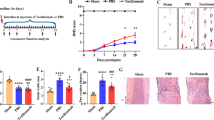
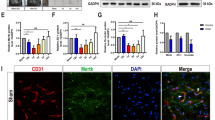
Abstract
Clearance of myelin debris caused by acute demyelination is an essential process for functional restoration following spinal cord injury (SCI). Microvascular endothelial cells, acting as “amateur” phagocytes, have been confirmed to engulf and degrade myelin debris, promoting the inflammatory response, robust angiogenesis, and persistent fibrosis. However, the effect of myelin debris engulfment on the function of endothelial tight junctions (TJs) remains unclear. Here, we demonstrate that myelin debris uptake impairs TJs and gap junctions of endothelial cells in the lesion core of the injured spinal cord and in vitro, resulting in increased permeability and leakage. We further show that myelin debris acts as an inducer to regulate the endothelial-to-mesenchymal transition in a dose-dependent manner and promotes endothelial cell migration through the PI3K/AKT and ERK signaling pathways. Together, our results indicate that myelin debris engulfment impairs TJs and promotes the migration of endothelial cells. Accelerating myelin debris clearance may help maintain blood–spinal cord barrier integrity, thus facilitating restoration of motor and sensory function following SCI.









Similar content being viewed by others
Data Availability
The data are available from the corresponding authors upon reasonable request.
References
Ahuja CS, Wilson JR, Nori S, Kotter MRN, Druschel C, Curt A, Fehlings MG (2017) Traumatic spinal cord injury. Nat Rev Dis Primers 3:17018. https://doi.org/10.1038/nrdp.2017.18
Anderson MA, Burda JE, Ren Y, Ao Y, O’Shea TM, Kawaguchi R, Coppola G, Khakh BS, Deming TJ, Sofroniew MV (2016) Astrocyte scar formation aids central nervous system axon regeneration. Nature 532(7598):195–200. https://doi.org/10.1038/nature17623
Beck KD, Nguyen HX, Galvan MD, Salazar DL, Woodruff TM, Anderson AJ (2010) Quantitative analysis of cellular inflammation after traumatic spinal cord injury: evidence for a multiphasic inflammatory response in the acute to chronic environment. Brain 133(Pt 2):433–447. https://doi.org/10.1093/brain/awp322
Bellver-Landete V, Bretheau F, Mailhot B, Vallieres N, Lessard M, Janelle ME, Vernoux N, Tremblay ME, Fuehrmann T, Shoichet MS, Lacroix S (2019) Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat Commun 10(1):518. https://doi.org/10.1038/s41467-019-08446-0
Ben-Zvi A, Liebner S (2021) Developmental regulation of barrier- and non-barrier blood vessels in the CNS. J Intern Med. https://doi.org/10.1111/joim.13263
Bhowmick S, D’Mello V, Caruso D, Wallerstein A, Abdul-Muneer PM (2019) Impairment of pericyte-endothelium crosstalk leads to blood–brain barrier dysfunction following traumatic brain injury. Exp Neurol 317:260–270. https://doi.org/10.1016/j.expneurol.2019.03.014
Boven LA, Van Meurs M, Van Zwam M, Wierenga-Wolf A, Hintzen RQ, Boot RG, Aerts JM, Amor S, Nieuwenhuis EE, Laman JD (2006) Myelin-laden macrophages are anti-inflammatory, consistent with foam cells in multiple sclerosis. Brain 129(Pt 2):517–526. https://doi.org/10.1093/brain/awh707
Chapouly C, Tadesse Argaw A, Horng S, Castro K, Zhang J, Asp L, Loo H, Laitman BM, Mariani JN, Straus Farber R, Zaslavsky E, Nudelman G, Raine CS, John GR (2015) Astrocytic TYMP and VEGFA drive blood–brain barrier opening in inflammatory central nervous system lesions. Brain 138(Pt 6):1548–1567. https://doi.org/10.1093/brain/awv077
Chen AQ, Fang Z, Chen XL, Yang S, Zhou YF, Mao L, **a YP, ** HJ, Li YN, You MF, Wang XX, Lei H, He QW, Hu B (2019) Microglia-derived TNF-alpha mediates endothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke. Cell Death Dis 10(7):487. https://doi.org/10.1038/s41419-019-1716-9
Cignarella F, Filipello F, Bollman B, Cantoni C, Locca A, Mikesell R, Manis M, Ibrahim A, Deng L, Benitez BA, Cruchaga C, Licastro D, Mihindukulasuriya K, Harari O, Buckland M, Holtzman DM, Rosenthal A, Schwabe T, Tassi I, Piccio L (2020) TREM2 activation on microglia promotes myelin debris clearance and remyelination in a model of multiple sclerosis. Acta Neuropathol 140(4):513–534. https://doi.org/10.1007/s00401-020-02193-z
Dias DO, Kim H, Holl D, Werne Solnestam B, Lundeberg J, Carlen M, Goritz C, Frisen J (2018) Reducing Pericyte-Derived Scarring Promotes Recovery after Spinal Cord Injury. Cell 173 (1):153–165 e122. https://doi.org/10.1016/j.cell.2018.02.004
Echeverry S, Shi XQ, Rivest S, Zhang J (2011) Peripheral nerve injury alters blood–spinal cord barrier functional and molecular integrity through a selective inflammatory pathway. J Neurosci 31(30):10819–10828. https://doi.org/10.1523/JNEUROSCI.1642-11.2011
Filbin MT (2003) Myelin-associated inhibitors of axonal regeneration in the adult mammalian CNS. Nat Rev Neurosci 4(9):703–713. https://doi.org/10.1038/nrn1195
Gao C, **e R, Li W, Zhou J, Liu S, Cao F, Liu Y, Ma R, Si Y, Liu Y, Bi Y, Gilbert GE, Shi J (2013) Endothelial cell phagocytosis of senescent neutrophils decreases procoagulant activity. Thromb Haemost 109(6):1079–1090. https://doi.org/10.1160/TH12-12-0894
Greenhalgh AD, David S (2014) Differences in the phagocytic response of microglia and peripheral macrophages after spinal cord injury and its effects on cell death. J Neurosci 34(18):6316–6322. https://doi.org/10.1523/JNEUROSCI.4912-13.2014
Hesp ZC, Goldstein EZ, Miranda CJ, Kaspar BK, McTigue DM (2015) Chronic oligodendrogenesis and remyelination after spinal cord injury in mice and rats. J Neurosci 35(3):1274–1290. https://doi.org/10.1523/JNEUROSCI.2568-14.2015
Huang C, Han X, Li X, Lam E, Peng W, Lou N, Torres A, Yang M, Garre JM, Tian GF, Bennett MV, Nedergaard M, Takano T (2012) Critical role of connexin 43 in secondary expansion of traumatic spinal cord injury. J Neurosci 32(10):3333–3338. https://doi.org/10.1523/JNEUROSCI.1216-11.2012
Hulshoff MS, Xu X, Krenning G, Zeisberg EM (2018) Epigenetic Regulation of Endothelial-to-Mesenchymal Transition in Chronic Heart Disease. Arterioscler Thromb Vasc Biol 38(9):1986–1996. https://doi.org/10.1161/ATVBAHA.118.311276
Ihezie SA, Mathew IE, McBride DW, Dienel A, Blackburn SL, Thankamani Pandit PK (2021) Epigenetics in blood–brain barrier disruption. Fluids Barriers CNS 18(1):17. https://doi.org/10.1186/s12987-021-00250-7
Kim KA, Kim D, Kim JH, Shin YJ, Kim ES, Akram M, Kim EH, Majid A, Baek SH, Bae ON (2020) Autophagy-mediated occludin degradation contributes to blood-brain barrier disruption during ischemia in bEnd.3 brain endothelial cells and rat ischemic stroke models. Fluids Barriers 17(1):21. https://doi.org/10.1186/s12987-020-00182-8
Kitchen P, Salman MM, Halsey AM, Clarke-Bland C, MacDonald JA, Ishida H, Vogel HJ, Almutiri S, Logan A, Kreida S, Al-Jubair T, Winkel Missel J, Gourdon P, Tornroth-Horsefield S, Conner MT, Ahmed Z, Conner AC, Bill RM (2020) Targeting aquaporin-4 subcellular localization to treat central nervous system edema. Cell 181 (4):784–799e719. https://doi.org/10.1016/j.cell.2020.03.037
Kobayakawa K, Ohkawa Y, Yoshizaki S, Tamaru T, Saito T, Kijima K, Yokota K, Hara M, Kubota K, Matsumoto Y, Harimaya K, Ozato K, Masuda T, Tsuda M, Tamura T, Inoue K, Edgerton VR, Iwamoto Y, Nakashima Y, Okada S (2019) Macrophage centripetal migration drives spontaneous healing process after spinal cord injury. Sci Adv 5(5):eaav5086. https://doi.org/10.1126/sciadv.aav5086
Kong FQ, Zhao SJ, Sun P, Liu H, Jie J, Xu T, Xu AD, Yang YQ, Zhu Y, Chen J, Zhou Z, Qian DF, Gu CJ, Chen Q, Yin GY, Zhang HW, Fan J (2020) Macrophage MSR1 promotes the formation of foamy macrophage and neuronal apoptosis after spinal cord injury. J Neuroinflammation 17(1):62. https://doi.org/10.1186/s12974-020-01735-2
Kopper TJ, Gensel JC (2018) Myelin as an inflammatory mediator: Myelin interactions with complement, macrophages, and microglia in spinal cord injury. J Neurosci Res 96(6):969–977. https://doi.org/10.1002/jnr.24114
Kotter MR, Li WW, Zhao C, Franklin RJ (2006) Myelin impairs CNS remyelination by inhibiting oligodendrocyte precursor cell differentiation. J Neurosci 26(1):328–332. https://doi.org/10.1523/JNEUROSCI.2615-05.2006
Kozlowski P, Rosicka P, Liu J, Yung AC, Tetzlaff W (2014) In vivo longitudinal Myelin Water Imaging in rat spinal cord following dorsal column transection injury. Magn Reson Imaging 32(3):250–258. https://doi.org/10.1016/j.mri.2013.12.006
Kumar R, Mani AM, Singh NK, Rao GN (2020) PKCtheta-JunB axis via upregulation of VEGFR3 expression mediates hypoxia-induced pathological retinal neovascularization. Cell Death Dis 11(5):325. https://doi.org/10.1038/s41419-020-2522-0
Li Y, Lucas-Osma AM, Black S, Bandet MV, Stephens MJ, Vavrek R, Sanelli L, Fenrich KK, Di Narzo AF, Dracheva S, Winship IR, Fouad K, Bennett DJ (2017) Pericytes impair capillary blood flow and motor function after chronic spinal cord injury. Nat Med 23(6):733–741. https://doi.org/10.1038/nm.4331
Li Y, Lui KO, Zhou B (2018) Reassessing endothelial-to-mesenchymal transition in cardiovascular diseases. Nat Rev Cardiol 15(8):445–456. https://doi.org/10.1038/s41569-018-0023-y
Liu J, Hou W, Guan T, Tang L, Zhu X, Li Y, Hou S, Zhang J, Chen H, Huang Y (2018) Slit2/Robo1 signaling is involved in angiogenesis of glomerular endothelial cells exposed to a diabetic-like environment. Angiogenesis 21(2):237–249. https://doi.org/10.1007/s10456-017-9592-3
Nesic O, Lee J, Ye Z, Unabia GC, Rafati D, Hulsebosch CE, Perez-Polo JR (2006) Acute and chronic changes in aquaporin 4 expression after spinal cord injury. Neuroscience 143(3):779–792. https://doi.org/10.1016/j.neuroscience.2006.08.079
Ng MT, Stammers AT, Kwon BK (2011) Vascular disruption and the role of angiogenic proteins after spinal cord injury. Transl Stroke Res 2(4):474–491. https://doi.org/10.1007/s12975-011-0109-x
Piera-Velazquez S, Jimenez SA (2019) Endothelial to mesenchymal transition: role in physiology and in the pathogenesis of human diseases. Physiol Rev 99(2):1281–1324. https://doi.org/10.1152/physrev.00021.2018
Rengarajan M, Hayer A, Theriot JA (2016) Endothelial Cells Use a Formin-Dependent Phagocytosis-Like Process to Internalize the Bacterium Listeria monocytogenes. PLoS Pathog 12(5):e1005603. https://doi.org/10.1371/journal.ppat.1005603
Rolfe AJ, Bosco DB, Broussard EN, Ren Y (2017) In Vitro Phagocytosis of Myelin Debris by Bone Marrow-Derived Macrophages. J vis Exp. https://doi.org/10.3791/56322
Squair JW, Gautier M, Mahe L, Soriano JE, Rowald A, Bichat A, Cho N, Anderson MA, James ND, Gandar J, Incognito AV, Schiavone G, Sarafis ZK, Laskaratos A, Bartholdi K, Demesmaeker R, Komi S, Moerman C, Vaseghi B, Scott B, Rosentreter R, Kathe C, Ravier J, McCracken L, Kang X, Vachicouras N, Fallegger F, Jelescu I, Cheng Y, Li Q, Buschman R, Buse N, Denison T, Dukelow S, Charbonneau R, Rigby I, Boyd SK, Millar PJ, Moraud EM, Capogrosso M, Wagner FB, Barraud Q, Bezard E, Lacour SP, Bloch J, Courtine G, Phillips AA (2021) Neuroprosthetic baroreflex controls haemodynamics after spinal cord injury. Nature 590(7845):308–314. https://doi.org/10.1038/s41586-020-03180-w
Tan S, Shan Y, Lin Y, Liao S, Zhang B, Zeng Q, Wang Y, Deng Z, Chen C, Hu X, Peng L, Qiu W, Lu Z (2019) Neutralization of interleukin-9 ameliorates experimental stroke by repairing the blood-brain barrier via down-regulation of astrocyte-derived vascular endothelial growth factor-A. FASEB J 33(3):4376–4387. https://doi.org/10.1096/fj.201801595RR
Tran AP, Warren PM, Silver J (2018) The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev 98(2):881–917. https://doi.org/10.1152/physrev.00017.2017
Wang X, Cao K, Sun X, Chen Y, Duan Z, Sun L, Guo L, Bai P, Sun D, Fan J, He X, Young W, Ren Y (2015) Macrophages in spinal cord injury: phenotypic and functional change from exposure to myelin debris. Glia 63(4):635–651. https://doi.org/10.1002/glia.22774
Wang S, Deng J, Fu H, Guo Z, Zhang L, Tang P (2020) Astrocytes directly clear myelin debris through endocytosis pathways and followed by excessive gliosis after spinal cord injury. Biochem Biophys Res Commun. https://doi.org/10.1016/j.bbrc.2020.02.069
Whetstone WD, Hsu JY, Eisenberg M, Werb Z, Noble-Haeusslein LJ (2003) Blood-spinal cord barrier after spinal cord injury: relation to revascularization and wound healing. J Neurosci Res 74(2):227–239. https://doi.org/10.1002/jnr.10759
Yang H, Li X, Liu Y, Li X, Li X, Wu M, Lv X, Chunhua C, Ding X, Zhang Y (2018) Crocin Improves the Endothelial Function. Cell Physiol Biochem 46(2):765–780. https://doi.org/10.1159/000488735
Zhang W, **ong Z, Wei T, Li Q, Tan Y, Ling L, Feng X (2018) Nuclear factor 90 promotes angiogenesis by regulating HIF-1alpha/VEGF-A expression through the PI3K/Akt signaling pathway in human cervical cancer. Cell Death Dis 9(3):276. https://doi.org/10.1038/s41419-018-0334-2
Zhou T, Zheng Y, Sun L, Badea SR, ** Y, Liu Y, Rolfe AJ, Sun H, Wang X, Cheng Z, Huang Z, Zhao N, Sun X, Li J, Fan J, Lee C, Megraw TL, Wu W, Wang G, Ren Y (2019) Microvascular endothelial cells engulf myelin debris and promote macrophage recruitment and fibrosis after neural injury. Nat Neurosci 22(3):421–435. https://doi.org/10.1038/s41593-018-0324-9
Zhu Y, Soderblom C, Krishnan V, Ashbaugh J, Bethea JR, Lee JK (2015) Hematogenous macrophage depletion reduces the fibrotic scar and increases axonal growth after spinal cord injury. Neurobiol Dis 74:114–125. https://doi.org/10.1016/j.nbd.2014.10.024
Acknowledgements
We are grateful for the support of the Scientific Research and Experimental Center of the Second Affiliated Hospital of Anhui Medical University. We thank BioRender (BioRender.com) for Fig. 9.
Funding
This work was founded by the Key Research and Development Program of Anhui Province (Grant Nos. 202004j07020042), the National Natural Science Foundation of China (Grant Nos. 81801220 and 81671204), and the Provincial Natural Science Research Key Project of Colleges and Universities of Anhui Province (Grant No. KJ2019A0257).
Author information
Authors and Affiliations
Contributions
JJ, MZ, and LC designed and supervised the study. FY, YL (2nd author), YC and YL (4th author) performed the experiments and data analysis with technical help from XH, XY, ZL, and YS. FY, YL (2nd author), ZL and DT edited and revised the manuscript. All authors have approved the final submitted manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The animal study was reviewed and approved by the Animal Ethics Committee of Anhui Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yao, F., Luo, Y., Chen, Y. et al. Myelin Debris Impairs Tight Junctions and Promotes the Migration of Microvascular Endothelial Cells in the Injured Spinal Cord. Cell Mol Neurobiol 43, 741–756 (2023). https://doi.org/10.1007/s10571-022-01203-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-022-01203-w