Abstract
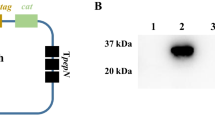
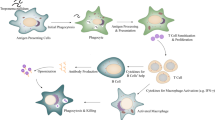
This study aimed to compare the immune response against Toxoplasma gondii (T. gondii) in BALB/c mice induced by excreted/secreted (E/S) antigens and mannose-modified nanoliposome of E/S antigens. Here, E/S antigens and mannose-modified nanoliposome of E/S antigens were firstly prepared, and then BALB/c female inbred mice were separately immunized. In the next step, anti-E/S antigen antibodies and the relative expression levels of IL-10 and IL-12 mRNA were detected by ELISA and real-time PCR, respectively. After immunization, mice were intraperitoneally challenged with 102 tachyzoites of T. gondii, and the survival rate was recorded. The ELISA analysis showed significant differences between the levels of anti-E/S antigen antibodies in the mice immunized by E/S antigens and those immunized by mannose-modified nanoliposome of E/S antigens at days 7, 10, 20, 25, and 30 (P < 0.05). Real-time PCR analysis showed that the relative expression of IL-10 was significantly decreased during 20 days. Yet, the relative expression of IL-12 was significantly increased during 20 days (P < 0.05). In T. gondii challenge test, significant differences were found between the survival rates of mice immunized by E/S antigens and mice immunized by mannose-modified nanoliposome with E/S antigens. This project evidenced that mannose-modified nanoliposome of E/S antigens induced a more powerful immune response against T. gondii in BALB/c mice when compared with excreted/secreted antigens alone.



Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Abdollahi SH, Arababadi MK, Hassanshahi G (2009) Evaluation of excreted/secreted antigens derived from peritoneal of toxoplasma infected small mice to detect IgG against Toxoplasma. Pak J Biol Sci 12(6):530–533
Almeida MPO, Ferro EAV, Briceño MPP, Oliveira MC, Barbosa BF, Silva NM (2019) Susceptibility of human villous (BeWo) and extravillous (HTR-8/SVneo) trophoblast cells to Toxoplasma gondii infection is modulated by intracellular iron availability. Parasitol Res 118:1559–1572
Angus C, Klivington-Evans D, Dubey J, Kovacs JA (2000) Immunization with a DNA plasmid encoding the SAG1 (P30) protein of Toxoplasma gondii is immunogenic and protective in rodents. J Infect Dis 181:317–324
Blader IJ, Saeij JP (2009) Communication between Toxoplasma gondii and its host: impact on parasite growth, development, immune evasion, and virulence. Apmis 117:458–476
Bottari NB et al (2020) Neuroprotective role of resveratrol mediated by purinergic signalling in cerebral cortex of mice infected by Toxoplasma gondii. Parasitol Res 119:2897–2905
Bülow R, Boothroyd J (1991) Protection of mice from fatal Toxoplasma gondii infection by immunization with p30 antigen in liposomes. J Immunol 147:3496–3500
Cui YL, He SY, Xue MF, Zhang J, Wang HX, Yao Y (2008) Protective effect of a multiantigenic DNA vaccine against Toxoplasma gondii with co-delivery of IL-12 in mice. Parasite Immunol 30:309–313
Darcy F et al (1988) Induction of a protective antibody-dependent response against toxoplasmosis by in vitro excreted/secreted antigens from tachyzoites of Toxoplasma gondii. Parasite Immunol 10:553–567
Daryani A, Hosseini AZ, Dalimi A (2003) Immune responses against excreted/secreted antigens of Toxoplasma gondii tachyzoites in the murine model. Vet Parasitol 113:123–134
Dziadek B, Gatkowska J, Brzostek A, Dziadek J, Dzitko K, Dlugonska H (2009) Toxoplasma gondii: the immunogenic and protective efficacy of recombinant ROP2 and ROP4 rhoptry proteins in murine experimental toxoplasmosis. Exp Parasitol 123:81–89
El-Malky M et al. (2005) Protective effect of vaccination with Toxoplasma lysate antigen and CpG as an adjuvant against Toxoplasma gondii in susceptible C57BL/6 mice. Microbiol Immunol 49:639– 646
Fakhravar Z, Ebrahimnejad P, Daraee H, Akbarzadeh A (2016) Nanoliposomes: synthesis methods and applications in cosmetics. J Cosmetic Laser Ther 18:174–181
Hiszczyńska-Sawicka E et al (2012) Induction of immune responses in sheep by vaccination with liposome-entrapped DNA complexes encoding Toxoplasma gondii MIC3 gene. Pol J Vet Sci 15(1):3–9
Innes EA, Bartley PM, Maley S, Katzer F, Buxton D (2009) Veterinary vaccines against Toxoplasma gondii Memórias do instituto oswaldo cruz 104:246-251
Konstantinovic N, Guegan H, Stäjner T, Belaz S, Robert-Gangneux F (2019) Treatment of toxoplasmosis: current options and future perspectives. Food Waterborne Parasitol 15:e00036
Kotresha D, Noordin R (2010) Recombinant proteins in the diagnosis of toxoplasmosis. APMIS 118:529–542
Lau YL, Fong MY (2008) Toxoplasma gondii: serological characterization and immunogenicity of recombinant surface antigen 2 (SAG2) expressed in the yeast Pichia pastoris. Exp Parasitol 119:373–378
Lee Y-H, Shin D-W, Lee J-H, Nam H-W, Ahn M-H (2007) Vaccination against murine toxoplasmosis using recombinant Toxoplasma gondii SAG3 antigen alone or in combination with Quil A. Yonsei Med J 48(3):396–404
Machado NI, Dos Santos TAT, de Souza W, DaMatta RA, Seabra SH (2020) Treatment with melatonin induces a reduction of Toxoplasma gondii development in LLC-MK2 cells. Parasitol Res 119:2703–2711
Nguyen TX, Huang L, Gauthier M, Yang G, Wang Q (2016) Recent advances in liposome surface modification for oral drug delivery. Nanomedicine 11:1169–1185
Perrie Y, Crofts F, Devitt A, Griffiths HR, Kastner, Elisabeth and Nadella, Vinod (2016) Designing liposomal adjuvants for the next generation of vaccines. Advanced Drug Delivery Reviews, 99 (Pt A). pp. 85–96. ISSN 0169–409X
Suzuki Y (2020) The immune system utilizes two distinct effector mechanisms of T cells depending on two different life cycle stages of a single pathogen, Toxoplasma gondii, to control its cerebral infection. Parasitol Int 76:102030
Wang N, Chen M, Wang T (2019) Liposomes used as a vaccine adjuvant-delivery system: From basics to clinical immunization. J Control Release 303:130–150
Yamamoto Y, Mineo J, Meneghisse C, Guimaraes A, Kawarabayashi M (1998) Detection in human sera to excreted secreted antigens Toxoplasma gondii by use immunoblot assay of IgG, IgM and IgA from of dot. ELISA Annals Trop Med Parasitol 92:23–30
Zhao Y et al (2020) Mannose-modified liposome co-delivery of human papillomavirus type 16 E7 peptide and CpG oligodeoxynucleotide adjuvant enhances antitumor activity against established large TC-1 grafted tumors in mice. Int J Nanomed 15:9571–9586
Acknowledgements
This study was performed in the Molecular Medicine Research Center, Research Institute of Basic Medical Sciences, Rafsanjan University of Medical Sciences, Rafsanjan, Iran. The authors would like to thank all the colleagues who contributed.
Funding
This study was financially supported by Rafsanjan University of Medical Sciences, Rafsanjan, Iran, under the ethical code of IR.RUMS.REC.1396.78 at RUMS.
Author information
Authors and Affiliations
Contributions
S.H.A., contributed to the design and implementation of the research. H.E.S., participated in the collection of data. S.K.F. and N.A., contributed substantially to the drafting, writing, and revising of the manuscript. M.K.A., contributed to the analysis and interpretation of data. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Nawal Hijjawi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdollahi, S.H., Shahmabadi, H.E., Arababadi, M.K. et al. The immune response against Toxoplasma gondii in BALB/c mice induced by mannose-modified nanoliposome of excreted/secreted antigens. Parasitol Res 120, 2855–2861 (2021). https://doi.org/10.1007/s00436-021-07232-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07232-6