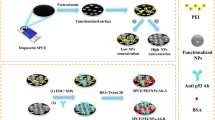
Abstract
A poly(thiophene acetic acid)/Au/poly(methylene blue) nanostructured interface was electrochemically assembled step-by-step on screen-printed carbon electrodes (SPCE) for label-free detection of p53 protein. The initial electrical conductive properties of the polymeric interface were increased with an additional layer of poly(methylene blue) electropolymerized in the presence of gold nanoparticles. The nano-immunosensing architecture was prepared by covalent immobilization of anti-p53 antibodies as bioreceptors through the poly(thiophene acetic acid) moieties. The nano-immunosensor assembly was extensively characterized by ultraviolet–visible spectrophotometry, dynamic and electrophoretic light scattering, scanning electron microscopy, X-ray photoelectron spectroscopy, Raman spectroscopy, atomic force microscopy, cyclic voltammetry, and electrochemical impedance spectroscopy. Under optimal conditions, p53 was specifically and selectively detected by square wave voltammetry in a linear range between 1 and 100 ng mL−1 with a limit of detection of 0.65 ng mL−1. In addition, the electrochemical nano-immunosensor detected p53 in spiked human serum samples and colorectal cancer cell lysates, and the results were validated with a standard spectrophotometric method using a paired samples t test, which did not exhibit significant differences between both methods. The resultant p53 nano-immunosensor is simple to assemble, robust, and has the potential for point-of-care biomarker detection applications.
Graphical abstract







Similar content being viewed by others
References
Aydemir N, Malmström J, Travas-Sejdic J (2016) Conducting polymer based electrochemical biosensors. Phys Chem Chem Phys 18:8264–8277. https://doi.org/10.1039/c5cp06830d
Ibanez JG, Rincón ME, Gutierrez-Granados S et al (2018) Conducting polymers in the fields of energy, environmental remediation, and chemical–chiral sensors. Chem Rev 118:4731–4816. https://doi.org/10.1021/acs.chemrev.7b00482
Cruz-Pacheco AF, Quinchia J, Orozco J (2022) Cerium oxide-doped PEDOT nanocomposite for label-free electrochemical immunosensing of anti-p53 autoantibodies. Microchim Acta 189:228. https://doi.org/10.1007/s00604-022-05322-5
Kakhki S, Barsan MM, Shams E, Brett CMA (2012) Development and characterization of poly(3,4-ethylenedioxythiophene)-coated poly(methylene blue)-modified carbon electrodes. Synth Met 161:2718–2726. https://doi.org/10.1016/j.synthmet.2011.10.007
Abad-Gil L, Brett CMA (2022) Poly(methylene blue)-ternary deep eutectic solvent/Au nanoparticle modified electrodes as novel electrochemical sensors: optimization, characterization and application. Electrochim Acta 434:141295. https://doi.org/10.1016/j.electacta.2022.141295
Qin J, Cho M, Lee Y (2019) Ultrasensitive detection of amyloid-β using cellular prion protein on the highly conductive Au nanoparticles-poly(3,4-ethylene dioxythiophene)-poly(thiophene-3-acetic acid) composite electrode. Anal Chem 91:11259–11265. https://doi.org/10.1021/acs.analchem.9b02266
Promsuwan K, Meng L, Suklim P et al (2020) Bio-PEDOT: modulating carboxyl moieties in poly(3,4-ethylenedioxythiophene) for enzyme-coupled bioelectronic interfaces. ACS Appl Mater Interfaces 12:39841–39849. https://doi.org/10.1021/acsami.0c10270
Attallah AM, Abdel-Aziz MM, El-Sayed AM, Tabll AA (2003) Detection of serum p53 protein in patients with different gastrointestinal cancers. Cancer Detect Prev 27:127–131. https://doi.org/10.1016/S0361-090X(03)00024-2
Deepa Nohwal B, Chaudhary R, Pundir CS (2022) Amperometric detection of tumor suppressor protein p53 via pencil graphite electrode for fast cancer diagnosis. Anal Biochem 639:114528. https://doi.org/10.1016/j.ab.2021.114528
Aydın M, Aydın EB, Sezgintürk MK (2018) A disposable immunosensor using ITO based electrode modified by a star-shaped polymer for analysis of tumor suppressor protein p53 in human serum. Biosens Bioelectron 107:1–9. https://doi.org/10.1016/j.bios.2018.02.017
Quinchia J, Echeverri D, Cruz-Pacheco AF et al (2020) Electrochemical biosensors for determination of colorectal tumor biomarkers. Micromachines 11:1–46. https://doi.org/10.3390/MI11040411
Rodrigues NR, Rowan A, Smith ME et al (1990) p53 mutations in colorectal cancer. Proc Natl Acad Sci 87:7555–7559. https://doi.org/10.1073/pnas.87.19.7555
Esokkiya A, Sudalaimani S, Sanjeev Kumar K et al (2021) Poly(methylene blue)-based electrochemical platform for label-free sensing of acrylamide. ACS Omega 6:9528–9536. https://doi.org/10.1021/acsomega.0c06315
Barsan MM, Pinto EM, Brett CMA (2011) Methylene blue and neutral red electropolymerisation on AuQCM and on modified AuQCM electrodes: an electrochemical and gravimetric study. Phys Chem Chem Phys 13:5462–5471. https://doi.org/10.1039/C1CP20418A
Barsan MM, Pinto EM, Brett CMA (2008) Electrosynthesis and electrochemical characterisation of phenazine polymers for application in biosensors. Electrochim Acta 53:3973–3982. https://doi.org/10.1016/j.electacta.2007.10.012
Marinho MIC, Cabral MF, Mazo LH (2012) Is the poly (methylene blue)-modified glassy carbon electrode an adequate electrode for the simple detection of thiols and amino acid-based molecules? J Electroanal Chem 685:8–14. https://doi.org/10.1016/j.jelechem.2012.08.023
Verma S, Singh A, Shukla A et al (2017) Anti-IL8/AuNPs-rGO/ITO as an immunosensing platform for noninvasive electrochemical detection of oral cancer. ACS Appl Mater Interfaces 9:27462–27474. https://doi.org/10.1021/acsami.7b06839
Brett CMA (2022) Electrochemical impedance spectroscopy in the characterisation and application of modified electrodes for electrochemical sensors and biosensors. Molecules 27:1497
Bertok T, Lorencova L, Chocholova E et al (2019) Electrochemical impedance spectroscopy based biosensors: mechanistic principles, analytical examples and challenges towards commercialization for assays of protein cancer biomarkers. ChemElectroChem 6:989–1003. https://doi.org/10.1002/celc.201800848
Echeverri D, Orozco J (2022) β-1,4-Galactosyltransferase-V colorectal cancer biomarker immunosensor with label-free electrochemical detection. Talanta 243:123337. https://doi.org/10.1016/j.talanta.2022.123337
González-Sánchez MI, Gómez-Monedero B, Agrisuelas J et al (2019) Electrochemical performance of activated screen printed carbon electrodes for hydrogen peroxide and phenol derivatives sensing. J Electroanal Chem 839:75–82. https://doi.org/10.1016/j.jelechem.2019.03.026
Galdino FE, Smith JP, Kwamou SI et al (2015) Graphite screen-printed electrodes applied for the accurate and reagentless sensing of pH. Anal Chem 87:11666–11672. https://doi.org/10.1021/acs.analchem.5b01236
Echeverri D, Cruz-Pacheco AF, Orozco J (2023) Capacitive nanobiosensing of β-1,4-galactosyltransferase-V colorectal cancer biomarker. Sensors Actuators B Chem 374:132784. https://doi.org/10.1016/j.snb.2022.132784
Rashed MA, Faisal M, Ahmed J et al (2022) Highly sensitive and selective amperometric hydrazine sensor based on Au nanoparticle-decorated conducting polythiophene prepared via oxidative polymerization and photo-reduction techniques. J Saudi Chem Soc 26:101480. https://doi.org/10.1016/j.jscs.2022.101480
Andreoli E, Rooney DA, Redington W et al (2011) Electrochemical deposition of hierarchical micro/nanostructures of copper hydroxysulfates on polypyrrole−polystyrene sulfonate films. J Phys Chem C 115:8725–8734. https://doi.org/10.1021/jp200465n
Eby DM, Artyushkova K, Paravastu AK, Johnson GR (2012) Probing the molecular structure of antimicrobial peptide-mediated silica condensation using X-ray photoelectron spectroscopy. J Mater Chem 22:9875–9883. https://doi.org/10.1039/C2JM30837A
Elshafey R, Siaj M, Tavares AC (2016) Au nanoparticle decorated graphene nanosheets for electrochemical immunosensing of p53 antibodies for cancer prognosis. Analyst 141:2733–2740. https://doi.org/10.1039/c6an00044d
Björneholm O, Federmann F, Kakar S, Möller T (1999) Between vapor and ice: free water clusters studied by core level spectroscopy. J Chem Phys 111:546–550. https://doi.org/10.1063/1.479334
Schroeder HW, Cavacini L (2010) Structure and function of immunoglobulins. J Allergy Clin Immunol 125:S41–S52. https://doi.org/10.1016/j.jaci.2009.09.046
Prasad KS, Chen J-C, Ay C, Zen J-M (2007) Mediatorless catalytic oxidation of NADH at a disposable electrochemical sensor. Sensors Actuators B Chem 123:715–719. https://doi.org/10.1016/j.snb.2006.10.012
Velický M, Toth PS, Woods CR et al (2019) Electrochemistry of the basal plane versus edge plane of graphite revisited. J Phys Chem C 123:11677–11685. https://doi.org/10.1021/acs.jpcc.9b01010
Li C, Huang Y, Lai K et al (2016) Analysis of trace methylene blue in fish muscles using ultra-sensitive surface-enhanced Raman spectroscopy. Food Control 65:99–105. https://doi.org/10.1016/j.foodcont.2016.01.017
Lorenzen AL, dos Santos AM, dos Santos LP et al (2022) PEDOT-AuNPs-based impedimetric immunosensor for the detection of SARS-CoV-2 antibodies. Electrochim Acta 404:139757. https://doi.org/10.1016/j.electacta.2021.139757
Pfaffen V, Ortiz PI, Córdoba de Torresi SI, Torresi RM (2010) On the pH dependence of electroactivity of poly(methylene blue) films. Electrochim Acta 55:1766–1771. https://doi.org/10.1016/j.electacta.2009.10.062
Gunda NSK, Singh M, Norman L et al (2014) Optimization and characterization of biomolecule immobilization on silicon substrates using (3-aminopropyl)triethoxysilane (APTES) and glutaraldehyde linker. Appl Surf Sci 305:522–530. https://doi.org/10.1016/j.apsusc.2014.03.130
Mitsakakis K, Lousinian S, Logothetidis S (2007) Early stages of human plasma proteins adsorption probed by atomic force microscope. Biomol Eng 24:119–124. https://doi.org/10.1016/j.bioeng.2006.05.013
Pedrero M, Manuel de Villena FJ, Muñoz-San Martín C et al (2016) Disposable amperometric immunosensor for the determination of human P53 protein in cell lysates using magnetic micro-carriers. Biosensors 6:56. https://doi.org/10.3390/bios6040056
Yeo J, Park J-Y, Bae WJ et al (2009) Label-free electrochemical detection of the p53 core domain protein on its antibody immobilized electrode. Anal Chem 81:4770–4777. https://doi.org/10.1021/ac900301h
García-Miranda Ferrari A, Rowley-Neale SJ, Banks CE (2021) Screen-printed electrodes: transitioning the laboratory in-to-the field. Talanta Open 3:100032. https://doi.org/10.1016/j.talo.2021.100032
Acknowledgements
We thank the Ruta N Complex and EPM for hosting the Max Planck Tandem Groups. Finally, we thank Dr. Ernesto Moreno and Dr. Marcela Rubio from the University of Medellín for donating cell lines.
Funding
The work has been funded by Minciencias, Mineducación, MINCIT, and ICETEX through the Program Ecosistema Científico Cod. FP44842-211–2018, project number 58536. J.O. thanks support from the University of Antioquia and the Max Planck Society through the cooperation agreement 566–1, 2014.
Author information
Authors and Affiliations
Contributions
Andrés F. Cruz-Pacheco, conceptualization, methodology, formal analysis, investigation, data curation, and writing, original draft. Jennifer Quinchia, investigation, data curation, and writing, original draft. Jahir Orozco, conceptualization; formal analysis; writing, review and editing; supervision, project administration; and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cruz-Pacheco, A.F., Quinchia, J. & Orozco, J. Nanostructured poly(thiophene acetic acid)/Au/poly(methylene blue) interface for electrochemical immunosensing of p53 protein. Microchim Acta 190, 136 (2023). https://doi.org/10.1007/s00604-023-05683-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-05683-5