a significant increase in BKα1 and BKβ3 expression (p < 0.05) but had no significant effect upon SK2 or SK3 expression (p > 0.05). Blocking FGF-2 produced significant reductions in KCa channel current density (p > 0.05) as well as BKα1 and BKβ3 expression in CNTF-ACM-treated neurons (p > 0.05). CNTF-ACM significantly enhances BK channel activity in rat cortical neurons and that FGF-2 is partially responsible for these effects. CNTF-induced astrocyte activation results in secretion of neuroactive factors which may affect neuronal excitability and resultant seizure activity in mammalian cortical neurons.





Similar content being viewed by others
References
Fisher RS, Acevedo C, Arzimanoglou A, Bogacz A, Cross JH, Elger CE, Engel J, Forsgren L, French JA, Glynn M (2014) ILAE official report: a practical clinical definition of epilepsy. Epilepsia 55:475–482
Nevalainen O, Simola M, Ansakorpi H, Raitanen J, Artama M, Isojärvi J, Auvinen A (2015) Epilepsy, excess deaths and years of life lost from external causes. Eur J Epidemiol 30:1–9
N’Gouemo P (2014) BKCa channel dysfunction in neurological diseases. Front Physiol 5:18–19
Sarafian TA, Montes C, Imura T, Qi J, Coppola G, Geschwind DH, Sofroniew MV (2010) Disruption of astrocyte STAT3 signaling decreases mitochondrial function and increases oxidative stress in vitro. PLoS One 5:e9532
Pekny M, Wilhelmsson U, Pekna M (2014) The dual role of astrocyte activation and reactive gliosis. Neurosci Lett 565:30–38
Pasquin S, Sharma M, Gauchat J-F (2015) Ciliary neurotrophic factor (CNTF): new facets of an old molecule for treating neurodegenerative and metabolic syndrome pathologies. Cytokine Growth Factor Rev 30:18–19
Albrecht PJ, Enterline JC, Cromer J, Levison SW (2007) CNTF-activated astrocytes release a soluble trophic activity for oligodendrocyte progenitors. Neurochem Res 32:263–271
Wang X, Zheng H, Liu C, Zhu C, Wang W, Li Z (2008) Ciliary neurotrophic factor-treated astrocyte conditioned medium regulates the L-type calcium channel activity in rat cortical neurons. Neurochem Res 33:826–832
Holm NR, Christophersen P, Olesen SP, Gammeltoft S (1997) Activation of calcium-dependent potassium channels in rat brain neurons by neurotrophin-3 and nerve growth factor. Proc Natl Acad Sci 94:1002–1006
Baldelli P, Forni PE, Carbone E (2000) BDNF, NT-3 and NGF induce distinct new Ca2+ channel synthesis in develo** hippocampal neurons. Eur J Neurosci 12:4017–4032
Stocker M, Krause M, Pedarzani P (1999) An apamin-sensitive Ca2+-activated K+ current in hippocampal pyramidal neurons. Proc Natl Acad Sci 96:4662–4667
Li W, Gao SB, Lv CX, Wu Y, Guo ZH, Ding JP, Xu T (2007) Characterization of voltage-and Ca2+-activated K+ channels in rat dorsal root ganglion neurons. J Cell Physiol 212:348–357
Sun X, Gu XQ, Haddad GG (2003) Calcium influx via L-and N-type calcium channels activates a transient large-conductance Ca2+-activated K+ current in mouse neocortical pyramidal neurons. J Neurosci 23:3639–3648
Chen SR, Cai YQ, Pan HL (2009) Plasticity and emerging role of BKCa channels in nociceptive control in neuropathic pain. J Neurochem 110:352–362
Joiner WJ, Khanna R, Schlichter LC, Kaczmarek LK (2001) Calmodulin regulates assembly and trafficking of SK4/IK1 Ca2+-activated K+ channels. J Biol Chem 276:37980–37985
Dominguez R, Micevych P (2010) Estradiol rapidly regulates membrane estrogen receptor α levels in hypothalamic neurons. J Neurosci 30:12589–12596
Yang J, Krishnamoorthy G, Saxena A, Zhang G, Shi J, Yang H, Delaloye K, Sept D, Cui J (2010) An epilepsy/dyskinesia-associated mutation enhances BK channel activation by potentiating Ca2+ sensing. Neuron 66:871–883
Wang B, Rothberg BS, Brenner R (2009) Mechanism of increased BK channel activation from a channel mutation that causes epilepsy. J Gen Physiol 133:283–294
Du W, Bautista JF, Yang H, Diez-Sampedro A, You S-A, Wang L, Kotagal P, Lüders HO, Shi J, Cui J (2005) Calcium-sensitive potassium channelopathy in human epilepsy and paroxysmal movement disorder. Nat Genet 37:733–738
Shruti S, Urban-Ciecko J, Fitzpatrick JA, Brenner R, Bruchez MP, Barth AL (2012) The brain-specific β4 subunit downregulates BK channel cell surface expression. PLoS One 7:e33429
Brenner R, Chen QH, Vilaythong A, Toney GM, Noebels JL, Aldrich RW (2005) BK channel β4 subunit reduces dentate gyrus excitability and protects against temporal lobe seizures. Nat Neurosci 8:1752–1759
Lorenz S, Heils A, Kasper JM, Sander T (2007) Allelic association of a truncation mutation of the KCNMB3 gene with idiopathic generalized epilepsy. Am J Med Genet Part B Neuropsychiatr Genet 144:10–13
Hu S, Labuda MZ, Pandolfo M, Goss GG, McDermid HE, Ali DW (2003) Variants of the KCNMB3 regulatory subunit of maxi BK channels affect channel inactivation. Physiol Genomics 15:191–198
Young KL, Villar D, Carson TL, Imerman PM, Moore RA, Bottoff MR (2003) Tremorgenic mycotoxin intoxication with penitrem A and roquefortine in two dogs. J Am Vet Med Assoc 222:52–53
Szebenyi G, Dent EW, Callaway JL, Seys C, Lueth H, Kalil K (2001) Fibroblast growth factor-2 promotes axon branching of cortical neurons by influencing morphology and behavior of the primary growth cone. J Neurosci 21:3932–3941
Liu Z, Holmes GL (1997) Basic fibroblast growth factor-induced seizures in rats. Neurosci Lett 233:85–88
Zucchini S, Buzzi A, Barbieri M, Rodi D, Paradiso B, Binaschi A, Coffin JD, Marzola A, Cifelli P, Belluzzi O (2008) Fgf-2 overexpression increases excitability and seizure susceptibility but decreases seizure-induced cell loss. J Neurosci 28:13112–13124
Davies PJ, Ireland DR, McLachlan EM (1996) Sources of Ca2+ for different Ca(2+)-activated K+ conductances in neurones of the rat superior cervical ganglion. J Physiol 495:353
Sah P (1995) Different calcium channels are coupled to potassium channels with distinct physiological roles in vagal neurons. Proc R Soc Lond B Biol Sci 260:105–111
Alvina K, Khodakhah K (2008) Selective regulation of spontaneous activity of neurons of the deep cerebellar nuclei by N-type calcium channels in juvenile rats. J Physiol 586:2523–2538
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 81171117), the Scientific Research Starting Foundation for the Returned Overseas Chinese Scholars (Chinese Ministry of Education, Grant No. 44), and the Science and Technology Program of Anhui Province (a Key Laboratory Project, Grant No. 1506c085014).
Author Contributions
X. J. W.: Conceived and designed the study. M. Q. S., H. L. L., H. B. X., and H. T. W: Performed the experiments. X. J. W.: Analyzed the data and drafted the manuscript
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Sun, M., Liu, H., Xu, H. et al. CNTF-Treated Astrocyte Conditioned Medium Enhances Large-Conductance Calcium-Activated Potassium Channel Activity in Rat Cortical Neurons. Neurochem Res 41, 1982–1992 (2016). https://doi.org/10.1007/s11064-016-1910-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1910-4