Abstract
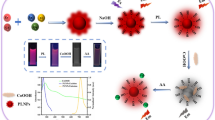
A novel, rapid, and accurate ultra-sensitive spectrofluorimetric method was developed and applied for the determination of spectinomycin hydrochloride (SPEC) in real human plasma and pharmaceutical dosage form. Silver nanoparticles (AgNPs) fluorescence was measured at 415 nm (λex at 340 nm). The method was used for SPEC quantification in the concentration range of 0.7–30 ng/mL with the limit of quantitation equal to 0.5 ng/mL and the limit of detection of 0.19 ng/mL. The fluorescence of AgNPs was quenched via a gradual increase in SPEC concentration. The developed method was validated using ICH guidelines and bio-analytically validated using US-FDA recommendations. The simplicity and high sensitivity of the proposed method make it suitable to monitor SPEC to conduct an extensive pharmacokinetic study for SPEC in human plasma. Moreover, SPEC stability was studied in real human plasma. SPEC was also subjected to acidic, basic, oxidative, and photolytic forced degradation conditions according to ICH guidelines. As observed, SPEC showed extensive degradation under acid, base, and oxidative hydrolysis stress conditions.





Similar content being viewed by others
REFERENCES
Szúnyog, J., Adams, E., Liekens, K., Roets, E., and Hoogmartens, J., J. Pharm. Biomed. Anal., 2002, vol. 29, p. 213. https://doi.org/10.1016/S0731-7085(02)00015-8
Galimand, M., Gerbaud, G., and Courvalin, P., Antimic. Agents Chemother., 2000, vol. 44, p. 1365. https://doi.org/10.1128/aac.44.5.1365-1366.2000
Stypulkowska, K., Blazewicz, A., Brudzikowska, A., Warowna–Grzeskiewicza, M., Sarna, K., and Fijaleka, Z., J. Pharm. Biomed. Anal., 2015, vol. 112, p. 8. https://doi.org/10.1016/j.jpba.2015.03.036
The United States Pharmacopoeia 30, US Pharmacopeial Convention, 2012.
Moalla, S.M.N., Hossny, N.M., Mostafa, E.R., and Amin, A.S., Int. J. Adv. Res. Sci., 2014, vol. 1, p. 21.
Wang, Y., Wang, M., Li, J., Yao, S.C., Xue, J., Zou, W.B., and Hu, C., Acta Chromatogr., 2015, vol. 27, p. 93. https://doi.org/10.1556/AChrom.27.2015.1.8
Abualhasan, M.N., Batrawi, N., Sutcliffe, O.B., and Zaid, A.N., Sci. Pharm., 2012, vol. 80, p. 977. https://doi.org/10.3797/scipharm.1207-13
Vučićević-Prčetića, K., Cservenák, R., and Radulović, J. Pharm. Biomed. Anal., 2011, vol. 56, p. 736. https://doi.org/10.1016/j.jpba.2011.07.031
Tao, Y., Chen, D., Yu, G., Yu, H., Pan, Y., Wang, Y., Huang, L., and Yuan, Z., Food Addit. Contam., Part A, 2011, vol. 28, p. 145. https://doi.org/10.1080/19440049.2010.538440
Weidong, C., Liu, J., Yang, X., and Wang, E., Electrophoresis, 2002, vol. 23, p. 3683. https://doi.org/10.1002/1522-2683(200211)23:21
Wei, W., Wei, M., Cai, Z., and Liu, S., Chromatographia, 2011, vol. 74, p. 349. https://doi.org/10.1007/s10337-011-2060-0
Hussein, S.A., Salman, B.I., Ali, M.F., and Marzouq, M.A., Luminescence, 2019, vol. 34, p. 902. https://doi.org/10.1002/bio.3688
Wang, P. and Zhang, Y., Talanta, 2015, vol. 146, p. 175. https://doi.org/10.1016/j.talanta.2015.07.065
Zaid, A.A., Bukhari, N., Haider, S., Wabaidur,S.M., and Alwarthan, A.A., Arab. J. Chem., 2010, vol. 3, p. 251. https://doi.org/10.1016/j.arabjc.2010.06.008
Caires, A., Costa, L., and Fernandes, J., Cent. Eur. J. Chem., 2013, vol. 11, p. 111. https://doi.org/10.2478/s11532-012-0139-6
Kholoud, M.M.A., Ala’a, E., Abdulrhman, A., and Reda, A.A., Arab. J. Chem., 2010, vol. 3, p. 135. https://doi.org/10.1016/j.arabjc.2010.04.008
Geddes, C.D. and Lakowicz, J.R., J. Fluoresc., 2002, vol. 12, p. 121.
Sokolov, K., George, C., and Therese, M.C., Anal. Chem., 1998, vol. 70, p. 3898. https://doi.org/10.1021/ac9712310
Yu, X., Jiang, Z., Wang, Q., and Guo, Y., Microchim. Acta, 2010, vol. 171, p. 17. https://doi.org/10.1007/s00604-010-0401-6
Zhang, R., Wang, Z., Song, C., Yang, J., Sadaf, A., and Cui, Y., J. Fluoresc., 2013, vol. 23, p. 71. https://doi.org/10.1007/s10895-012-1117-2
Vilela, D., González, M.C., and Escarpa, A., Anal. Chim. Acta, 2012, vol. 751, p. 24. https://doi.org/10.1016/j.aca.2012.08.043
Jamal, F., Jean-Sébastien, G., Penhoat, M., Payen, E., and Rolando, C., Microsyst. Technol., 2012, vol. 18, p. 151. https://doi.org/10.1007/s00542-011-1369-9
Chang, H., Tsung, T.T., Yang, Y.C., Chen, L.C., Lin, H.M., Lin, C.K., and Jwo, C.S., Int. J. Adv. Manuf. Technol., 2005, vol. 26, p. 552. https://doi.org/10.1007/s00170-003-2029-8
Anderson, C., Luciano, C., and Joelson, F., Cent. Eur. J. Chem., 2013, vol. 11, p. 111. https://doi.org/10.2478/s11532-012-0139-6
Kusumi, R., Metzler, C., and Fass, R., Chemotherapy, 1981, vol. 27, p. 95. https://doi.org/10.1159/000237962
Pesez, M.J.B., Colorimetric and Fluorimetric Analysis of Organic Compounds and Drugs, New York: Marcel Dekker, 1974.
Ali, L. and Jamshid, L.M., Luminescence, 2016, vol. 31, p. 1349. https://doi.org/10.1002/bio.3114
International Conference on Harmonization, Topic Q2 (R1) Validation of Analytical Procedures: Text and Methodology, 2005.
Joseph, R.L., Anal. Biochem., 2001, vol. 298, p. 1. https://doi.org/10.1006/abio.2001.5377
Ali, M.F., Salman, B.I., Hussein, S.A., and Marzouq, M.A., RSC Adv., 2020, vol. 10, p. 44065. https://doi.org/10.1039/D0RA07960J
Ali, H.R.H., Hassan, A.I., Hassan, Y.F., and El-Wekil, M.M., Anal. Bioanal. Chem., 2020, vol. 412, p. 1363.
Zimmer, D., Bioanalysis, 2014, vol. 6, p. 13. https://doi.org/10.4155/bio.13.298
Sweetman, S.C., Martindale: the Complete Drug Reference, London: Pharm. Press, 2009, 36th ed.
Ming-juan, W. and Chang-qin, H., Chromatographia, 2006, vol. 63, p. 260. https://doi.org/10.1365/s10337-006-0730-0
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Salman, B., Eltoukhi, W.E. & Hassan, Y.F. Applicability of Silver Nanoparticles as a Fluorescent Precursor for the Determination of Spectinomycin Hydrochloride in Human Plasma. J Anal Chem 77, 1440–1447 (2022). https://doi.org/10.1134/S1061934822110107
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934822110107