Abstract
Cancer is the second leading cause of mortality worldwide. Despite recent advances in cancer treatment including immunotherapy with immune checkpoint inhibitors, new unconventional biomarkers and targets for the detection, prognosis, and treatment of cancer are still in high demand. Tumor cells are characterized by mutations that allow their unlimited growth, program their local microenvironment to support tumor growth, and spread towards distant sites. While a major focus has been on altered tumor genomes and proteomes, crucial signaling molecules such as lipids have been underappreciated. One of these molecules is the membrane phospholipid phosphatidylserine (PS) that is usually found at cytosolic surfaces of cellular membranes but can be rapidly and massively shuttled to the extracellular leaflet of the plasma membrane during apoptosis to serve as a limiting factor for immune responses. These immunosuppressive interactions are exploited by tumor cells to evade the immune system. In this review, we describe mechanisms of immune regulation in tumors, discuss if PS may constitute an inhibitory immune checkpoint, and describe current and future strategies for targeting PS to reactivate the tumor-associated immune system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer is the collective term for a large number of diseases that are caused by somatic mutations, leading to unchecked expansion of the cells in which the mutations occurred. This expansion eventually disturbs the physiology of the organ in which the transformation occurred. Moreover, during cancer development, which in humans can take decades, transformed cell can acquire the ability to leave the organ of origin and colonize distant sites to form metastases. Distant metastasis is a major reason why, despite considerable progress in cancer therapy, cancer remains a leading cause of non-natural death globally [17]. While the goal of cancer therapy is to remove the transformed cells from the body, it has become apparent that focusing therapeutic efforts merely on the cancer cells alone may not be sufficient. Rather, the complex interplay of the cancer cells with their cellular and molecular neighborhood, the tumor microenvironment (TME), needs to be considered and ideally be targeted to aid in removing the transformed cells. This is of particular importance because cancer cells acquire the ability to subvert their local microenvironment from a naturally tumor-suppressive into a tumor-supporting state [14, 25, 30].
The TME is composed of a specific extracellular matrix, various gradients of nutrients and gaseous molecules, and different cell types such as vascular and lymphatic endothelial cells, mural cells, fibroblasts, and immune cells [33]. A major limiting factor of tumor growth is the immune system, which is able to sense neo-antigens and stress-induced molecules in transformed cells, which under normal circumstances leads to eradication of the develo** cancer [7, 71, 83]. The occurrence of clinically detectable tumors therefore indicates that immune control has failed. Indeed, many advanced tumors are characterized by a tumor-promoting rather than protective immune response. Identifying the factors that shape the formation of such a detrimental immune environment with the aim to interfere with their action to re-activate the immune system against a tumor has consequentially become a major research goal. Hereby, tumors appear to exploit physiological mechanisms that have evolved to limit immune reactions during disturbed tissue homeostasis or to switch off immunity once the trigger of disturbed homeostasis has been removed. These central mechanisms that decide if an immune reaction is initiated, persists, or is terminated are called immune checkpoints [73]. They are commonly viewed as intercellular receptor-ligand interactions. In this review, we discuss under which conditions PS may serve as an immune checkpoint in cancer and discuss the underlying mechanisms and strategies to target PS to overcome tumor-associated immune suppression.
Immune checkpoints in cancer
A considerable part of the attention related to research for cancer therapy within the last two decades has been directed to the field of cancer immunotherapy. This is due to several key findings including the prognostic relevance of the immune response in patients, the success of cellular immunotherapy, and immune checkpoint blockade (ICB). The cellular composition of the tumor-associated immune system and the functional profile of these cells have emerged as a powerful predictor of prognosis for cancer patients [8]. The presence of immune cells with an activation profile reminiscent of anti-microbial or anti-viral defense responses such as CD8 + T cells, T helper 1 (TH1)-polarized CD4 + T cells, memory T cells, NK cells, γδ T cells, B cells, and activated myeloid cells in the TME is associated with a favorable outcome, while immunosuppressive myeloid cells and regulatory T cells (Treg) that are able to limit the anti-tumor properties of the former cells predict poor prognosis in most cancers [8, 116]. An immunosuppressive TME is not only prognostically relevant, but also a major hurdle for immunotherapy approaches that aim at supplying protective immune cells to cancer patients [55]. The mechanisms that render the TME immunosuppressive are highly diverse in nature [93], but disrupting inhibitory immune checkpoints has taken center stage due to its clinical efficacy [106, 107].
ICB is an approach to prevent immunosuppressive molecular interactions mediated by inhibitory immune checkpoints by the administration of neutralizing antibodies. Immune checkpoints are defined as the engagement of a receptor expressed on immune cells by its ligands, whose expression is often more widespread. Receptor engagement either enhances or suppresses effector function of the cells expressing the receptor to constitute either co-stimulatory or co-inhibitory immune checkpoints [107]. Physiologically, co-inhibitory immune checkpoints are engaged to prevent autoimmunity and to modulate the duration and extent of immune responses [73]. These inhibitory checkpoints are usually not expressed or expressed at low levels on resting cells but are upregulated upon activation to provide negative feedback signaling opportunities. A major focus has been on such immune checkpoints expressed by T cells that modulate the second signal of T cell activation following the recognition of antigens presented by major histocompatibility molecules via their specific T cell receptor [85]. The most understood co-inhibitory immune checkpoint molecules are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed death 1 (PD-1), both regulating distinct non-redundant inhibitory signaling pathways [106]. CTLA-4 is upregulated on T cells after antigen recognition and competes with the co-stimulatory molecule CD28 for its ligands, the B7 family molecules CD80 and CD86 [45], which are expressed on antigen-presenting cells following their activation. CTLA-4 negatively regulates T cell activation to fine-tune or terminate immune reactions by a remarkable plethora of mechanisms, including the removal of the CD28 ligands from the immunological synapse by trans-endocytosis [76, 106]. Allison and colleagues demonstrated that the application of CTLA-4 blocking antibodies resulted in complete tumor rejection and long-lasting immunity in mice due to a potentiated anti-tumor immune response [49]. PD-1 is also upregulated on T cells after antigen-dependent activation but is expressed by other cells such as B cells and macrophages as well [1]. PD-1 interaction with its ligands PD-L1 and PD-L2, which in comparison to CD28/CTLA-4 ligands are widely expressed on a variety of cell types [69], predominantly interferes negatively with T cell receptor-dependent signaling [23]. Honjo and colleagues demonstrated that PD-1-deficient mice were prone to develop autoimmunity indicating PD-1 as a crucial negative regulator of lymphocyte signaling [34, 70]. These remarkable pre-clinical findings sparked further research efforts, culminating in the first immune checkpoint disrupting antibodies entering clinical trials. Remarkable clinical response rates for CTLA-4 and PD-1 blocking antibodies were observed in melanoma patients [29, 48, 81], which was followed by a number of further clinical studies in other cancer entities [106]. Most importantly, ICB yielded long-term survival benefits in some cancer patients. Interfering with the PD-1/PD-L1 immune checkpoint showed superior clinical benefit compared to CTLA-4 blockade, which might be due to the less restricted expression pattern of PD-1 and its ligands, particularly PD-L1 in tumor cells.
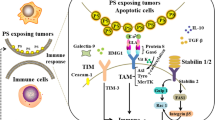
Despite this remarkable success story, ICB only works in a subgroup of patients, and induced resistance to ICB has emerged from follow-up studies [86]. However, the predictive power of the immune response holds even in cancer entities where current ICB strategies show low efficacy. This indicates that new targets to activate the immune system against cancer are needed. Hereby, other immune cells, apart from those that interact primarily or directly against the tumor, should be considered. For instance, tumor-associated macrophages are key contributors to sha** the tumor-promoting TME and are able to remove tumor cells via phagocytosis [122]. The signals that educate macrophages to support tumor growth are manifold, including the recognition of dying tumor cells [24, 108]. Physiological cell death and particularly the recognition of dying cell-derived molecules can serve as a signal limiting inflammatory macrophage activation under physiological conditions, and may therefore be viewed as a myeloid cell-targeted immune checkpoint [109]. Hereby, PS is a central signal to recognize dying cells, induce their removal by efferocytosis, and induce repair pathways in macrophages (Fig. 1).
The PS immune checkpoint. PS is externalized under conditions of tissue damage, e.g., during inflammation. Externalized PS is recognized by macrophages, which are then programmed towards resolution of inflammation and tissue repair. Thus, PS is a signal to limit and terminate immune reactions and can therefore be considered a non-conventional inhibitory immune checkpoint
Phosphatidylserine metabolism
PS is an essential anionic glycerophospholipid that comprises about 3–10% of total phospholipids in mammalian cells under physiological conditions, which is the scenario the following description is focusing on. Glycerophospholipids are amphiphilic molecules that are the major constituents of all cellular membranes. They are usually composed of a central glycerol moiety with two hydrophobic ester-linked fatty acyl groups in the sn1 and sn2 position and various different hydrophilic headgroups linked to the glycerol via a phosphate molecule in the sn3 position, which makes them amphiphilic. Lysophospholipids with a fatty acid only in the sn1 or sn2 position and glycerophospholipids with ether-linked fatty alcohols also exist, albeit in lower quantities. Importantly, the distribution of glycerophospholipids in membrane bilayers, generally being arranged in a manner that the hydrophobic tails are directed towards the center of the bilayer and the hydrophilic heads are oriented towards intracellular or extracellular fluids, is not uniform. Rather, a discrete distribution of glycerophospholipids with different headgroups is essential for eukaryotic cell physiology [27, 118]. PS is highly enriched in the cytosolic layer of cellular membranes, even though it can be found at the luminal side of the membrane bilayer in cellular organelles such as the endoplasmic reticulum, mitochondria, and the Golgi complex [18, 97]. The asymmetric distribution of PS in cellular membranes is established and maintained by ATP-dependent flippases that catalyze the localization of PS to the cytosolic leaflet of the plasma membrane [68]. This resulting state appears to be highly stable [43], but specific PS scramblases exist that are able to rapidly interfere with the asymmetrical distribution of PS in an ATP-independent manner, resulting in its exposure at the extracellular leaflet of the plasma membrane [68].
PS is synthesized by two distinct phosphatidylserine synthase (PTDSS) enzymes, PTDSS1 and PTDSS2 [104]. Both enzymes are localized in the endoplasmic reticulum in mitochondria-associated membranes [103], where they catalyze a base-exchange reaction replacing the polar headgroup of phosphatidylcholine (PC) or phosphatidylethanolamine (PE) with serine. PTDSS1 and PTDSS2 differ in their expression profiles in distinct cell types [3, 5, 92] and in their substrate specificity. While PTDSS2 utilizes exclusively PE as substrate, PTDSS1 appears to be more promiscuous, accepting both PC and PE [42]. Particularly, PTDSS1 may be involved in generating ether-linked PS species [84], even though PTDSS2 can also accept ether-linked PE as substrate with relatively low efficacy [41]. Ether-linked PS are probably rare in cells even though data are limited. In the CNS, 13% and 0.3% 1-O-alkenyl hydrocarbon chains that result from linking fatty alcohols to glycerol were found in PS in white matter and gray matter, respectively, while PE contained 47% and 21% 1-O-alkenyl hydrocarbon chains in white and gray matter [39]. Thus, ether-linked PS levels may vary between 0.01 and 1.3% among glycerophospholipids in tissues. Despite these substrate preferences, PTDSS isoforms are able to compensate for each other. Even though mice lacking Ptdss1 showed 85% reduced serine-exchange capability, the overall PS content was unaltered due to the compensatory action of PTDSS2. A similar phenomenon was reported vice-versa in Ptdss2−/− mice, while double deletion of PTDSS1 and PTDSS2 was lethal [3]. Degradation of PS occurs in mitochondria by phosphatidylserine decarboxylase (PISD), which decarboxylates the serine headgroup in PS to ethanolamine thereby generating PE. Transport of PS from mitochondria-associated membranes to other cellular membranes, including the plasma membrane, has been reviewed in detail [37, 42] and mainly involves vesicular transport and a minor contribution of soluble lipid transfer proteins.
Localization of PS both at the cytosolic and the extracellular leaflet of the plasma membrane mediates crucial cellular functions, which are largely defined by its anionic headgroup. Hereby, the net negative charge results from the combination of the serine and phosphate moieties. Cytosolically oriented PS provides a major proportion of the negative charge density of the plasma membrane’s inner leaflet, allowing a number of proteins to interact with PS. This occurs either in a non-specific manner due to the presence of polycationic regions in their primary structure, which are found for instance in Ras- and Rho-GTPases and the protein kinase Src, or in a specific manner via C2 domains which bind Ca2+ to link proteins such as protein kinase C or phosphoinositide-3-kinase to PS and, consequently, the plasma membrane [37, 59]. Localization of these proteins to the plasma membrane is essential for their functionality, which includes triggering major survival pathways in cells. This is in line with findings that higher eukaryotic cells lacking the machinery to synthesize PS do not survive [3]. These intracellular signaling properties of PS are active during steady-state conditions. In contrast, PS is externalized or otherwise visible to the extracellular milieu when cellular homeostasis is disturbed. Here, it may under some circumstances act similar to an inhibitory immune checkpoint.
PS and its receptors in immunity
Intracellular PS is part of platforms for triggering crucial signaling modules for immune cell activation such as protein kinase C [2] or pathways downstream of antigen recognition by T cell receptors [113]. Its exposure at the extracellular leaflet of the plasma membrane occurs in a variety of conditions, including immune cell activation, platelet activation, and cell death, and usually requires suppression of flippase activity, induction of scramblase activity, or both [6, 37]. Hereby, the expression of PS at the outer plasma membrane leaflet of viable cells is generally a transient event that is required during developmental processes such as myoblast or osteoclast fusion. PS exposure on activated immune cells is known for a long time, but the physiological relevance remains unknown. Activated neutrophils transiently expose PS, which is not related to cell death [19]. In mast cells, PS exposure is induced after stimulation of the IgE receptor or experimental elevation of cytosolic Ca2+, and degranulation occurs, indicating that PS exposure could be associated with exocytosis [60]. Indeed, there is further evidence supporting a role in exocytosis, since PS exposure is observed at exocytic sites of activated chromaffin cells [72]. T cells may also express PS in the outer leaflet of the plasma membrane without undergoing apoptosis [87]. Interestingly, non-apoptotic PS exposure on T cells promotes infection with human immunodeficiency virus (HIV)-1 and is triggered by the viral envelope glycoprotein of HIV-1 itself interacting with CD4. PS exposure during HIV-1 infection depends on Ca2+-dependent activation of TMEM16F scramblase, and blocking PS exposure protects from infection [120]. Also during platelet activation, Ca2+-dependent activation of TMEM16F scramblase and probably inactivation of P4 ATPase family flippases result in PS exposure, serving as a platform for serine protease activation during coagulation [68]. Hereby, serine proteases in the coagulation cascade bind to PS at the platelet surface via their Ca2+-containing gamma-carboxyglutamic (Gla) domain. Production of active thrombin during that process can in turn propagate inflammation [10].
In contrast to these mechanisms that may promote immune reactions, PS exposed on the surface of apoptotic cells avoids or even actively prevents and resolves inflammation [6]. PS is exposed on the surface of apoptotic cells due to caspase-dependent inactivation of P4 ATPase family flippases and activation of XK-related protein family scramblases, particularly Xkr8 [68]. PS exposure is just one of the consequences of apoptotic cell death. Downstream effects of caspase activation to execute apoptosis comprise loss of organelle integrity, DNA fragmentation, and plasma membrane blebbing with preserved plasma membrane integrity to generate apoptotic bodies. These are then removed by macrophages before secondary necrosis can occur. Apoptosis occurs very frequently during homeostatic conditions and is therefore non-inflammatory and non-immunogenic to maintain tolerance and to prevent a systemic auto-immune response [61, 108, 110]. It is important to note that in the context of cell death, PS exposure does not selectively occur during apoptosis. Various modes of cell death including apoptosis, necrosis, and necroptosis are characterized by the exposure of PS towards the extracellular milieu. However, the specific mechanisms and kinetics appear to be different, including the loss of overall plasma membrane integrity and accessibility of the extracellular milieu to its inner leaflet during necrosis, ion fluctuations (Ca2+ and Cl−), and caspase activity, which affect flippase and scramblase activity [88]. In each of these cases, PS serves as a signal to remove the cellular debris, a so-called eat-me signal. However, particularly during apoptosis, its properties surpass serving as a first signal for debris removal. Recognition of PS by a large array of receptors is one of the reasons why apoptosis is non-immunogenic, inducing active signaling events to counteract inflammatory signaling and to activate tolerogenic pathways [6]. This property of PS may even be used to actively trigger the resolution of inflammation as PS-containing liposomes acting on local macrophages in inflammation models restricted inflammatory leukocyte recruitment, while PC-containing liposomes did not [90].
Sensing of PS in the extracellular space requires dedicated receptors. There are several PS receptors that may act redundantly, but also in a context-specific manner to allow tailored reactions to dying cells [61] (Table 1). PS receptors include brain angiogenesis inhibitor 1 (BAI1) [46], stabilin-1/2, αVβ3-5 integrins [26, 78], CD300 family proteins [12, 65, 89], T cell/transmembrane, immunoglobulin, and mucin (TIM) family proteins, and TYRO3, AXL, and MER (TAM) receptor tyrosine kinases. BAI1, stabilin-1/2, and the TIM family proteins directly bind to PS, while TAM and integrin receptors indirectly bind to PS via bridging molecules [74]. TAM receptors bind PS via the Gla domain-containing proteins growth arrest-specific 6 (GAS6) and protein S (PROS1), and integrin receptors bind to PS via MFGE8 [6, 61].
Stabilin-1 and stabilin-2 are type I membrane protein receptors expressed in primary and secondary hematopoietic organs and other immune-related organs such as lymph nodes, spleen, bone marrow, and the liver that upon binding to PS on apoptotic cells initiate phagocytosis [40]. Anti-inflammatory macrophages that are associated with tissue repair have been shown to express stabilin-1 [75]. Stabilin-2 is also expressed in macrophages and participates in TGF-β production, which is an immunosuppressive cytokine produced by efferocytic macrophages that regulates immune cell generation and function [75].
The TIM family consists of type I cell surface glycoproteins, which have been identified as direct PS receptors. TIM-1 is expressed on CD4 T cells, mast cells, and B cells. Interaction of TIM-1 with PS reduces T cell activation and cytokine production [20, 99, 117]. TIM-4 is expressed on dendritic cells and macrophages [112]. Particularly in liver macrophages expressing TIM-4, PS recognition via TIM-4 attenuated NLRP3 inflammasome activation in the context of nonalcoholic fatty liver diseases [57]. TIM-3 is a co-inhibitory receptor that is designated as an immune checkpoint and is expressed on a variety of cells including diverse T cell subsets, dendritic cells, and macrophages [63]. Several ligands for TIM-3 beside PS were identified, explaining its broad anti-inflammatory relevance in transplant tolerance, autoimmunity, viral infections, and cancer [111].
The TAM family proteins are linked to PS via the bridging molecules GAS6 and PROS1, where the amino terminus binds to PS in a Ca2+-dependent manner while the carboxy terminus binds to the individual TAM receptor. The three TAM receptors share a similar structural organization but are expressed on different cells, show different modes of interaction with GAS6 and PROS1, and may be linked to different signaling pathways [54, 82, 96]. For instance, mice lacking TYRO3 show neurological disorders, mice lacking AXL show vascular problems, while MERTK-deficient mice present autoimmunity issues. However, all three appear to contribute to efferocytosis and regulate inflammation [15]. MERTK, the most studied TAM receptor regarding immune response, is mainly expressed by macrophages, even though expression on other cells such as dendritic cells, natural killer cells, B cells, and T cells has been noted [21]. MERTK signaling in macrophages plays a role in both inflammatory disease and cancer and has generally been connected with the resolution of inflammation and tissue repair. In contrast, triggering AXL-dependent signaling on macrophages can have detrimental and even diametrically opposed functional outcomes compared to MERTK [15, 119].
The data summarized above indicate that PS recognition on the surface of apoptotic cells is usually anti-inflammatory, but context-specific responses are enabled by receptor diversity. It is important to highlight that not all externalized PS causes the equal responses. Externalized PS on viable cells or on activated platelets does not classically result in their removal by macrophages, and PS externalized in the context of necrosis does at least not strongly limit the inflammatory properties of necrotic cells [88]. The downstream effects of recognition of externalized PS probably depend on concentration, topology, or its clustering capacity, as demonstrated for its interaction with AXL [62]. Future studies will need to address the composition of the lipid and protein environment of PS, saturation of fatty acids and fatty alcohols in PS, and oxidative status, e.g., by mass spectrometry to assess under which conditions the presentation of PS to the extracellular space leads to immunosuppressive or stimulatory outcomes. However, it appears reasonable to assume that tumors may utilize the undeniable immunosuppressive potential of PS to evade the immune system.
Role of PS in cancer
Assuming that tumors may exploit the immunosuppressive nature of PS would indicate that a significant degree of cell death happens in tumors. This assumption is not trivial as protection from cell death is a hallmark of cancer. While this is certainly true in a subpopulation of cancer cells, the proliferative index in tumors usually comes along with a higher degree of cell death compared to the parent tissue [108, 110]. Thus, tumors as such can use the non-immunogenic properties of PS to hinder their eradication by the immune system by sacrificing a proportion of their own. While this occurs under steady-state conditions, the immunosuppressive properties of PS may become even more relevant upon cancer treatment. The main therapy regimens used in clinical practice including surgery, chemo-, and radiotherapy induce either cell death in tumor cells themselves or may cause death of local tissue cells or rapidly dividing cells systemically, which will cause a significant degree of immunosuppressive PS exposure [38]. Also, killing tumor cells by current immunotherapies including ICB will induce significant PS exposure on the dying tumor cells. Consequently, targeting PS and its receptors in cancer might produce synergistic effects when combined with current therapy regimens.
Indeed, several PS receptors have been connected to tumor immunity. For instance, mice with a deletion of stabilin-1 in macrophages showed reduced tumor growth compared to control mice [75]. The TAM family receptors are often classified as proto-oncogenic receptors in tumor cells themselves and have been described in different types of cancers, playing a role in proliferation, migration, survival, and chemo-resistance properties. High expression of these receptors is related with poor prognosis and cell aggressiveness. This may, among others, also stem from their role in recognizing PS expressed on (dying) tumor cells [66]. Indeed, the removal of apoptotic cells by MERTK-expressing macrophages induced an anti-inflammatory phenotype and promoted metastasis in breast cancer [91]. MERTK blockade by antibodies led to an anti-tumor response characterized by type 1 interferon production, with additive effects after combination with ICB [123]. Mice harboring MERTK-deficient myeloid cells showed tumor resistance, slower tumor growth, and enhanced expression of inflammatory cytokines, accompanied by higher presence of CD8 T cells. Depletion of CD8 T cells restored tumor growth in this model. These effects were observed in melanoma and mammary carcinoma models, but MERTK blockade caused an increase in the growth of colorectal cancer models [13]. Such opposite effects in different cancer models may be related to a heterogeneous expression of PS in the tumor environments and/or the presence of alternative PS receptors. However, in a model of acute lymphoblastic leukemia (ALL), the inhibition of MERTK decreased tumor burden and prolonged survival. MERTK inhibition decreased the expression of PD-L1 and PD-L2 on monocytes/macrophages and decreased PD-1 expression in T cells, leading to increased CD4 + and CD8 + T cell activation [50]. Also, AXL/GAS6 signaling was actively exploited by leukemic cells to generate a suppressive TME by driving macrophages towards a tumor-promoting phenotype. Depletion of both AXL or GAS6 in macrophages stimulated NK cell- and T cell-dependent immunity against the leukemia cells and sensitized to ICB [95]. These data summarized above suggest that targeting the TAM receptor family holds the potential to potentiate anti-tumor effects of ICB in different types of cancer.
Other efforts to utilize PS dysregulation in the TME have focused on targeting PS itself. Some blocking strategies consist of the administration of PS ligands such as Annexin A5 (AnxA5) or others, which may slow tumor progression and increase the immunogenicity of tumor cells [56]. In addition to merely blocking PS recognition with AnxA5, fusing AnxA5 with other peptides or proteins has been investigated. For instance, bacterial L-methionase, which catalyzes the degradation of the essential amino acid L-methionine, was linked to AnxA5 to target it specifically to tumor cells. This led to reduced methionine availability and furthermore induced the conversion of selenomethionine into toxic methylselenol, thereby killing tumor cells and inhibiting tumor growth [102]. Moreover, tagging tumor antigens to AnxA5 significantly enhanced its immunogenicity and anti-tumor efficacy when administered after chemotherapy, which as expected increased PS exposure. The efficacy of re-activating the immune system with AnxA5-dependent delivery of tumor antigens was further enhanced by ICB [36, 52]. Besides AnxA5, antibodies against PS were tested in tumor models. When the PS-binding antibody 2aG4 was used in a model of prostate cancer in combination with chemotherapy, the immune profile of the TME shifted to stimulate anti-tumor responses, which was characterized by an increased abundance of pro-inflammatory macrophages and mature dendritic cells suppressing progression of the tumors [114]. Another preclinical PS targeting antibody, mch1N11, improved the anti-tumor immune response of radiotherapy in a melanoma model, which was further improved when ICB was added. The combination generated a potent cytotoxic T cell response even against contra-laterally placed non-irradiated tumors [9]. Lactadherin, another multifunctional glycoprotein, binds PS-enriched cell surfaces in a Ca2+-independent manner. This interaction is crucial for regulation of blood coagulation, but it was also observed that lactadherin affects the reprogramming of pro-tumoral anti-inflammatory macrophages, reducing glioma growth [35], probably by preventing the recognition of PS.
In addition to targeting PS receptors or PS exposure to the extracellular milieu, recent findings have suggested that reducing PS synthesis may be beneficial in cancer. PTDSS1 expression was frequently upregulated in different cancer types and was associated with poor survival [84]. At least in murine breast tumor cells, increased PTDSS1 expression resulted in increased PS levels [84]. Deletion of PTDSS1 in breast cancer cells reduced PS levels and tumor growth, which was associated with reduced abundance of macrophages, due to reduced proliferation, in tumors. This phenotype was mimicked in mice lacking MERTK in macrophages, suggesting that PS/MERTK signaling was needed for macrophage proliferation and subsequent support of tumor growth [84]. Hereby, PTDSS1 deletion mainly affected the production of ether-linked PS species but reduced PS exposure upon apoptosis. How ether-linked PS regulates PS exposure during apoptosis remains unclear. Interestingly, increased PS exposure upon PTDSS1 overexpression has been reported previously [100], and early reports suggested that PS exposed during apoptosis was derived from a pool of newly synthesized PS, but a dependency on PTDSS1 or PTDSS2 was not found [105]. Thus, PTDSS1 dependency may be exclusive to tumor cells. This may be of particular relevance given the fact that PTDSS2 expression is lost in a variety of cancer types [115]. Disrupting PTDSS1 activity may therefore specifically affect tumor cells rather than untransformed cells that still express PTDSS2 to produce PS. Accordingly, potent and selective PTDSS1 inhibitors were developed, and their application in PTDSS2-deficient tumor models resulted in tumor regression [115]. This inhibition caused the activation of the endoplasmic reticulum stress response, which mediated cell death and anti-tumor immunity activation, specifically stimulating dendritic cells. Importantly, increased immunogenicity translated into protection against PTDSS2-WT tumor cells in the surrounding, indicating the induction of overall protective immunity [115]. Thus, targeting PTDSS1 appears to interfere with the immunosuppressive TME by reducing the exposure of PS in the outer membrane leaflet, which importantly may not affect efferocytosis per se [6, 84]. The use of pharmacological compounds targeting PTDSS1 may be of interest for tumor therapy. A potential synergy with ICB would also need to be explored, based on the reports above indicating such a synergy between inhibiting PS receptors and ICB.
Another potential role played by PS during tumor immune responses is being a tumor antigen itself, which would be highly expressed due to increased PTDSS1-dependent production in tumors. The inherent diversity of lipids makes them excellent candidates as antigens [64]. Lipid antigens are presented on the surface of antigen-presenting cells via CD1 molecules and are sensed by natural killer T cells (NKT cells), a heterogeneous population of lymphocytes [101]. Among NKT cells, tumor-suppressive type I NKT and tumor-promoting type II NKT cells have been identified [44, 94]. Indeed, lyso-PS species can be recognized by type II NKT cells [84]. Whether and how PS-reactive type II NKT cells affect tumor immunity remains to be determined. Interestingly, CD1d-bound PS may be recognized by PS receptors as well, adding a potential new twist to the immunoregulatory potential of PS [47].
Clinical implications and strategies to utilize PS exposure for cancer therapy
The evidence summarized above supports the rationale for targeting PS in cancer to overcome immunosuppression [11]. Different pre-clinical and clinical strategies are currently being developed to interfere with PS in cancer (Fig. 2). Besides being a target for immune activation against tumors, PS has been proposed as a marker for clinical monitoring of the efficacy of cytotoxic therapy, i.e., to determine the amount of cell death induced. A number of clinical studies have been performed to investigate the suitability of (99 m)Tc-Annexin A5 as a molecular imaging agent [4]. While data are promising, there appears to be a need for standardization for clinical use. Increased PS exposure on tumor cells and tumor-infiltrating blood vessels is also being tested as a target to direct liposomes specifically towards tumors. Liposomes are lipid vesicles that can be used as drug carriers. Liposomal carriers were synthesized to bind to PS via Saposin C (SapC), a glycoprotein involved in the activation of lysosomal enzymes and ceramide production, which shows a strong affinity for PS. Vesicles containing SapC and dioleylphosphatidylserine (SapC-DOPS) have been used to selectively target different types of cancer in vitro and in vivo while these liposomes do not bind to normal tissue due to low cell surface PS expression. Moreover, they can be used in combination with standard of care therapies [67]. A phase 1 clinical trial with SapC-DOPS is running since 2019, and the last update suggested a tolerable safety profile [80].
Preclinical and clinical strategies to target phosphatidylserine (PS) in cancer. PS in tumor cells is produced particularly via PS synthase 1 (PTDSS1). It is exposed on the tumor-associated vasculature, living tumor cells, and particularly on dying tumor cells via scramblases. PS is recognized by macrophages, but also other cells, via a divergent set of PS receptors including TAM family receptors, TIM family receptors, and stabilins. PS serves as a signal to remove cellular debris by efferocytosis and induces anti-inflammatory macrophage activation and proliferation. This interaction promotes tumor progression among others by limiting the activity of cytotoxic lymphocytes. PS recognition by macrophages can be interrupted by PS neutralizing antibodies, Annexin A5, and similar PS binding proteins and small molecule inhibitors of PS receptors. Reactivation of cytotoxic lymphocytes by these approaches synergizes with immune checkpoint blockade (ICB). Clinically tested PS antibodies require β2-glycoprotein 1 (β2GP1) to bind to PS and appear to act predominantly at the tumor-associated vasculature. Annexin A5 and similar PS binding proteins can also be linked to imaging or cytotoxic agents or tumor antigens to aid in tumor cell detection or killing. Details can be found in the main text
The most prominent approach to interfere with PS for immunotherapy is masking externalized PS with an antibody. The monoclonal antibody bavituximab binds to β2-glycoprotein I to subsequently induce its binding to PS with high affinity [58]. Bavituximab has been and is being investigated in clinical trials addressing different types of cancer such as lung, breast, pancreatic, and hepatocellular carcinoma [6]. Unfortunately, a phase III clinical trial for late-stage non-squamous non-small cell lung carcinoma showed no significant improvement of overall survival for patients who received the combination of docetaxel and bavituximab compared to docetaxel alone. However, a potential benefit of combining bavituximab with ICB emerged from this trial [22]. The clinical potential of combining bavituximab with the ICB antibody pembrolizumab is currently explored in phase II trials in gastric and hepatocellular carcinoma [32, 51]. The data reported so far indicate the efficacy of the combination at least in a subgroup of patients that can be identified by specific biomarkers [98]. One major limitation of bavituximab and the preclinical antibodies indicated above may be the requirement of β2GP1 to target PS instead of targeting PS directly. Moreover, these antibodies preferentially bind to the tumor-associated vasculature, which may restrict their potential to reach (dying) tumor cells with externalized PS in order to disrupt their interaction with macrophages and other immune cells [6, 58]. Thus, a major driver of efficacy of these antibodies may be innate immune-driven antibody-dependent cellular cytotoxicity against tumor endothelial cells [77] rather than blocking the immunosuppressive interaction between dying tumor cells and phagocytes. On the other hand, preferential targeting of the tumor-associated vasculature rather than dying tumor cells may avoid undesired side effects. Since PS is a major eat-me signal on dying cells, decreased recognition by phagocytes may lead to an accumulation of apoptotic cells which subsequently can become secondary necrotic, with toxic effects including the tumor lysis syndrome or auto-immune reactions such as the antiphospholipid syndrome [16, 31]. However, as indicated above, decreasing PS levels in tumor cells by deleting PTDSS1 did affect macrophage activation but not efferocytosis [84]. Other eat-me signals together with reduced amounts of exposed PS may suffice for efficient removal of dying cells [61]. Thus, PS-recognizing antibodies with a different mode of action or alternative strategies to interfere with PS recognition should be clinically tested in the future. These would include PTDSS1 inhibitors, particularly in PTDSS2-deficient tumors. Moreover, interfering with PS transport from the endoplasmic reticulum to the plasma membrane may be worth investigating. Generally, mechanisms underlying the synthesis of PS, its transport, distribution, and recycling, need to be studied in more depth to identify new and hopefully selective targets to decrease or increase PS bioavailability depending on the type of therapy needed and the type of cell involved. Of course, the broader the biological function of the target, the more the likelihood of severe side effects increases given the important role of PS as scaffold for intracellular signaling pathways and the fact that billions of cells are cleared daily by efferocytosis.
Conclusions and outlook
The evidence summarized above supports an important role for PS as an immunomodulatory signal that can be clinically targeted in cancer, even though optimized strategies need to be developed. The outcome of clinical trials of combining bavituximab with ICB in different cancers will provide further insights into the clinical potential of PS-targeting agents. However, alternative approaches to interfere with externalized PS cancer need to be studied as well. These include molecules derived from AnxA5 and alternative agents such as GlaS, a protein derived from the Gla domain of PROS1. Such agents could be used not only to mask PS but also to deliver drugs specifically to the TME [28].
Extracellularly approachable PS probably cannot be considered a prototypical immune checkpoint, even though its upregulation on the extracellular dying cells occurs during an overshooting immune reaction with collateral damage, and is able to limit the ongoing immune reaction to avoid further damage (Fig. 2). PS is upregulated also on living cells and during immune cell activation, where it may promote rather than restrict inflammation. Moreover, some PS receptors such as AXL may under specific circumstances promote detrimental immune reactions. However, PS can serve as a prototypical example for molecular interactions that are frequently overlooked in cancer, i.e., interactions of lipids with other signaling molecules. Considerable efforts have been and are being undertaken to acquire genomic and proteomic information of cancer patients with the idea to identify druggable alterations in these molecules. The overall metabolic consequences of tumor-associated mutations are still largely unexplored, including changes in the lipidome [53]. The few studies that have addressed the tumor lipidome have found remarkable alterations in the composition of lipids forming biological membranes. These alterations affect parameters such as membrane fluidity, which determines mechanic properties of cells, cellular signaling, e.g., due to an altered formation of membrane microdomains, drug uptake, and interaction of cancer cells with the TME [79, 121]. Future systematic efforts are needed to study the cancer lipidome and its impact on the TME. Recent advances in spatial lipidomics will be instrumental towards the latter. This notion also applies to studying the biology of PS per se, particularly the different modes of PS externalization and its recognition. It is still unclear how much PS is actually needed to serve as an eat-me signal if there is a hierarchy of receptors with different affinity and avidity to specific PS species including ether-linked PS, local concentrations, arrangement, or distribution. Increasing sensitivity and development of standards and protocols for unbiased and targeted lipid mass spectrometry, as well as tracing studies using high resolution microscopy, will be helpful to determine these parameters.
Data availability
No datasets were generated or analysed during the current study.
References
Agata Y, Kawasaki A, Nishimura H, Ishida Y, Tsubata T, Yagita H, Honjo T (1996) Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes. Int Immunol 8:765–772. https://doi.org/10.1093/intimm/8.5.765
Altman A, Kong KF (2016) Protein kinase C enzymes in the hematopoietic and immune systems. Annu Rev Immunol 34:511–538. https://doi.org/10.1146/annurev-immunol-041015-055347
Arikketh D, Nelson R, Vance JE (2008) Defining the importance of phosphatidylserine synthase-1 (PSS1): unexpected viability of PSS1-deficient mice. J Biol Chem 283:12888–12897. https://doi.org/10.1074/jbc.M800714200
Belhocine TZ, Blankenberg FG, Kartachova MS, Stitt LW, Vanderheyden JL, Hoebers FJ, Van de Wiele C (2015) (99m)Tc-Annexin A5 quantification of apoptotic tumor response: a systematic review and meta-analysis of clinical imaging trials. Eur J Nucl Med Mol Imaging 42:2083–2097. https://doi.org/10.1007/s00259-015-3152-0
Bergo MO, Gavino BJ, Steenbergen R, Sturbois B, Parlow AF, Sanan DA, Skarnes WC, Vance JE, Young SG (2002) Defining the importance of phosphatidylserine synthase 2 in mice. J Biol Chem 277:47701–47708. https://doi.org/10.1074/jbc.M207734200
Birge RB, Boeltz S, Kumar S, Carlson J, Wanderley J, Calianese D, Barcinski M, Brekken RA, Huang X, Hutchins JT, Freimark B, Empig C, Mercer J, Schroit AJ, Schett G, Herrmann M (2016) Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer. Cell Death Differ 23:962–978. https://doi.org/10.1038/cdd.2016.11
Boon T, Cerottini JC, Van den Eynde B, van der Bruggen P, Van Pel A (1994) Tumor antigens recognized by T lymphocytes. Annu Rev Immunol 12:337–365. https://doi.org/10.1146/annurev.iy.12.040194.002005
Bruni D, Angell HK, Galon J (2020) The immune contexture and immunoscore in cancer prognosis and therapeutic efficacy. Nat Rev Cancer 20:662–680. https://doi.org/10.1038/s41568-020-0285-7
Budhu S, Giese R, Gupta A, Fitzgerald K, Zappasodi R, Schad S, Hirschhorn D, Campesato LF, De Henau O, Gigoux M, Liu CL, Mazo G, Deng L, Barker CA, Wolchok JD, Merghoub T (2021) Targeting phosphatidylserine enhances the anti-tumor response to tumor-directed radiation therapy in a preclinical model of melanoma. Cell Reports 34. ARTN 108620.https://doi.org/10.1016/j.celrep.2020.108620
Burzynski LC, Humphry M, Pyrillou K, Wiggins KA, Chan JNE, Figg N, Kitt LL, Summers C, Tatham KC, Martin PB, Bennett MR, Clarke MCH (2019) The coagulation and immune systems are directly linked through the activation of interleukin-1alpha by thrombin. Immunity 50(1033–1042):e1036. https://doi.org/10.1016/j.immuni.2019.03.003
Chang W, Fa H, **ao D, Wang J (2020) Targeting phosphatidylserine for cancer therapy: prospects and challenges. Theranostics 10:9214–9229. https://doi.org/10.7150/thno.45125
Choi SC, Simhadri VR, Tian LJ, Gil-Krzewska A, Krzewski K, Borrego F, Coligan JE (2011) Cutting edge: mouse CD300f (CMRF-35-like molecule-1) recognizes outer membrane-exposed phosphatidylserine and can promote phagocytosis. J Immunol 187:3483–3487. https://doi.org/10.4049/jimmunol.1101549
Cook RS, Jacobsen KM, Wofford AM, DeRyckere D, Stanford J, Prieto AL, Redente E, Sandahl M, Hunter DM, Strunk KE, Graham DK, Earp HS 3rd (2013) MerTK inhibition in tumor leukocytes decreases tumor growth and metastasis. J Clin Invest 123:3231–3242. https://doi.org/10.1172/JCI67655
Coussens LM, Zitvogel L, Palucka AK (2013) Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science 339:286–291. https://doi.org/10.1126/science.1232227
DeBerge M, Glinton K, Subramanian M, Wilsbacher LD, Rothlin CV, Tabas I, Thorp EB (2021) Macrophage AXL receptor tyrosine kinase inflames the heart after reperfused myocardial infarction. J Clin Invest 131. https://doi.org/10.1172/JCI139576
Ding ZT, Pan HY, Yang ZX, Yang CD, Shi H (2023) Beyond the classics: the emerging value of anti-phosphatidylserine/ prothrombin antibodies in antiphospholipid syndrome. Clinical Immunology 256. ARTN 109804.https://doi.org/10.1016/j.clim.2023.109804
Diseases GBD, Injuries C (2020) Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1204–1222. https://doi.org/10.1016/S0140-6736(20)30925-9
Fairn GD, Schieber NL, Ariotti N, Murphy S, Kuerschner L, Webb RI, Grinstein S, Parton RG (2011) High-resolution map** reveals topologically distinct cellular pools of phosphatidylserine. J Cell Biol 194:257–275. https://doi.org/10.1083/jcb.201012028
Frasch SC, Henson PM, Nagaosa K, Fessler MB, Borregaard N, Bratton DL (2004) Phospholipid flip-flop and phospholipid scramblase 1 (PLSCR1) co-localize to uropod rafts in formylated Met-Leu-Phe-stimulated neutrophils. J Biol Chem 279:17625–17633. https://doi.org/10.1074/jbc.M313414200
Freeman GJ, Casasnovas JM, Umetsu DT, DeKruyff RH (2010) genes: a family of cell surface phosphatidylserine receptors that regulate innate and adaptive immunity. Immunol Rev 235:172–189
Gautier EL, Chow A, Spanbroek R, Marcelin G, Greter M, Jakubzick C, Bogunovic M, Leboeuf M, van Rooijen N, Habenicht AJ, Merad M, Randolph GJ (2012) Systemic analysis of PPARgamma in mouse macrophage populations reveals marked diversity in expression with critical roles in resolution of inflammation and airway immunity. J Immunol 189:2614–2624. https://doi.org/10.4049/jimmunol.1200495
Gerber DE, Horn L, Boyer M, Sanborn R, Natale R, Palmero R, Bidoli P, Bondarenko I, Germonpre P, Ghizdavescu D, Kotsakis A, Lena H, Losonczy G, Park K, Su WC, Tang M, Lai J, Kallinteris NL, Shan JS, Reck M, Spigel DR (2018) Randomized phase III study of docetaxel plus bavituximab in previously treated advanced non-squamous non-small-cell lung cancer. Ann Oncol 29:1548–1553. https://doi.org/10.1093/annonc/mdy177
Greenwald RJ, Freeman GJ, Sharpe AH (2005) The B7 family revisited. Annu Rev Immunol 23:515–548. https://doi.org/10.1146/annurev.immunol.23.021704.115611
Gregory CD, Paterson M (2018) An apoptosis-driven ‘onco-regenerative niche’: roles of tumour-associated macrophages and extracellular vesicles. Philos Trans R Soc Lond B Biol Sci 373. https://doi.org/10.1098/rstb.2017.0003
Greten FR, Grivennikov SI (2019) Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51:27–41. https://doi.org/10.1016/j.immuni.2019.06.025
Hanayama R, Nagata S (2005) Impaired involution of mammary glands in the absence of milk fat globule EGF factor 8. Proc Natl Acad Sci U S A 102:16886–16891. https://doi.org/10.1073/pnas.0508599102
Harayama T, Riezman H (2018) Understanding the diversity of membrane lipid composition. Nat Rev Mol Cell Biol 19:281–296. https://doi.org/10.1038/nrm.2017.138
Hardy J, Bauzon M, Chan CKF, Makela AV, Kanada M, Schneider D, Blankenberg F, Contag CH, Hermiston T (2023) Gla-domain mediated targeting of externalized phosphatidylserine for intracellular delivery. FASEB J 37:e23113. https://doi.org/10.1096/fj.202201250RRR
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723. https://doi.org/10.1056/NEJMoa1003466
Hou J, Karin M, Sun B (2021) Targeting cancer-promoting inflammation - have anti-inflammatory therapies come of age? Nat Rev Clin Oncol 18:261–279. https://doi.org/10.1038/s41571-020-00459-9
Howard SC, Jones DP, Pui CH (2011) The tumor lysis syndrome. New Engl J Med 364:1844–1854. https://doi.org/10.1056/NEJMra0904569
Hsieh D, Kainthla R, Zhu H, Beg MS (2023) Phase 2 trial of pembrolizumab (pembro) and bavituximab (bavi) in advanced hepatocellular carcinoma (HCC). J Clin Oncol 41:584–584
Hui L, Chen Y (2015) Tumor microenvironment: sanctuary of the devil. Cancer Lett 368:7–13. https://doi.org/10.1016/j.canlet.2015.07.039
Ishida Y, Agata Y, Shibahara K, Honjo T (1992) Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J 11:3887–3895. https://doi.org/10.1002/j.1460-2075.1992.tb05481.x
Kaminska A, Enguita FJ, Stepien EL (2018) Lactadherin: an unappreciated haemostasis regulator and potential therapeutic agent. Vascul Pharmacol 101:21–28. https://doi.org/10.1016/j.vph.2017.11.006
Kang TH, Park JH, Yang A, Park HJ, Lee SE, Kim YS, Jang GY, Farmer E, Lam B, Park YM, Hung CF (2020) Annexin A5 as an immune checkpoint inhibitor and tumor-homing molecule for cancer treatment. Nat Commun 11:1137. https://doi.org/10.1038/s41467-020-14821-z
Kay JG, Fairn GD (2019) Distribution, dynamics and functional roles of phosphatidylserine within the cell. Cell Commun Signal 17:126. https://doi.org/10.1186/s12964-019-0438-z
Kaynak A, Davis HW, Kogan AB, Lee JH, Narmoneva DA, Qi X (2022) Phosphatidylserine: the unique dual-role biomarker for cancer imaging and therapy. Cancers (Basel) 14. https://doi.org/10.3390/cancers14102536
Kim HY, Huang BX, Spector AA (2014) Phosphatidylserine in the brain: metabolism and function. Prog Lipid Res 56:1–18. https://doi.org/10.1016/j.plipres.2014.06.002
Kim S, Park SY, Kim SY, Bae DJ, Pyo JH, Hong M, Kim IS (2012) Cross talk between engulfment receptors stabilin-2 and integrin alphavbeta5 orchestrates engulfment of phosphatidylserine-exposed erythrocytes. Mol Cell Biol 32:2698–2708. https://doi.org/10.1128/MCB.06743-11
Kimura AK, Kim HY (2013) Phosphatidylserine synthase 2: high efficiency for synthesizing phosphatidylserine containing docosahexaenoic acid. J Lipid Res 54:214–222. https://doi.org/10.1194/jlr.M031989
Kimura AK, Kimura T (2021) Phosphatidylserine biosynthesis pathways in lipid homeostasis: toward resolution of the pending central issue for decades. FASEB J 35:e21177. https://doi.org/10.1096/fj.202001802R
Kornberg RD, McConnell HM (1971) Lateral diffusion of phospholipids in a vesicle membrane. Proc Natl Acad Sci U S A 68:2564–2568. https://doi.org/10.1073/pnas.68.10.2564
Krijgsman D, Hokland M, Kuppen PJK (2018) The role of natural killer T cells in cancer-a phenotypical and functional approach. Front Immunol 9:367. https://doi.org/10.3389/fimmu.2018.00367
Krummel MF, Allison JP (1995) CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med 182:459–465. https://doi.org/10.1084/jem.182.2.459
Lala T, Doan JK, Takatsu H, Hartzell HC, Shin HW, Hall RA (2022) Phosphatidylserine exposure modulates adhesion GPCR BAI1 (ADGRB1) signaling activity. J BiolChem 298. ARTN 102685. https://doi.org/10.1016/j.jbc.2022.102685
Lameris R, Shahine A, Veth M, Westerman B, Godfrey DI, Lutje Hulsik D, Brouwer P, Rossjohn J, de Gruijl TD, van der Vliet HJ (2023) Enhanced CD1d phosphatidylserine presentation using a single-domain antibody promotes immunomodulatory CD1d-TIM-3 interactions. J Immunother Cancer 11. https://doi.org/10.1136/jitc-2023-007631
Larkin J, Hodi FS, Wolchok JD (2015) Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med 373:1270–1271. https://doi.org/10.1056/NEJMc1509660
Leach DR, Krummel MF, Allison JP (1996) Enhancement of antitumor immunity by CTLA-4 blockade. Science 271:1734–1736. https://doi.org/10.1126/science.271.5256.1734
Lee-Sherick AB, Jacobsen KM, Henry CJ, Huey MG, Parker RE, Page LS, Hill AA, Wang X, Frye SV, Earp HS, Jordan CT, DeRyckere D, Graham DK (2018) MERTK inhibition alters the PD-1 axis and promotes anti-leukemia immunity. JCI Insight 3. https://doi.org/10.1172/jci.insight.97941
Lee J, Shergill A, Park H, Lee KW, Arkenau HT, Kang YK, Olowokure OO, Kim JG, Oh SY, Oh DY, Costin D, Pippas AW, Chen JS, Chaney MF, Koustenis A, Youssoufian H, Makris L, Culm K, Mockbee CM, Chau I (2023) Phase 2 study of bavituximab (bavi), a first-in-class antibody targeting phosphatidylserine (PS), plus pembrolizumab (P) in advanced gastric or gastroesophageal junction (GEJ) cancer. J Clin Oncol 41(16):e16023. https://doi.org/10.1200/JCO.2023.41.16_suppl.e16023
Lee SE, Lee CM, Won JE, Jang GY, Lee JH, Park SH, Kang TH, Han HD, Park YM (2022) Enhancement of anticancer immunity by immunomodulation of apoptotic tumor cells using Annexin A5 protein-labeled nanocarrier system. Biomaterials 288:121677. https://doi.org/10.1016/j.biomaterials.2022.121677
Letai A (2017) Functional precision cancer medicine-moving beyond pure genomics. Nat Med 23:1028–1035. https://doi.org/10.1038/nm.4389
Lew ED, Oh J, Burrola PG, Lax I, Zagorska A, Traves PG, Schlessinger J, Lemke G (2014) Differential TAM receptor-ligand-phospholipid interactions delimit differential TAM bioactivities. eLife 3. https://doi.org/10.7554/eLife.03385
Lim WA, June CH (2017) The principles of engineering immune cells to treat cancer. Cell 168:724–740. https://doi.org/10.1016/j.cell.2017.01.016
Lima LG, Chammas R, Monteiro RQ, Moreira ME, Barcinski MA (2009) Tumor-derived microvesicles modulate the establishment of metastatic melanoma in a phosphatidylserine-dependent manner. Cancer Lett 283:168–175. https://doi.org/10.1016/j.canlet.2009.03.041
Liu W, Bai F, Wang H, Liang Y, Du X, Liu C, Cai D, Peng J, Zhong G, Liang X, Ma C, Gao L (2019) Tim-4 inhibits NLRP3 inflammasome via the LKB1/AMPKalpha pathway in macrophages. J Immunol 203:990–1000. https://doi.org/10.4049/jimmunol.1900117
Luster TA, He J, Huang XM, Maiti SN, Schroit AJ, de Groot PG, Thorpe PE (2006) Plasma protein β-2-glycoprotein 1 mediates interaction between the anti-tumor monoclonal antibody 3G4 and anionic phospholipids on endothelial cells. J Biol Chem 281:29863–29871. https://doi.org/10.1074/jbc.M605252200
Ma X, Li X, Wang W, Zhang M, Yang B, Miao Z (2022) Phosphatidylserine, inflammation, and central nervous system diseases. Front Aging Neurosci 14:975176. https://doi.org/10.3389/fnagi.2022.975176
Martin S, Pombo I, Poncet P, David B, Arock M, Blank U (2000) Immunologic stimulation of mast cells leads to the reversible exposure of phosphatidylserine in the absence of apoptosis. Int Arch Allergy Immunol 123:249–258. https://doi.org/10.1159/000024451
Mehrotra P, Ravichandran KS (2022) Drugging the efferocytosis process: concepts and opportunities. Nat Rev Drug Discov 21:601–620. https://doi.org/10.1038/s41573-022-00470-y
Meyer AS, Zweemer AJ, Lauffenburger DA (2015) The AXL receptor is a sensor of ligand spatial heterogeneity. Cell Syst 1:25–36. https://doi.org/10.1016/j.cels.2015.06.002
Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel RA, Freeman GJ, Kuchroo VK (2002) Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415:536–541. https://doi.org/10.1038/415536a
Moody DB (2006) The surprising diversity of lipid antigens for CD1-restricted T cells. Adv Immunol 89:87–139. https://doi.org/10.1016/S0065-2776(05)89003-0
Murakami Y, Tian L, Voss OH, Margulies DH, Krzewski K, Coligan JE (2014) CD300b regulates the phagocytosis of apoptotic cells via phosphatidylserine recognition. Cell Death Differ 21:1746–1757. https://doi.org/10.1038/cdd.2014.86
Myers KV, Amend SR, Pienta KJ (2019) Targeting Tyro3, Axl and MerTK (TAM receptors): implications for macrophages in the tumor microenvironment. Mol Cancer 18:94. https://doi.org/10.1186/s12943-019-1022-2
N’Guessan KF, Patel PH, Qi X (2020) SapC-DOPS - a phosphatidylserine-targeted nanovesicle for selective cancer therapy. Cell Commun Signal 18:6. https://doi.org/10.1186/s12964-019-0476-6
Nagata S, Sakuragi T, Segawa K (2020) Flippase and scramblase for phosphatidylserine exposure. Curr Opin Immunol 62:31–38. https://doi.org/10.1016/j.coi.2019.11.009
Nguyen LT, Ohashi PS (2015) Clinical blockade of PD1 and LAG3–potential mechanisms of action. Nat Rev Immunol 15:45–56. https://doi.org/10.1038/nri3790
Nishimura H, Minato N, Nakano T, Honjo T (1998) Immunological studies on PD-1 deficient mice: implication of PD-1 as a negative regulator for B cell responses. Int Immunol 10:1563–1572. https://doi.org/10.1093/intimm/10.10.1563
Olesch C, Bruene B, Weigert A (2022) Keep a little fire burning-the delicate balance of targeting sphingosine-1-phosphate in cancer immunity. Int J MolSci 23. ARTN 1289. https://doi.org/10.3390/ijms23031289
Ory S, Ceridono M, Momboisse F, Houy S, Chasserot-Golaz S, Heintz D, Calco V, Haeberle AM, Espinoza FA, Sims PJ, Bailly Y, Bader MF, Gasman S (2013) Phospholipid scramblase-1-induced lipid reorganization regulates compensatory endocytosis in neuroendocrine cells. J Neurosci 33:3545–3556. https://doi.org/10.1523/JNEUROSCI.3654-12.2013
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. https://doi.org/10.1038/nrc3239nrc3239[pii]
Park SY, Kim IS (2017) Engulfment signals and the phagocytic machinery for apoptotic cell clearance. Exp Mol Med 49:e331. https://doi.org/10.1038/emm.2017.52
Park SY, Kim IS (2019) Stabilin receptors: role as phosphatidylserine receptors. Biomolecules 9. https://doi.org/10.3390/biom9080387
Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, Baker J, Jeffery LE, Kaur S, Briggs Z, Hou TZ, Futter CE, Anderson G, Walker LS, Sansom DM (2011) Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science 332:600–603
Ran S, He J, Huang XM, Soares M, Scothorn D, Thorpe PE (2005) Antitumor effects of a monoclonal antibody that binds anionic phospholipids on the surface of tumor blood vessels in mice. Clin Cancer Res 11:1551–1562. https://doi.org/10.1158/1078-0432.Ccr-04-1645
Ravichandran KS (2011) Beginnings of a good apoptotic meal: the find-me and eat-me signaling pathways. Immunity 35:445–455. https://doi.org/10.1016/j.immuni.2011.09.004
Rivel T, Ramseyer C, Yesylevskyy S (2019) The asymmetry of plasma membranes and their cholesterol content influence the uptake of cisplatin. Sci Rep 9:5627. https://doi.org/10.1038/s41598-019-41903-w
Rixe O, Morris JC, Puduvalli VK, Villano JL, Wise-Draper TM, Wesolowski R, Yilmaz E, Lanverman SM, Karivedu V, Patterson MT, Qi XY (2019) Safety and pharmacokinetics of BXQ-350 in a phase 1a and 1b trial of solid tumors and high-grade glioma. J ClinOncol 37. https://doi.org/10.1200/JCO.2019.37.15_suppl.e13531
Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, Daud A, Carlino MS, McNeil C, Lotem M, Larkin J, Lorigan P, Neyns B, Blank CU, Hamid O, Mateus C, Shapira-Frommer R, Kosh M, Zhou H, Ibrahim N, Ebbinghaus S, Ribas A, Investigators K- (2015) Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med 372:2521–2532. https://doi.org/10.1056/NEJMoa1503093
Rothlin CV, Carrera-Silva EA, Bosurgi L, Ghosh S (2015) TAM receptor signaling in immune homeostasis. Annu Rev Immunol 33:355–391. https://doi.org/10.1146/annurev-immunol-032414-112103
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331:1565–1570
Sekar D, Dillmann C, Sirait-Fischer E, Fink AF, Zivkovic A, Baum N, Strack E, Klatt S, Zukunft S, Wallner S, Descot A, Olesch C, da Silva P, von Knethen A, Schmid T, Grosch S, Savai R, Ferreiros N, Fleming I, Ghosh S, Rothlin CV, Stark H, Medyouf H, Brune B, Weigert A (2022) Phosphatidylserine synthase PTDSS1 shapes the tumor lipidome to maintain tumor-promoting inflammation. Cancer Res 82:1617–1632. https://doi.org/10.1158/0008-5472.CAN-20-3870
Sharma P, Allison JP (2015) The future of immune checkpoint therapy. Science 348:56–61. https://doi.org/10.1126/science.aaa8172
Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A (2017) Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 168:707–723. https://doi.org/10.1016/j.cell.2017.01.017
Shin HW, Takatsu H (2020) Phosphatidylserine exposure in living cells. Crit Rev Biochem Mol Biol 55:166–178. https://doi.org/10.1080/10409238.2020.1758624
Shlomovitz I, Speir M, Gerlic M (2019) Flip** the dogma - phosphatidylserine in non-apoptotic cell death. Cell Commun Signal 17:139. https://doi.org/10.1186/s12964-019-0437-0
Simhadri VR, Andersen JF, Calvo E, Choi SC, Coligan JE, Borrego F (2012) Human CD300a binds to phosphatidylethanolamine and phosphatidylserine, and modulates the phagocytosis of dead cells. Blood 119:2799–2809. https://doi.org/10.1182/blood-2011-08-372425
Sordi R, Bet AC, Della Justina AM, Ramos GC, Assreuy J (2020) The apoptosis clearance signal phosphatidylserine inhibits leukocyte migration and promotes inflammation resolution in vivo. Eur J Pharmacol 173095. https://doi.org/10.1016/j.ejphar.2020.173095
Stanford JC, Young C, Hicks D, Owens P, Williams A, Vaught DB, Morrison MM, Lim J, Williams M, Brantley-Sieders DM, Balko JM, Tonetti D, Earp HS 3rd, Cook RS (2014) Efferocytosis produces a prometastatic landscape during postpartum mammary gland involution. J Clin Invest 124:4737–4752. https://doi.org/10.1172/JCI76375
Sturbois-Balcerzak B, Stone SJ, Sreenivas A, Vance JE (2001) Structure and expression of the murine phosphatidylserine synthase-1 gene. J Biol Chem 276:8205–8212. https://doi.org/10.1074/jbc.M009776200
Tang T, Huang X, Zhang G, Hong Z, Bai X, Liang T (2021) Advantages of targeting the tumor immune microenvironment over blocking immune checkpoint in cancer immunotherapy. Signal Transduct Target Ther 6:72. https://doi.org/10.1038/s41392-020-00449-4
Terabe M, Berzofsky JA (2008) The role of NKT cells in tumor immunity. Adv Cancer Res 101:277–348
Tirado-Gonzalez I, Descot A, Soetopo D, Nevmerzhitskaya A, Schaffer A, Kur IM, Czlonka E, Wachtel C, Tsoukala I, Muller L, Schafer AL, Weitmann M, Dinse P, Alberto E, Buck MC, Landry JJ, Baying B, Slotta-Huspenina J, Roesler J, Harter PN, Kubasch AS, Meinel J, Elwakeel E, Strack E, Quang CT, Abdel-Wahab O, Schmitz M, Weigert A, Schmid T, Platzbecker U, Benes V, Ghysdael J, Bonig H, Gotze KS, Rothlin CV, Ghosh S, Medyouf H (2021) AXL inhibition in macrophages stimulates host-versus-leukemia immunity and eradicates naive and treatment-resistant leukemia. Cancer Discov 11:2924–2943. https://doi.org/10.1158/2159-8290.CD-20-1378
Tsou WI, Nguyen KQ, Calarese DA, Garforth SJ, Antes AL, Smirnov SV, Almo SC, Birge RB, Kotenko SV (2014) Receptor tyrosine kinases, TYRO3, AXL, and MER, demonstrate distinct patterns and complex regulation of ligand-induced activation. J Biol Chem 289:25750–25763. https://doi.org/10.1074/jbc.M114.569020
Tsuji T, Cheng J, Tatematsu T, Ebata A, Kamikawa H, Fujita A, Gyobu S, Segawa K, Arai H, Taguchi T, Nagata S, Fujimoto T (2019) Predominant localization of phosphatidylserine at the cytoplasmic leaflet of the ER, and its TMEM16K-dependent redistribution. P Natl Acad Sci USA 116:13368–13373. https://doi.org/10.1073/pnas.1822025116
Uhlik M, Pointing D, Iyer S, Ausec L, Stajdohar M, Cvitkovic R, Zganec M, Culm K, Santos VC, Pytowski B, Malafa M, Liu H, Krieg AM, Lee J, Rosengarten R, Benjamin L (2023) Xerna™ TME Panel is a machine learning-based transcriptomic biomarker designed to predict therapeutic response in multiple cancers. Front Oncol 13. ARTN 1158345. https://doi.org/10.3389/fonc.2023.1158345
Umetsu SE, Lee WL, McIntire JJ, Downey L, Sanjanwala B, Akbari O, Berry GJ, Nagumo H, Freeman GJ, Umetsu DT, DeKruyff RH (2005) TIM-1 induces T cell activation and inhibits the development of peripheral tolerance. Nat Immunol 6:447–454. https://doi.org/10.1038/ni1186
Vallabhapurapu SD, Blanco VM, Sulaiman MK, Vallabhapurapu SL, Chu Z, Franco RS, Qi X (2015) Variation in human cancer cell external phosphatidylserine is regulated by flippase activity and intracellular calcium. Oncotarget 6:34375–34388. https://doi.org/10.18632/oncotarget.6045
Van Rhijn I, Godfrey DI, Rossjohn J, Moody DB (2015) Lipid and small-molecule display by CD1 and MR1. Nat Rev Immunol 15:643–654. https://doi.org/10.1038/nri3889
Van Rite BD, Krais JJ, Cherry M, Sikavitsas VI, Kurkjian C, Harrison RG (2013) Antitumor activity of an enzyme prodrug therapy targeted to the breast tumor vasculature. Cancer Invest 31:505–510. https://doi.org/10.3109/07357907.2013.840383
Vance JE (2014) MAM (mitochondria-associated membranes) in mammalian cells: lipids and beyond. Biochim Biophys Acta 1841:595–609. https://doi.org/10.1016/j.bbalip.2013.11.014
Vance JE (2018) Historical perspective: phosphatidylserine and phosphatidylethanolamine from the 1800s to the present. J Lipid Res 59:923–944. https://doi.org/10.1194/jlr.R084004
Vance JE, Steenbergen R (2005) Metabolism and functions of phosphatidylserine. Prog Lipid Res 44:207–234. https://doi.org/10.1016/j.plipres.2005.05.001
Waldman AD, Fritz JM, Lenardo MJ (2020) A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol 20:651–668. https://doi.org/10.1038/s41577-020-0306-5
Wei SC, Duffy CR, Allison JP (2018) Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov 8:1069–1086. https://doi.org/10.1158/2159-8290.CD-18-0367
Weigert A, Mora J, Sekar D, Syed S, Brune B (2016) Killing is not enough: how apoptosis hijacks tumor-associated macrophages to promote cancer progression. Adv Exp Med Biol 930:205–239. https://doi.org/10.1007/978-3-319-39406-0_9
Werfel TA, Cook RS (2018) Efferocytosis in the tumor microenvironment. Semin Immunopathol 40:545–554. https://doi.org/10.1007/s00281-018-0698-5
Willems JJ, Arnold BP, Gregory CD (2014) Sinister self-sacrifice: the contribution of apoptosis to malignancy. Front Immunol 5:299. https://doi.org/10.3389/fimmu.2014.00299
Wolf Y, Anderson AC, Kuchroo VK (2020) TIM3 comes of age as an inhibitory receptor. Nat Rev Immunol 20:173–185. https://doi.org/10.1038/s41577-019-0224-6
Wong K, Valdez PA, Tan C, Yeh S, Hongo JA, Ouyang W (2010) Phosphatidylserine receptor Tim-4 is essential for the maintenance of the homeostatic state of resident peritoneal macrophages. P Natl Acad Sci USA 107:8712–8717. https://doi.org/10.1073/pnas.0910929107
Xu C, Gagnon E, Call ME, Schnell JR, Schwieters CD, Carman CV, Chou JJ, Wucherpfennig KW (2008) Regulation of T cell receptor activation by dynamic membrane binding of the CD3epsilon cytoplasmic tyrosine-based motif. Cell 135:702–713. https://doi.org/10.1016/j.cell.2008.09.044
Yin Y, Huang X, Lynn KD, Thorpe PE (2013) Phosphatidylserine-targeting antibody induces M1 macrophage polarization and promotes myeloid-derived suppressor cell differentiation. Cancer Immunol Res 1:256–268. https://doi.org/10.1158/2326-6066.CIR-13-0073
Yoshihama Y, Namiki H, Kato T, Shimazaki N, Takaishi S, Kadoshima-Yamaoka K, Yukinaga H, Maeda N, Shibutani T, Fujimoto K, Hirasawa M, Goto H, Wada N, Tsutsumi S, Hirota Y, Ishikawa T, Yamamoto S (2022) Potent and selective PTDSS1 inhibitors induce collateral lethality in cancers with PTDSS2 deletion. Cancer Res 82:4031–4043. https://doi.org/10.1158/0008-5472.CAN-22-1006
You X, Koop K, Weigert A (2023) Heterogeneity of tertiary lymphoid structures in cancer. Front Immunol 14:1286850. https://doi.org/10.3389/fimmu.2023.1286850
Younan P, Iampietro M, Nishida A, Ramanathan P, Santos RI, Dutta M, Lubaki NM, Koup RA, Katze MG, Bukreyev A (2017) Ebola virus binding to Tim-1 on T lymphocytes induces a cytokine storm. mBio 8. https://doi.org/10.1128/mBio.00845-17
Zachowski A (1993) Phospholipids in animal eukaryotic membranes: transverse asymmetry and movement. Biochem J 294(Pt 1):1–14. https://doi.org/10.1042/bj2940001
Zagorska A, Traves PG, Jimenez-Garcia L, Strickland JD, Oh J, Tapia FJ, Mayoral R, Burrola P, Copple BL, Lemke G (2020) Differential regulation of hepatic physiology and injury by the TAM receptors Axl and Mer. Life Sci Alliance 3. https://doi.org/10.26508/lsa.202000694
Zaitseva E, Zaitsev E, Melikov K, Arakelyan A, Marin M, Villasmil R, Margolis LB, Melikyan GB, Chernomordik LV (2017) Fusion stage of HIV-1 entry depends on virus-induced cell surface exposure of phosphatidylserine. Cell Host Microbe 22(99–110):e117. https://doi.org/10.1016/j.chom.2017.06.012
Zalba S, Ten Hagen TL (2017) Cell membrane modulation as adjuvant in cancer therapy. Cancer Treat Rev 52:48–57. https://doi.org/10.1016/j.ctrv.2016.10.008
Zheng X, Turkowski K, Mora J, Brune B, Seeger W, Weigert A, Savai R (2017) Redirecting tumor-associated macrophages to become tumoricidal effectors as a novel strategy for cancer therapy. Oncotarget 8:48436–48452. https://doi.org/10.18632/oncotarget.17061
Zhou Y, Fei M, Zhang G, Liang WC, Lin W, Wu Y, Piskol R, Ridgway J, McNamara E, Huang H, Zhang J, Oh J, Patel JM, Jakubiak D, Lau J, Blackwood B, Bravo DD, Shi Y, Wang J, Hu HM, Lee WP, Jesudason R, Sangaraju D, Modrusan Z, Anderson KR, Warming S, Roose-Girma M, Yan M (2020) Blockade of the phagocytic receptor MerTK on tumor-associated macrophages enhances P2X7R-dependent STING activation by tumor-derived cGAMP. Immunity 52(357–373):e359. https://doi.org/10.1016/j.immuni.2020.01.014
Acknowledgements
We apologize to researchers whose important primary contributions to the matter were cited indirectly by referring to current reviews.
Funding
Open Access funding enabled and organized by Projekt DEAL. AW is supported by Deutsche Krebshilfe (70114051), Deutsche Forschungsgemeinschaft (SFB 1039, TP B06; GRK 2336, TP1), the LOEWE Center Frankfurt Cancer Institute (FCI), and the EnABLE Cluster funded by the Hessen State Ministry for Higher Education, Research and the Arts.
Author information
Authors and Affiliations
Contributions
IK and AW wrote the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the special issue on Signalling by Fatty Acid Derivatives and Sphingolipids in Health and Disease in Pflügers.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kur, IM., Weigert, A. Phosphatidylserine externalization as immune checkpoint in cancer. Pflugers Arch - Eur J Physiol (2024). https://doi.org/10.1007/s00424-024-02948-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00424-024-02948-7