Abstract
Background
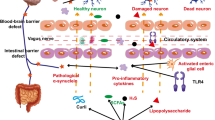
Parkinson’s disease (PD) is viewed as a progressively deteriorating neurodegenerative disorder, the exact etiology of which remains not fully deciphered to this date. The gut microbiota could play a crucial role in PD development by modulating the human immune system.
Objective
This study aims to explore the relationship between gut microbiota and PD, focusing on how immune characteristics may both directly and indirectly influence their interaction.
Methods
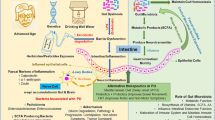
Utilizing cumulative data from genome-wide association studies (GWAS), our research conducted a two-sample Mendelian randomization (MR) analysis to clarify the association between the gut microbiome and PD. Additionally, by employing a two-step MR approach, we assessed the impact of gut microbiota on PD development via immune characteristics and quantified HLA-DR mediation effect on plasmacytoid dendritic cells (pDCs).
Results
We discovered significant associations between PD and microbiota, comprising one class, one order, two families, and two genera. Furthermore, we explored the extent to which HLA-DR on pDCs mediates the effect of Butyrivibrio gut microbiota on PD.
Conclusion
Our study emphasizes the complex interactions between the gut microbiota, immune characteristics, and PD. The relationships and intermediary roles identified in our research provide important insights for develo** potential therapies that target the gut microbiome to alleviate symptoms in PD patients.



Similar content being viewed by others
Data availability
The comprehensive dataset utilized in this research can be accessed publicly through https://gwas.mrcieu.ac.uk/. Study data are accessible upon request from the corresponding author.
References
Miao Y, Meng H (2024) The involvement of α-synucleinopathy in the disruption of microglial homeostasis contributes to the pathogenesis of Parkinson’s disease. Cell Commun Signal 22:31. https://doi.org/10.1186/s12964-023-01402-y
GBD (2015) Neurological Disorders Collaborator Group (2017) Global, regional, and national burden of neurological disorders during 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol 16:877–897. https://doi.org/10.1016/S1474-4422(17)30299-5
Janz C, Timpka J, Rosqvist K et al (2024) Non-motor symptom management: insights into adherence to treatment guidelines in Parkinson’s disease patients. J Park Dis. https://doi.org/10.3233/JPD-230263
Dorsey ER, Bloem BR (2024) Parkinson’s disease is predominantly an environmental disease. J Park Dis. https://doi.org/10.3233/JPD-230357
Tosin MH, Goetz CG, Stebbins GT (2024) Patient with Parkinson disease and care partner perceptions of key domains affecting health-related quality of life. Neurology 102:e208028. https://doi.org/10.1212/WNL.0000000000208028
Jellinger KA (2023) Pathobiology of cognitive impairment in Parkinson disease: challenges and outlooks. Int J Mol Sci 25:498. https://doi.org/10.3390/ijms25010498
Patwardhan A, Kamble N, Bhattacharya A et al (2024) Impact of non-motor symptoms on quality of life in patients with early-onset Parkinson’s disease. Can J Neurol Sci 1–29. https://doi.org/10.1017/cjn.2023.336
Morrone Parfitt G, Coccia E, Goldman C et al (2024) Disruption of lysosomal proteolysis in astrocytes facilitates midbrain organoid proteostasis failure in an early-onset Parkinson’s disease model. Nat Commun 15:447. https://doi.org/10.1038/s41467-024-44732-2
Emmi A, Sandre M, Russo FP et al (2023) Duodenal alpha-synuclein pathology and enteric gliosis in advanced Parkinson’s disease. Mov Disord 38:885–894. https://doi.org/10.1002/mds.29358
Guo B, Zhang J, Zhang W, et al (2023) Gut microbiota-derived short chain fatty acids act as mediators of the gut-brain axis targeting age-related neurodegenerative disorders: a narrative review. Crit Rev Food Sci Nutr 1–22. https://doi.org/10.1080/10408398.2023.2272769
Cheng Y, Tan G, Zhu Q et al (2023) Efficacy of fecal microbiota transplantation in patients with Parkinson’s disease: clinical trial results from a randomized, placebo-controlled design. Gut Microbes 15:2284247. https://doi.org/10.1080/19490976.2023.2284247
Elangovan A, Dahiya B, Kirola L et al (2023) Does gut brain axis has an impact on Parkinson’s disease (PD)? Ageing Res Rev 94:102171. https://doi.org/10.1016/j.arr.2023.102171
Duan W-X, Wang F, Liu J-Y, Liu C-F (2023) Relationship between short-chain fatty acids and Parkinson’s disease: a review from pathology to clinic. Neurosci Bull. https://doi.org/10.1007/s12264-023-01123-9
Nishijima H, Tomiyama M (2024) GABA storage and release from direct pathway neurons account for the enhanced short-duration response of L-dopa in Parkinson’s disease. J Neurol Sci 456:122844. https://doi.org/10.1016/j.jns.2023.122844
Pidcock SE, Skvortsov T, Santos FG et al (2021) Phylogenetic systematics of Butyrivibrio and Pseudobutyrivibrio genomes illustrate vast taxonomic diversity, open genomes and an abundance of carbohydrate-active enzyme family isoforms. Microb Genom 7:000638. https://doi.org/10.1099/mgen.0.000638
Palevich N, Kelly WJ, Leahy SC et al (2019) Comparative genomics of rumen Butyrivibrio spp. uncovers a continuum of polysaccharide-degrading capabilities. Appl Environ Microbiol 86:e01993-e2019. https://doi.org/10.1128/AEM.01993-19
Guo T-T, Zhang Z, Sun Y et al (2023) Neuroprotective effects of sodium butyrate by restoring gut microbiota and inhibiting TLR4 signaling in mice with MPTP-induced Parkinson’s disease. Nutrients 15:930. https://doi.org/10.3390/nu15040930
Ozono T, Kimura Y, Suenaga T et al (2023) Extracellular transportation of α-synuclein by HLA class II molecules. Biochem Biophys Res Commun 644:25–33. https://doi.org/10.1016/j.bbrc.2022.12.082
Barrow AD, Cella M, Edeling MA et al (2023) Cutting edge: PDGF-DD binding to NKp44 costimulates TLR9 signaling and proinflammatory cytokine secretion in human plasmacytoid dendritic cells. J Immunol ji2200496. https://doi.org/10.4049/jimmunol.2200496
Pihlstrøm L, Wiethoff S, Houlden H (2017) Genetics of neurodegenerative diseases: an overview. Handb Clin Neurol 145:309–323. https://doi.org/10.1016/B978-0-12-802395-2.00022-5
Lopera-Maya EA, Kurilshikov A, van der Graaf A et al (2022) Effect of host genetics on the gut microbiome in 7,738 participants of the Dutch Microbiome Project. Nat Genet 54:143–151. https://doi.org/10.1038/s41588-021-00992-y
Orrù V, Steri M, Sidore C et al (2020) Complex genetic signatures in immune cells underlie autoimmunity and inform therapy. Nat Genet 52:1036–1045. https://doi.org/10.1038/s41588-020-0684-4
Genomes Project Consortium, Auton A, Brooks LD et al (2015) A global reference for human genetic variation. Nature 526:68–74. https://doi.org/10.1038/nature15393
Hartikainen AK, Khan I, Karjalainen EK et al (2024) Microbiota and mucosal gene expression of fecal microbiota transplantation or placebo treated patients with chronic pouchitis. Gut Microbes 16:2295445. https://doi.org/10.1080/19490976.2023.2295445
Vascellari S, Palmas V, Melis M et al (2020) Gut microbiota and metabolome alterations associated with Parkinson’s disease. mSystems 5:e00561–20. https://doi.org/10.1128/mSystems.00561-20
**e A, Ensink E, Li P et al (2022) Bacterial butyrate in Parkinson’s disease is linked to epigenetic changes and depressive symptoms. Mov Disord 37:1644–1653. https://doi.org/10.1002/mds.29128
Wei B-R, Zhao Y-J, Cheng Y-F et al (2024) Helicobacter pylori infection and Parkinson’s disease: etiology, pathogenesis and levodopa bioavailability. Immun Ageing 21:1. https://doi.org/10.1186/s12979-023-00404-1
Huang G, Khan R, Zheng Y et al (2023) Exploring the role of gut microbiota in advancing personalized medicine. Front Microbiol 14:1274925. https://doi.org/10.3389/fmicb.2023.1274925
Stolzer I, Scherer E, Süß P et al (2023) Impact of microbiome-brain communication on neuroinflammation and neurodegeneration. Int J Mol Sci 24:14925. https://doi.org/10.3390/ijms241914925
Wang H, Zhang M, Li J et al (2022) Gut microbiota is causally associated with poststroke cognitive impairment through lipopolysaccharide and butyrate. J Neuroinflammation 19:76. https://doi.org/10.1186/s12974-022-02435-9
Engen PA, Dodiya HB, Naqib A et al (2017) The potential role of gut-derived inflammation in multiple system atrophy. J Parkinsons Dis 7:331–346. https://doi.org/10.3233/JPD-160991
Kurita N, Yamashiro K, Kuroki T et al (2020) Metabolic endotoxemia promotes neuroinflammation after focal cerebral ischemia. J Cereb Blood Flow Metab 40:2505–2520. https://doi.org/10.1177/0271678X19899577
Zhou X, Gao Y, Wei J et al (2023) The intestinal microbiota exerts a sex-specific influence on neuroinflammation in a Parkinson’s disease mouse model. Neurochem Int 173:105661. https://doi.org/10.1016/j.neuint.2023.105661
Jia X, Chen Q, Zhang Y, Asakawa T (2023) Multidirectional associations between the gut microbiota and Parkinson’s disease, updated information from the perspectives of humoral pathway, cellular immune pathway and neuronal pathway. Front Cell Infect Microbiol 13:1296713. https://doi.org/10.3389/fcimb.2023.1296713
**e Z, Zhang M, Luo Y et al (2023) Healthy human fecal microbiota transplantation into mice attenuates MPTP-induced neurotoxicity via AMPK/SOD2 pathway. Aging Dis 14:2193–2214. https://doi.org/10.14336/AD.2023.0309
Acknowledgements
The authors express their gratitude to all participants and researchers involved in the GWAS study, the IEU OpenGWAS project, the NHGRI-EBI Catalog of Human Genome-Wide Association Studies, and other GWAS datasets for their contributions and data sharing.
Funding
This research was supported by the Autonomous Region Key Research and Development Project (Grant No. 2023B03003), the Tian-Shan Talent Program (Grant No. 2022TSYCLJ0066), the National Natural Science Foundation of China (Grant No. 82371258), and the Central Guiding Local Science and Technology Development Special Fund Project (Grant No. ZYYD2022C17).
Author information
Authors and Affiliations
Contributions
Zihao Wang: research design and manuscript drafting. Huan **a and Tingting Feng: data analysis. Adilai Aibibuli: manuscript editing and language polishing.**nling Yang and Mingyang Zhang: research design and peer review. All authors have reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The utilized data were sourced from accessible online public databases, and the submission has already received approval from the hospital’s ethics committee report in accordance, rendering further ethical approval unnecessary.
Consent to participate
This study did not involve any human experiments. All analyses were based on publicly available whole-genome association study data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The manuscript has not been published elsewhere and is not under consideration by any other journal.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Z., **a, H., Feng, T. et al. The role of HLA-DR on plasmacytoid dendritic cells in mediating the effects of Butyrivibrio gut microbiota on Parkinson’s disease. Neurol Sci (2024). https://doi.org/10.1007/s10072-024-07467-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10072-024-07467-y