Abstract
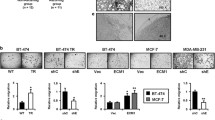
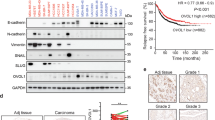
EMMPRIN (extracellular matrix metalloproteinase inducer, EMN, CD147) is a member of the immunoglobulin superfamily expressed in numerous cell types both as a soluble and a membrane-spanning glycoprotein. It is involved in many physiological processes, as well as in cancer. This study addresses mechanisms of crosstalk between EMN-driven cancer-related cellular responses and the canonical Wnt-pathway in MCF-7 breast carcinoma cells. Genetic knockdown of EMN in MCF-7 resulted in characteristic changes in cellular shape, organization of the actin cytoskeleton and malignancy profile, indicating that EMN expression represses cell motility, but, in contrast, exerts a stimulatory effect on cell proliferation and invasive properties. Increased invasiveness coincided with elevated expression of Wnt-target genes and established invasion driver matrix metalloproteinase MMP14. Activation of the downstream Wnt-pathway by means of heterologous β-catenin and/or TCF-4 expression, through inhibition of GSK-3β by LiCl treatment, or by cell stimulation with insulin-like growth factor-1 (IGF-1) resulted in increased EMN expression. EMN over-expression raised the ratio of the two opposing Wnt pathway-driven transcription factors Sp1 and Sp5, leading to stimulation of the EMN promoter. Furthermore, the EMN promoter was activated by a feed-forward circuit involving an EMN-dependent drop in expression of the repressive signal transducer and activator of transcription 1 (STAT1). Taken together, we show that the influence of EMMPRIN on malignancy-related properties of breast cancer cells is functionally connected to both Wnt- and JAK/STAT pathways.








Similar content being viewed by others
References
Muramatsu T (2016) Basigin (CD147), a multifunctional transmembrane glycoprotein with various binding partners. J Biochem 159:481–490
Hu J, Dang N, Yao H, Li Y, Zhang H, Yang X, Xu J, Bian H, **ng J, Zhu P, Chen Z (2010) Involvement of HAb18G/CD147 in T cell activation and immunological synapse formation. J Cell Mol Med 14:2132–2143
Schmidt R, Bultmann A, Fischel S, Gillitzer A, Cullen P, Walch A, Jost P, Ungerer M, Tolley ND, Lindemann S, Gawaz M, Schomig A, May AE (2008) Extracellular matrix metalloproteinase inducer (CD147) is a novel receptor on platelets, activates platelets, and augments nuclear factor kappaB-dependent inflammation in monocytes. Circ Res 102:302–309
Biswas C, Zhang Y, DeCastro R, Guo H, Nakamura T, Kataoka H, Nabeshima K (1995) The human tumor cell-derived collagenase stimulatory factor (renamed EMMPRIN) is a member of the immunoglobulin superfamily. Cancer Res 55:434–439
Fan QW, Yuasa S, Kuno N, Senda T, Kobayashi M, Muramatsu T, Kadomatsu K (1998) Expression of basigin, a member of the immunoglobulin superfamily, in the mouse central nervous system. Neurosci Res 30:53–63
Chen H, Lam Fok K, Jiang X, Chan HC (2012) New insights into germ cell migration and survival/apoptosis in spermatogenesis: lessons from CD147. Spermatogenesis 2:264–272
Szymanowska M, Hendry KA, Robinson C, Kolb AF (2009) EMMPRIN (basigin/CD147) expression is not correlated with MMP activity during adult mouse mammary gland development. J Cell Biochem 106:52–62
Iacono KT, Brown AL, Greene MI, Saouaf SJ (2007) CD147 immunoglobulin superfamily receptor function and role in pathology. Exp Mol Pathol 83:283–295
Weidle UH, Scheuer W, Eggle D, Klostermann S, Stockinger H (2010) Cancer-related issues of CD147. Cancer Genom Proteom 7:157–169
Riethdorf S, Reimers N, Assmann V, Kornfeld JW, Terracciano L, Sauter G, Pantel K (2006) High incidence of EMMPRIN expression in human tumors. Int J Cancer 119:1800–1810
Liu F, Cui L, Zhang Y, Chen L, Wang Y, Fan Y, Lei T, Gu F, Lang R, Pringle GA, Zhang X, Chen Z, Fu L (2010) Expression of HAb18G is associated with tumor progression and prognosis of breast carcinoma. Breast Cancer Res Treat 124:677–688
Zhao S, Ma W, Zhang M, Tang D, Shi Q, Xu S, Zhang X, Liu Y, Song Y, Liu L, Zhang Q (2013) High expression of CD147 and MMP-9 is correlated with poor prognosis of triple-negative breast cancer (TNBC) patients. Med Oncol 30:335
Kataoka H, DeCastro R, Zucker S, Biswas C (1993) Tumor cell-derived collagenase-stimulatory factor increases expression of interstitial collagenase, stromelysin, and 72-kDa gelatinase. Cancer Res 53:3154–3158
Sameshima T, Nabeshima K, Toole BP, Yokogami K, Okada Y, Goya T, Koono M, Wakisaka S (2000) Glioma cell extracellular matrix metalloproteinase inducer (EMMPRIN) (CD147) stimulates production of membrane-type matrix metalloproteinases and activated gelatinase A in co-cultures with brain-derived fibroblasts. Cancer Lett 157:177–184
Guo H, Zucker S, Gordon MK, Toole BP, Biswas C (1997) Stimulation of matrix metalloproteinase production by recombinant extracellular matrix metalloproteinase inducer from transfected Chinese hamster ovary cells. J Biol Chem 272:24–27
Berditchevski F, Chang S, Bodorova J, Hemler ME (1997) Generation of monoclonal antibodies to integrin-associated proteins. Evidence that alpha3beta1 complexes with EMMPRIN/basigin/OX47/M6. J Biol Chem 272:29174–29180
Dai JY, Dou KF, Wang CH, Zhao P, Lau WB, Tao L, Wu YM, Tang J, Jiang JL, Chen ZN (2009) The interaction of HAb18G/CD147 with integrin alpha6beta1 and its implications for the invasion potential of human hepatoma cells. BMC Cancer 9:337
Zhao Y, Chen S, Gou WF, Niu ZF, Zhao S, **ao LJ, Takano Y, Zheng HC (2013) The role of EMMPRIN expression in ovarian epithelial carcinomas. Cell Cycle 12:2899–29913
Wang B, Xu YF, He BS, Pan YQ, Zhang LR, Zhu C, Qu LL, Wang SK (2010) RNAi-mediated silencing of CD147 inhibits tumor cell proliferation, invasion and increases chemosensitivity to cisplatin in SGC7901 cells in vitro. J Exp Clin Cancer Res 29:61
Knutti N, Kuepper M, Friedrich K (2015) Soluble extracellular matrix metalloproteinase inducer (EMMPRIN, EMN) regulates cancer-related cellular functions by homotypic interactions with surface CD147. FEBS J 282:4187–4200
Menashi S, Serova M, Ma L, Vignot S, Mourah S, Calvo F (2003) Regulation of extracellular matrix metalloproteinase inducer and matrix metalloproteinase expression by amphiregulin in transformed human breast epithelial cells. Cancer Res 63:7575–7580
Chen Y, Gou X, Ke X, Cui H, Chen Z (2012) Human tumor cells induce angiogenesis through positive feedback between CD147 and insulin-like growth factor-I. PLoS ONE 7:e40965
Wu J, Ru NY, Zhang Y, Li Y, Wei D, Ren Z, Huang XF, Chen ZN, Bian H (2011) HAb18G/CD147 promotes epithelial–mesenchymal transition through TGF-beta signaling and is transcriptionally regulated by Slug. Oncogene 30:4410–4427
Ke X, Fei F, Chen Y, Xu L, Zhang Z, Huang Q, Zhang H, Yang H, Chen Z, **ng J (2012) Hypoxia upregulates CD147 through a combined effect of HIF-1alpha and Sp1 to promote glycolysis and tumor progression in epithelial solid tumors. Carcinogenesis 33:1598–1607
Sidhu SS, Nawroth R, Retz M, Lemjabbar-Alaoui H, Dasari V, Basbaum C (2010) EMMPRIN regulates the canonical Wnt/beta-catenin signaling pathway, a potential role in accelerating lung tumorigenesis. Oncogene 29:4145–4156
Howe LR, Brown AM (2004) Wnt signaling and breast cancer. Cancer Biol Ther 3:36–41
Tetsu O, McCormick F (1999) Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 398:422–426
Huang Q, Li J, **ng J, Li W, Li H, Ke X, Zhang J, Ren T, Shang Y, Yang H, Jiang J, Chen Z (2014) CD147 promotes reprogramming of glucose metabolism and cell proliferation in HCC cells by inhibiting the p53-dependent signaling pathway. J Hepatol 61:859–866
Takahashi M, Nakamura Y, Obama K, Furukawa Y (2005) Identification of SP5 as a downstream gene of the beta-catenin/Tcf pathway and its enhanced expression in human colon cancer. Int J Oncol 27:1483–1487
Fujimura N, Vacik T, Machon O, Vlcek C, Scalabrin S, Speth M, Diep D, Krauss S, Kozmik Z (2007) Wnt-mediated down-regulation of Sp1 target genes by a transcriptional repressor Sp5. J Biol Chem 282:1225–1237
Aberle H, Bauer A, Stappert J, Kispert A. Kemler R (1997) beta-catenin is a target for the ubiquitin–proteasome pathway. EMBO J 16:3797–3804
Lacroix M, Leclercq G (2004) Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83:249–289
Till A, Rosenstiel P, Brautigam K, Sina C, Jacobs G, Oberg HH, Seegert D, Chakraborty T, Schreiber S (2008) A role for membrane-bound CD147 in NOD2-mediated recognition of bacterial cytoinvasion. J Cell Sci 121:487–495
Weiske J, Albring KF, Huber O (2007) The tumor suppressor Fhit acts as a repressor of beta-catenin transcriptional activity. Proc Natl Acad Sci USA 104:20344–20349
Kramer OH, Baus D, Knauer SK, Stein S, Jager E, Stauber RH, Grez M, Pfitzner E, Heinzel T (2006) Acetylation of Stat1 modulates NF-kappaB activity. Genes Dev 20:473–485
Kong LM, Liao CG, Fei F, Guo X, **ng JL, Chen ZN (2010) Transcription factor Sp1 regulates expression of cancer-associated molecule CD147 in human lung cancer. Cancer Sci 101:1463–1470
Niyazi M, Niyazi I, Belka C (2007) Counting colonies of clonogenic assays by using densitometric software. Radiat Oncol 2:4
Messeguer X, Escudero R, Farre D, Nunez O, Martinez J, Alba MM (2002) PROMO: detection of known transcription regulatory elements using species-tailored searches. Bioinformatics 18:333–334
Sato T, Watanabe M, Hashimoto K, Ota T, Akimoto N, Imada K, Nomizu M, Ito A (2012) A novel functional site of extracellular matrix metalloproteinase inducer (EMMPRIN) that limits the migration of human uterine cervical carcinoma cells. Int J Oncol 40:236–242
Li Y, Wu J, Song F, Tang J, Wang SJ, Yu XL, Chen ZN, Jiang JL (2012) Extracellular membrane-proximal domain of HAb18G/CD147 binds to metal ion-dependent adhesion site (MIDAS) motif of integrin beta1 to modulate malignant properties of hepatoma cells. J Biol Chem 287:4759–4772
Desgrosellier JS, Cheresh DA (2010) Integrins in cancer: biological implications and therapeutic opportunities. Nat Rev Cancer 10:9–22
Ratnikov BI, Rozanov DV, Postnova TI, Baciu PG, Zhang H, DiScipio G, Chestukhina GG, Smith JW, Deryugina EI, Strongin AY (2002) An alternative processing of integrin alpha(v) subunit in tumor cells by membrane type-1 matrix metalloproteinase. J Biol Chem 277:7377–7385
Guvakova MA, Surmacz E (1997) Overexpressed IGF-I receptors reduce estrogen growth requirements, enhance survival, and promote E-cadherin-mediated cell-cell adhesion in human breast cancer cells. Exp Cell Res 231:149–162
Mauro L, Bartucci M, Morelli C, Ando S, Surmacz E (2001) IGF-I receptor-induced cell-cell adhesion of MCF-7 breast cancer cells requires the expression of junction protein ZO-1. J Biol Chem 276:39892–39897
Tapia R, Huerta M, Islas S, Avila-Flores A, Lopez-Bayghen E, Weiske J, Huber O, Gonzalez-Mariscal L (2009) Zona occludens-2 inhibits cyclin D1 expression and cell proliferation and exhibits changes in localization along the cell cycle. Mol Biol Cell 20:1102–1117
Gonzalez-Mariscal L, Bautista P, Lechuga S, Quiros M (2012) ZO-2, a tight junction scaffold protein involved in the regulation of cell proliferation and apoptosis. Ann N Y Acad Sci 1257:133–141
Friedl P, Gilmour D (2009) Collective cell migration in morphogenesis, regeneration and cancer. Nat Rev Mol Cell Biol 10:445–457
Schneckenleithner C, Bago-Horvath Z, Dolznig H, Neugebauer N, Kollmann K, Kolbe T, Decker T, Kerjaschki D, Wagner KU, Muller M, Stoiber D, Sexl V (2011) Putting the brakes on mammary tumorigenesis: loss of STAT1 predisposes to intraepithelial neoplasias. Oncotarget 2:1043–1054
Bailey SG, Cragg MS, Townsend PA (2012) Role of STAT1 in the breast. JAKSTAT 1:197–199
Acknowledgements
This work was supported by the Thüringer Aufbaubank (European Regional Development Fund Grants FE 9034 and FE 9053) and the Interdisziplinäres Zentrum für klinische Forschung (IZKF), Jena. We are grateful to Andreas Till for EMN cDNA and the EMN-knockdown constructs, to Oliver Krämer for the STAT1 expression construct and to Sonnhild Mittag for valuable discussions.
Author information
Authors and Affiliations
Contributions
NK performed and documented all experiments, OH designed, supervised and interpreted experiments concerned with Wnt pathway analysis, KF designed and supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Knutti, N., Huber, O. & Friedrich, K. CD147 (EMMPRIN) controls malignant properties of breast cancer cells by interdependent signaling of Wnt and JAK/STAT pathways. Mol Cell Biochem 451, 197–209 (2019). https://doi.org/10.1007/s11010-018-3406-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3406-9