Abstract
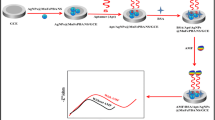
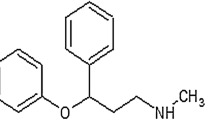
The present work describes a dual-readout assay for the determination of an antipsychotic drug olanzapine using Rhodamine B modified silver nanoparticles (AgNPs). AgNPs, when mixed with Rhodamine B, quenched its fluorescence emission with high quenching efficiency as evident from the Stern Volmer plot. Transmission electron microscopy image and Dynamic Light Scattering histogram of Rhodamine B bound AgNPs showed a stable monodispersed nanosuspension. Addition of olanzapine to Rhodamine B-bound AgNPs resulted in reappearance of fluorescence, which was dependent on the amount of olanzapine added to the system. Besides displacing the surface bound Rhodamine B molecules, it caused aggregation of AgNPs which formed the basis of dual-readout sensor. Several parameters such as pH, reaction time and order of addition of the three components which may influence the analytical signal were studied and optimized. The method was validated for linearity, sensitivity, selectivity, accuracy, precision and recovery. Based on this dual-readout system, linear concentration range was established from 0.05 to 10 µM (fluorescence measurement) and 5.0 to 50 µM (colorimetric response) for olanzapine. The limit of detection (LOD) using fluorescence and colorimetric approach was 0.013 µM and 1.25 µM, respectively. The proposed method showed excellent selectivity for olanzapine in presence of several antipsychotic drugs, cations, sugars and amino acids. Finally, the method was successfully applied to a pharmacokinetic study of olanzapine in rats and also for analyzing pharmaceutical formulations.











Similar content being viewed by others
Data Availability
Not applicable.
References
Global Leading Categories of Diseases/Disorders (YLDs). https://www.nimh.nih.gov/Health/statistics/global/global-leading-categories-of-diseases-disorders-ylds.shtml. Accessed 25 Jan 2019
Eli Lilly, Company, ZYPREXA (olanzapine) tablet for oral use: Prescribing Information (2018) https://www.fda.gov/media/75933/download. Accessed 25 Jan 2020
Carlson CD, Cavazzoni PA, Berg PH, Wei H, Kane JM (2003) An integrated analysis of acute treatment emergent extrapyramidal syndrome in patients with schizophrenia during olanzapine clinical trials: comparisons with placebo, haloperidol, risperidone or clozapine. J Clin Psychiatry 64:898–906. https://doi.org/10.4088/jcp.v64n0807
Carrillo JA, Herráiz AG, Ramos SI, Gervasini G, Vizcaíno S, Benítez J (2003) Role of the smoking induced cytochrome P450 (CYP)1A2 and polymorphic CYP2D6 in steady-state concentration of olanzapine. J Clin Psychopharmacol 23:119–127. https://doi.org/10.1097/00004714-200304000-00003
Gex-fabry M, Balant-gorgia AE, Balant LP (2003) Therapeutic drug monitoring of olanzapine: the combined effect of age, gender, smoking, and comedication. Ther Drug Monit 25:46–53. https://doi.org/10.1097/00007691-200302000-00007
Ghosh D, Chattopadhyay N (2015) Gold and silver nanoparticles based super quenching of fluorescence: A review. J Luminescence 160: 223–232. https://doi.org/10.1016/j.jlumin.2014.12.018
Oliveira E, Núñez C, Santos HM, Fernández-Lodeiro J, Fernández-Lodeiro A, Capelo JL, Lodeiro C (2015) Revisiting the use of gold and silver functionalised nanoparticles as colorimetric and fluorometric chemosensors for metal ions. Sen Actuators B: Chem 212:297–328. https://doi.org/10.1016/j.snb.2015.02.026
Vilela D, González MC, Escarpa A (2012) Sensing colorimetric approaches based on gold and silver nanoparticles aggregation: Chemical creativity behind the assay- A review. Anal Chim Acta 751:24–43. https://doi.org/10.1016/j.aca.2012.08.043
Emrani AS, Danesh NM, Lavaee P, Jalalian SH, Ramezani M, Abnous K, Taghdisi SM (2015) Sensitive and selective detection of digoxin based on fluorescence quenching and colorimetric aptasensors. Anal Methods 7:3419–3424. https://doi.org/10.1039/C5AY00235D
Emrani AS, Danesh NM, Lavaee P, Ramezani M, Abnous K, Taghdisi SM (2016) Colorimetric and fluorescence quenching aptasensors for detection of streptomycin in blood serum and milk based on double-stranded DNA and gold nanoparticles. Food Chem 190:115–121. https://doi.org/10.1016/j.foodchem.2015.05.079
Zhu X, **ao Y, Jiang X, Li J, Qin H, Huang H, Zhang Y, He X, Wang K (2016) A ratiometric nanosensor based on conjugated polyelectrolyte-stabilized AgNPs for ultrasensitive fluorescent and colorimetric sensing of melamine. Talanta 151:68–74. https://doi.org/10.1016/j.talanta.2016.01.012
Cheng C, Chen HY, Wu CS, Meena JS, Simon T, Ko FH (2016) A highly sensitive and selective cyanide detection using a gold nanoparticle-based dual fluorescence–colorimetric sensor with a wide concentration range. Sens Actuators B Chem 227:283–290. https://doi.org/10.1016/j.snb.2015.12.057
Firdous S, Aman T, Nisa AU (2005) Determination of olanzapine by UV spectrophotometry and non-aqueous titration. J Chem Soc Pak 27:163–167. https://www.researchgate.net/publication/283874978
Raggi MA, Casamenti G, Mandrioli R, Izzo G, Kenndler E (2000) Quantitation of olanzapine in tablets by HPLC, CZE, derivative spectrometry and linear voltammetry. J Pharm Biomed Anal 23:973–981. https://doi.org/10.1016/S0731-7085(00)00382-4
Biryol I, Erk N (2003) Voltammetric, spectrophotometric, and high performance liquid chromatographic analysis of olanzapine. Anal Lett 36: 2497–2513. https://doi.org/10.1081/AL-120024338
Tantawy MA, Hassan NY, Elragehy NA, Abdelkawy M (2013) Simultaneous determination of olanzapine and fluoxetine hydrochloride in capsules by spectrophotometry, TLC-spectrodensitometry and HPLC. J Adv Res 4: 173–180. https://doi.org/10.1016/j.jare.2012.05.004
Revanasiddappa HD, Deepakumari HN (2014) Highly sensitive spectrophotometric method for the quantitative determination of olanzapine in its pure and in pharmaceutical dosage forms. J Sci Ind Res 73:41–45. http://nopr.niscair.res.in/bitstream/123456789/25429/1/JSIR%2073%281%29%2041-45.pdf
Basavaiah K, Abdulrahman SAM (2012) Utility of N-bromosuccinimide as an environmental-friendly reagent for sensitive determination of olanzapine in pharmaceuticals. Jordan J Pharm Sci 4:209–221. https://journals.ju.edu.jo/JJPS/article/view/2570/2336
Upadhyay K, Asthana A, Tiwari N (2013) Sensitive and selective methods for determination of antipsychotic drug olanzapine in pharmaceuticals. Res Chem Intermed 39:2629–2640. https://doi.org/10.1007/s11164-012-0786-4
Rajendraprasad N, Basavaiah K (2010) Highly sensitive spectrophotometric determination of olanzapine using cerium(IV) and iron(II) complexes of 1,10-phenanthroline and 2,2'-bipyridyl. J Anal Chem 65: 482–488. https://doi.org/10.1134/S1061934810050084
Krebs A, Starczewska B, Puzanowska-Tarasiewicz H, Sledz J (2006) Spectrophotometric determination of olanzapine by its oxidation with N-bromosuccinimide and cerium(IV)sulphate. Anal Sci 22:829–833. https://doi.org/10.2116/analsci.22.829
Rajendraprasad N, Basavaiah K (2009) Determination of olanzapine by spectrophotometry using permanganate. Braz J Pharm Sci 45:539–550. https://doi.org/10.1590/S1984-82502009000300020
Basavaiah K, Abdulrahman SAM, Basavaiah KV (2009) Simple and sensitive spectrophotometric determination of olanzapine in pharmaceutical formulations using two sulphonphthalein acid dyes. J Food Drug Anal 17:434–442. https://www.researchgate.net/ publication/228679479
Belal F, El-Brashy A, El-Enany N, El-Bahay N (2008) Spectrofluorometric determination of olanzapine and fluphenazine hydrochloride in pharmaceutical preparations and human plasma using eosin: Application to stability studies. J AOAC Int 91: 1309–1317. https://doi.org/10.1093/jaoac/91.6.1309
Zhao F, Fan Q, Cai H (2014) Flow-injection chemiluminescence determination of olanzapine using N-chlorosuccinimide–calcein reaction sensitized by zinc (II). Luminescence 29:219–224. https://doi.org/10.1002/bio.2531
Ahmed HM, Mohamed MA, Salem WM (2015) New voltammetric analysis of olanzapine in tablets and human urine samples using a modified carbon paste sensor electrode incorporating gold nanoparticles and glutamine in a micellar medium. Anal Methods 7:581–598. https://doi.org/10.1039/C4AY02450H
Albayrak M, Kadioglu Y, Yaman ME, Senol O, Oral E (2019) Determination of olanzapine for therapeutic drug monitoring in schizophrenia patients by LC/MS method. Biomed Chromatogr 33:e4468. https://doi.org/10.1002/bmc.4468
Du P, Li P, Zhao R, Liu H, Liu L (2019) Optimized UPLC-MS/MS method for the quantitation of olanzapine in human plasma: application to a bioequivalence study. Bioanalysis 11:1291–1302. https://doi.org/10.4155/bio-2019-0114
Gangu Naidu C, Nageswara Rao R, Prasada Rao AV, Nagesh Kumar K, Padiya R, Madhusudhan Rao V (2018) Supported liquid extraction and LC-MS-MS determination of iloperidone and olanzapine in rat plasma: Application to a pharmacokinetic study. J Chromatogr Sci 56:879–887. https://doi.org/10.1093/chromsci/bmy063
Ruan CJ, Guo W, Zhou M, Guo GX, Wang CY, Li WB, de Leon J (2018) Quantitative determination of risperidone, paliperidone and olanzapine in human serum by liquid chromatography-tandem mass spectrometry coupled with on-line solid-phase extraction. Biomed Chromatogr 32:e4209. https://doi.org/10.1002/bmc.4209
Chavada VD, Bhatt NM, Sanyal M, Shrivastav PS (2017) Surface plasmon resonance based selective and sensitive colorimetric determination of azithromycin using unmodified silver nanoparticles in pharmaceuticals and human plasma. Spectrochim Acta Part A: Mol Biomol Spectrosc 170:97–103. https://doi.org/10.1016/j.saa.2016.07.011
Kanjanawarut R, Su XD (2009) Colorimetric detection of DNA using unmodified metallic nanoparticles and peptide nucleic acid probes. Anal Chem 81:6122–6129. https://doi.org/10.1021/ac900525k
Patel DS, Sharma N, Patel MC, Patel BN, Shrivastav PS, Sanyal M (2012) LC–MS/MS assay for olanzapine in human plasma and its application to a bioequivalence study. Acta Pharm Sin B 2:481–494. https://doi.org/10.1016/j.apsb.2012.02.009
Cui Z, Han C, Li H (2011) Dual-signal fenamithion probe by combining fluorescence with colorimetry based on Rhodamine B modified silver nanoparticles. Analyst 136:1351–1356. https://doi.org/10.1039/c0an00617c
Liu D, Chen W, Tian Y, He S, Zheng W, Sun J, Wang Z, Jiang X (2012) A highly sensitive gold-nanoparticle-based assay for acetylcholinesterase in cerebrospinal fluid of transgenic mice with Alzheimer’s disease. Adv Healthc Mat 1:90–95. https://doi.org/10.1002/adhm.201100002
ICH (2005) Validation of Analytical Procedures: Text and Methodology, Q2 (R1). International Conference on Harmonization. https://database.ich.org/sites/default/files/Q2_R1_Guideline.pdf. Accessed 25 Jan 2020
Ravinder S, Bapuji AT, Mukkanti K, Reddy DC (2013) Simultaneous determination of olanzapine and fluoxetine in human plasma by LC-MS/MS and its application to pharmacokinetic study. J Liq Chromatogr Relat Technol 36: 2651–2668. https://doi.org/10.1080/10826076.2012.723098
Bonde SL, Bhadane RP, Gaikwad A, Gavali SR, Katale DU, Narendiran AS (2014) Simultaneous determination of olanzapine and fluoxetine in human plasma by LC-MS/MS: its pharmacokinetic application. J Pharm Biomed Anal 90:64–71. https://doi.org/10.1016/j.jpba.2013.10.033
Pathak A, Rajput SJ (2009) Development of a stability-indicating HPLC method for simultaneous determination of olanzapine and fluoxetine in combined dosage forms. J Chromatogr Sci 47:605–611. https://doi.org/10.1093/chromsci/47.7.605
Ding N, Zhao H, Peng W, He Y, Zhou Y, Yuan L, Zhang Y (2012) A simple colorimetric sensor based on anti-aggregation of gold nanoparticles for Hg2+ detection. Colloids Surf A: Physicochem Eng Asp 395: 161–167. https://doi.org/10.1016/j.colsurfa.2011.12.024
Chiu JA, Franklin RB (1996) Analysis and pharmacokinetics of olanzapine (LY170053) and two metabolites in rat plasma using reversed-phase HPLC with electrochemical detection. J Pharm Biomed Anal 14:609–615. https://doi.org/10.1016/0731-7085 (95)01651-1
Aravagiri M, Teper Y, Marder SR (1999) Pharmacokinetics and tissue distribution of olanzapine in rats. Biopharm Drug Dispos 20: 369–377. https://doi.org/10.1002/1099-081X(199911)20:8<369::AID-BDD200>3.0.CO;2-6
Acknowledgements
The authors thank Department of Chemistry, Gujarat University for supporting this work and Department of Zoology, Gujarat University for pharmacokinetic study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. VDC and NMB did the formal analysis, investigation, writing - original draft and data Curation. MS designed the methodology and involved with project administration, generation of resources and software. PSS contributed towards conceptualization, supervision, writing - review & editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval/Standards
All procedures performed in studies involving animals were in accordance with the ethical standards of the institutional review board and/or national research committee, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All the experimental protocols were approved by the Committee for the Purpose of Control and Supervision of Experiment on Animals (Reg. – 167/1999/CPCSEA), New Delhi, India
Consent to Participate
Not applicable.
Consent for Publication
All authors have given their consent for publication of this work.
Code Availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chavada, V.D., Bhatt, N.M., Sanyal, M. et al. Dual Fluorescence-colorimetric Silver Nanoparticles Based Sensor for Determination of Olanzapine: Analysis in Rat Plasma and Pharmaceuticals. J Fluoresc 30, 955–967 (2020). https://doi.org/10.1007/s10895-020-02568-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-020-02568-1