Abstract
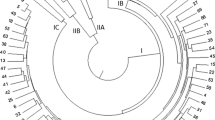
Rapeseed-mustard is one of the most important oilseed crops and providing a major source of edible oil in the world besides having other economic importance like leafy vegetables, ornamentals, and hedge crops. However, the genetic diversity present in the Brassica gene pool has not been investigated in detail. To address this problem, a study was conducted on 76 genotypes of B. juncea including cultivars, exotic lines, registered genetic stocks, advanced breeding lines, and germplasm lines. The genetic diversity was analyzed with the help of 50 polymorphic SSR and EST-SSR markers. For these genotype-marker combinations, a total of 126 alleles were amplified. All the genotypes were grouped into 5 clusters based on their dissimilarity matrix. Population structure analysis grouped the genotypes in 8 clusters and various degrees of admixture was also observed. The grou** of the genotypes appears effective as per their pedigree. The marker data was found more accurate to characterize the diversity and study the population structure than the quantitative trait data. The results of the present investigation will provide useful information for the identification of important alleles for future studies and pave the way to enhance genetic gains in Indian mustard.



Similar content being viewed by others
References
Bansal P, Kaur P, Banga SK, Banga SS (2009) Augmenting genetic diversity in Brassica juncea through its resynthesis using purposely selected diploid progenitors. Int J Plant Breed 3:41–45
Bodh PC, Yadav RK, Sharma A, Yadav M, Arora N, Dawar S, Prasad G (2020) Pocket book of agricultural statistics. In: Dir. Econ. Stat. Dep. Agric. Coop. Farmers Welfare. Minist. Agric. Farmers Welfare, Gov. India. Accessed 14 Jul 2021
Chen S, Wan Z, Nelson MN et al (2013) Evidence from genome-wide simple sequence repeat markers for a polyphyletic origin and secondary centers of genetic diversity of Brassica juncea in China and India. J Hered 104:416–427. https://doi.org/10.1093/jhered/est015
Chen R, Hara T, Ohsawa R, Yoshioka Y (2017) Analysis of genetic diversity of rapeseed genetic resources in Japan and core collection construction. Breed Sci 67:239–247. https://doi.org/10.1270/jsbbs.16192
Chen R, Shimono A, Aono M et al (2020) Genetic diversity and population structure of feral rapeseed (Brassica napus L.) in Japan. PLoS ONE 15:1–14. https://doi.org/10.1371/journal.pone.0227990
Duminil J, Di Michele M (2009) Plant species delimitation: a comparison of morphological and molecular markers. Plant Biosyst 143:528–542. https://doi.org/10.1080/11263500902722964
Ghosh KK, Haque ME, Parvin S et al (2009) Genetic diversity analysis in Brassica varieties through RAPD markers. Bangladesh J Agric Res 34:493–503
Ghosh S, Mazumder M, Mondal B et al (2019) Morphological and SSR marker-based genetic diversity analysis of Indian mustard (Brassica juncea L.) differing in Alternaria brassicicola tolerance. Euphytica 215:1–19. https://doi.org/10.1007/s10681-019-2523-1
Gupta VP, Sekhon MS, Satija DR (1991) Studies on genetic diversity, heterosis and combining ability in Indian mustard [Brassica juncea L. (Czern & Coss)]. Indian J Genet 51:448–453
Huangfu C, Song X, Qiang S (2009) ISSR variation within and among wild Brassica juncea populations: implication for herbicide resistance evolution. Genet Resour Crop Evol 56:913–924
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332
Jain A, Bhatia S, Banga SS et al (1994) Potential use of random amplified polymorphic DNA (RAPD) technique to study the genetic diversity in Indian mustard (Brassica juncea) and its relationship to heterosis. Theor Appl Genet 88:116–122
Kumar R, Gupta VP (1985) Isozyme studies in Indian mustard (Brassica juncea L.). Theor Appl Genet 70:107–110
Kumar M, Choi JY, Kumari N et al (2015) Molecular breeding in Brassica for salt tolerance: importance of microsatellite (SSR) markers for molecular breeding in Brassica. Front Plant Sci 6:1–13. https://doi.org/10.3389/fpls.2015.00688
Lodhi B, Thakral NK, Singh D et al (2016) Genetic diversity analysis in Indian mustard (Brassica juncea). J Oilseed Brassica 1:57–60
Luo Z, Brock J, Dyer JM et al (2019) Genetic diversity and population structure of a Camelina sativa spring panel. Front Plant Sci 10:1–12. https://doi.org/10.3389/fpls.2019.00184
Mishra A, Kumar P, Shamim M et al (2019) Genetic diversity and population structure analysis of Asian and African aromatic rice (Oryza sativa L.) genotypes. J Genet 98. https://doi.org/10.1007/s12041-019-1131-0
Perrier X, Jacquemoud-Collet JP (2006) DARwin software: dissimilarity analysis and representation for windows. Website http//darwinciradfr/darwin. Accessed 1 March 2013
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Saleem N, Jan SA, Atif MJ et al (2017) Multivariate based variability within diverse Indian mustard (Brassica juncea L.) genotypes. Open J Genet 7:69
Shi J, Huang S, Zhan J et al (2014) Erratum: Genome-wide microsatellite characterization and marker development in the sequenced brassica crop species (DNA Research (2014) 21 (53–68). https://doi.org/10.1093/dnares/dst040). DNA Res 21:229. https://doi.org/10.1093/dnares/dsu011
Siddiqui SA, Rai PK, Kaushal P et al (2011) Efficient modified DNA extraction method for marker identification. Natl Acad Sci Lett 34:239–245
Singh KH, Shakya R, Thakur AK et al (2013b) Genetic diversity in indian mustard [Brassica juncea (L.) Czernj & Cosson] as revealed by agronomic traits and RAPD markers. Natl Acad Sci Lett 36:419–427. https://doi.org/10.1007/s40009-013-0149-8
Singh N, Vasudev S, Kumar Yadava D et al (2013c) Assessment of genetic diversity in Brassica juncea Brassicaceae genotypes using phenotypic differences and SSR markers. Rev Biol Trop 61:1919–1934
Singh KH, Shakya R, Mahawar R (2014) Genetic diversity and patterns of variation among indian mustard (Brassica juncea (L.) Czernj. & Cosson) varieties. SABRAO J Breed Genet 46:329–339
Singh BK, Mishra DC, Yadav S et al (2016a) Identification, characterization, validation and cross-species amplification of genic-SSRs in Indian Mustard (Brassica juncea). J Plant Biochem Biotechnol 25:410–420. https://doi.org/10.1007/s13562-016-0353-y
Singh R, Semwal DP, Bhatt KC (2016b) Genetic variability and diversity pattern for agromorphological traits in Indian mustard germplasm. Indian J Plant Genet Resour 29:52–58
Singh J, Sharma PC, Sharma SK, Kumar A (2013a) Standardization of the fourier transform near-infrared reflectance spectroscopy for estimation of some oil quality parameters in mustard (Brassica spp.)
Snowdon RJ, Friedt W (2004) Molecular markers in Brassica oilseed breeding: current status and future possibilities. Plant Breed 123:1–8. https://doi.org/10.1111/j.1439-0523.2003.00968.x
Song KM, Osborn TC, Williams PH (1988) Brassica taxonomy based on nuclear restriction fragment length polymorphisms (RFLPs)-1. Genome evolution of diploid and amphidiploid species. Theor Appl Genet 75:784–794. https://doi.org/10.1007/BF00265606
Stift M, Kolář F, Meirmans PG (2019) Structure is more robust than other clustering methods in simulated mixed-ploidy populations. Heredity (edinb) 123:429–441. https://doi.org/10.1038/s41437-019-0247-6
Team RC (2021) R: a language and environment for statistical computing
Vaishnava A, Sachan JN, Tewari SK (2006) Genetic divergence for important quantitative traits in Indian mustard {Brassica juncea (l.) czern and coss}. Agric Sci Dig 26:269–272
Yadava DK, Sapra RL, Sujata V et al (2009) Selection of high diversity with a minimal set of accessions from Indian mustard (Brassica juncea) germplasm collection. Indian J Agric Sci 79:552–554
Yadava DK, Giri SC, Vignesh M et al (2011) Genetic variability and trait association studies in Indian mustard (Brassica juncea). Indian J Agric Sci 81:712
Yadava DK, Singh N, Vasudev S et al (2012) Combining ability and heterobeltiosis for yield and yield-contributing traits in Indian mustard (Brassica juncea). Indian J Agri Sci 82:563–567
Yang J, Zhang C, Zhao N et al (2018) Chinese root-type mustard provides phylogenomic insights into the evolution of the multi-use diversified allopolyploid Brassica juncea. Mol Plant 11:512–514. https://doi.org/10.1016/j.molp.2017.11.007
Zhang T, Ni XL, Jiang KF et al (2010) Relationship between heterosis and parental genetic distance based on molecular markers for functional genes related to yield traits in rice. Rice Sci 17:288–295. https://doi.org/10.1016/S1672-6308(09)60029-9
Acknowledgements
The authors gratefully acknowledge the Indian Council of Agricultural Research, New Delhi to provide funds and Germplasm section of ICAR-Directorate of Rapeseed-Mustard Research, Bharatpur for providing the germplasm.
Funding
The funding was provided by Indian Council of Agricultural research, New Delhi.
Author information
Authors and Affiliations
Contributions
PY designed the experiments and PKR supervised the study. PY, SY, AK and HSM helped in conducting the experiments. AM and RC helped in data analysis and SY helped in writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yadav, P., Yadav, S., Mishra, A. et al. Molecular distinction and population structure of Indian mustard [Brassica juncea (L.) Czern.]. Genet Resour Crop Evol 69, 1855–1866 (2022). https://doi.org/10.1007/s10722-022-01346-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-022-01346-1