Abstract
Background
Natalizumab (NTZ) is an effective treatment for relapsing–remitting multiple sclerosis (RRMS). However, patients and physicians may consider discontinuing NTZ therapy due to safety or efficacy issues. The aim of our study was to evaluate the NTZ discontinuation rate and reasons of discontinuation in a large Italian population of RRMS patients.
Materials and methods
The data were extracted from the Italian MS registry in May 2018 and were collected from 51,845 patients in 69 Italian multiple sclerosis centers. MS patients with at least one NTZ infusion in the period between June 1st 2012 to May 15th 2018 were included. Discontinuation rates at each time point were calculated. Reasons for NTZ discontinuation were classified as “lack of efficacy”, “progressive multifocal leukoencephalopathy (PML) risk” or “other”.
Results
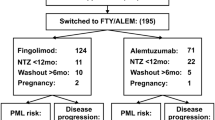
Out of 51,845, 5151 patients, 3019 (58.6%) females, with a mean age of 43.6 ± 10.1 years (median 40), were analyzed. Out of 2037 (39.5%) who discontinued NTZ, a significantly higher percentage suspended NTZ because of PML risk compared to lack of efficacy [1682 (32.7% of 5151) vs 221 (4.3%), p < 0.001]; other reasons were identified for 99 (1.9%) patients. Patients discontinuing treatment were older, had longer disease duration and worse EDSS at the time of NTZ initiation and at last follow-up on NTZ treatment. The JCV index and EDSS at baseline were predictors for stop** therapy (HR 2.94, 95% CI 1.22–4.75; p = 0.02; HR 1.36, 95% CI 1.18–5.41; p = 0.04).
Conclusions
Roughly 60% of MS patients stayed on NTZ treatment during the observation period. For those patients in whom NTZ discontinuation was required, it was mainly due to PML concerns.


Similar content being viewed by others
Availability of data and material
Dataset is available under reasonable request to the corresponding author.
References
Polman CH, O’Connor PW, Havrdova E, Hutchinson M, Kappos L, Miller DH, Phillips JT, Lublin FD, Giovannoni G, Wajgt A, Toal M, Lynn F, Panzara MA, Sandrock AW, Investigators A (2006) A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 354(9):899–910. https://doi.org/10.1056/NEJMoa044397
Radue EW, Stuart WH, Calabresi PA, Confavreux C, Galetta SL, Rudick RA, Lublin FD, Weinstock-Guttman B, Wynn DR, Fisher E, Papadopoulou A, Lynn F, Panzara MA, Sandrock AW, Investigators S (2010) Natalizumab plus interferon beta-1a reduces lesion formation in relapsing multiple sclerosis. J Neurol Sci 292(1–2):28–35. https://doi.org/10.1016/j.jns.2010.02.012
Fernandez O, Oreja-Guevara C, Arroyo R, Izquierdo G, Perez JL, Montalban X (2012) Natalizumab treatment of multiple sclerosis in Spain: results of an extensive observational study. J Neurol 259(9):1814–1823. https://doi.org/10.1007/s00415-012-6414-9
Prosperini L, Gianni C, Barletta V, Mancinelli C, Fubelli F, Borriello G, Pozzilli C (2012) Predictors of freedom from disease activity in natalizumab treated-patients with multiple sclerosis. J Neurol Sci 323(1–2):104–112. https://doi.org/10.1016/j.jns.2012.08.027
Baldwin KJ, Hogg JP (2013) Progressive multifocal leukoencephalopathy in patients with multiple sclerosis. Curr Opin Neurol 26(3):318–323. https://doi.org/10.1097/WCO.0b013e328360279f
Vukusic S, Rollot F, Casey R, Pique J, Marignier R, Mathey G, Edan G, Brassat D, Ruet A, De Seze J, Maillart E, Zephir H, Labauge P, Derache N, Lebrun-Frenay C, Moreau T, Wiertlewski S, Berger E, Moisset X, Rico-Lamy A, Stankoff B, Bensa C, Thouvenot E, Heinzlef O, Al-Khedr A, Bourre B, Vaillant M, Cabre P, Montcuquet A, Wahab A, Camdessanche JP, Tourbah A, Guennoc AM, Hankiewicz K, Patry I, Nifle C, Maubeuge N, Labeyrie C, Vermersch P, Laplaud DA, Investigators O (2019) Progressive multifocal leukoencephalopathy incidence and risk stratification among natalizumab users in France. JAMA Neurol. https://doi.org/10.1001/jamaneurol.2019.2670
D’Amico E, Zanghi A, Leone C, Tumani H, Patti F (2016) Treatment-related progressive multifocal leukoencephalopathy in multiple sclerosis: a comprehensive review of current evidence and future needs. Drug Saf 39(12):1163–1174. https://doi.org/10.1007/s40264-016-0461-6
McGuigan C, Craner M, Guadagno J, Kapoor R, Mazibrada G, Molyneux P, Nicholas R, Palace J, Pearson OR, Rog D, Young CA (2016) Stratification and monitoring of natalizumab-associated progressive multifocal leukoencephalopathy risk: recommendations from an expert group. J Neurol Neurosurg Psychiatry 87(2):117–125. https://doi.org/10.1136/jnnp-2015-311100
Fox RJ, Cree BA, De Seze J, Gold R, Hartung HP, Jeffery D, Kappos L, Kaufman M, Montalban X, Weinstock-Guttman B, Anderson B, Natarajan A, Ticho B, Duda P, Restore (2014) MS disease activity in RESTORE: a randomized 24 week natalizumab treatment interruption study. Neurology 82(17):1491–1498. https://doi.org/10.1212/WNL.0000000000000355
Rammohan KW, Ortega MR, Delgado SR, Tornes L (2011) Disease activity return during natalizumab treatment interruption in patients with multiple sclerosis. Neurology 77(21):1930. https://doi.org/10.1212/WNL.0b013e31823c1200 (discussion 1930–1931)
Berger JR, Centonze D, Comi G, Confavreux C, Cutter G, Giovannoni G, Gold R, Hartung HP, Lublin F, Miravalle A, Montalban X, O’Connor P, Olsson T, Polman CH, Stuve O, Wolinsky JS, Ziemssen T (2010) Considerations on discontinuing natalizumab for the treatment of multiple sclerosis. Ann Neurol 68(3):409–411. https://doi.org/10.1002/ana.22083
Evdoshenko E, Stepanova A, Shumilina M, Davydovskaya M, Khachanova N, Neofidov N, Kalinin I, Popova E, Dubchenko E, Pozhidaeva N, Volkov A, Sivertseva S, Prilenskaya A, Malkova N, Korobko D, Vergunova I, Shchur S, Makshakov G (2019) Real-world study of efficacy, risk management and reasons for discontinuation of natalizumab for treatment of multiple sclerosis in Russia. PLoS ONE 14(5):e0217303. https://doi.org/10.1371/journal.pone.0217303
Patti F, Leone C, Zappia M (2015) Clinical and radiologic rebound after discontinuation of natalizumab therapy in a highly active multiple sclerosis patient was not halted by dimethyl-fumarate: a case report. BMC Neurol 15:252. https://doi.org/10.1186/s12883-015-0512-0
Vellinga MM, Castelijns JA, Barkhof F, Uitdehaag BM, Polman CH (2008) Postwithdrawal rebound increase in T2 lesional activity in natalizumab-treated MS patients. Neurology 70(13 Pt 2):1150–1151. https://doi.org/10.1212/01.wnl.0000265393.03231.e5
Miravalle A, Jensen R, Kinkel RP (2011) Immune reconstitution inflammatory syndrome in patients with multiple sclerosis following cessation of natalizumab therapy. Arch Neurol 68(2):186–191. https://doi.org/10.1001/archneurol.2010.257
Borriello G, Prosperini L, Marinelli F, Fubelli F, Pozzilli C (2011) Observations during an elective interruption of natalizumab treatment: a post-marketing study. Mult Scler 17(3):372–375. https://doi.org/10.1177/1352458510392098
Havla J, Gerdes LA, Meinl I, Krumbholz M, Faber H, Weber F, Pellkofer HL, Hohlfeld R, Kumpfel T (2011) De-escalation from natalizumab in multiple sclerosis: recurrence of disease activity despite switching to glatiramer acetate. J Neurol 258(9):1665–1669. https://doi.org/10.1007/s00415-011-5996-y
West TW, Cree BA (2010) Natalizumab dosage suspension: are we hel** or hurting? Ann Neurol 68(3):395–399. https://doi.org/10.1002/ana.22163
Montalban X, Gold R, Thompson AJ, Otero-Romero S, Amato MP, Chandraratna D, Clanet M, Comi G, Derfuss T, Fazekas F, Hartung HP, Havrdova E, Hemmer B, Kappos L, Liblau R, Lubetzki C, Marcus E, Miller DH, Olsson T, Pilling S, Selmaj K, Siva A, Sorensen PS, Sormani MP, Thalheim C, Wiendl H, Zipp F (2018) ECTRIMS/EAN guideline on the pharmacological treatment of people with multiple sclerosis. Eur J Neurol 25(2):215–237. https://doi.org/10.1111/ene.13536
Fine AJ, Sorbello A, Kortepeter C, Scarazzini L (2014) Progressive multifocal leukoencephalopathy after natalizumab discontinuation. Ann Neurol 75(1):108–115. https://doi.org/10.1002/ana.24051
Trojano M, Bergamaschi R, Amato MP, Comi G, Ghezzi A, Lepore V, Marrosu MG, Mosconi P, Patti F, Ponzio M, Zaratin P, Battaglia MA, Italian Multiple Sclerosis Register Centers G (2019) The Italian multiple sclerosis register. Neurol Sci 40(1):155–165. https://doi.org/10.1007/s10072-018-3610-0
Prosperini L, de Rossi N, Scarpazza C, Moiola L, Cosottini M, Gerevini S, Capra R, Italian PMLsg (2016) Natalizumab-related progressive multifocal leukoencephalopathy in multiple sclerosis: findings from an Italian independent registry. PLoS ONE 11(12):e0168376. https://doi.org/10.1371/journal.pone.0168376
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O’Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Wolinsky JS, Narayana PA, O’Connor P, Coyle PK, Ford C, Johnson K, Miller A, Pardo L, Kadosh S, Ladkani D, Group PRTS (2007) Glatiramer acetate in primary progressive multiple sclerosis: results of a multinational, multicenter, double-blind, placebo-controlled trial. Ann Neurol 61(1):14–24. https://doi.org/10.1002/ana.21079
Butzkueven H, Kappos L, Pellegrini F, Trojano M, Wiendl H, Patel RN, Zhang A, Hotermans C, Belachew S, Investigators TOP (2014) Efficacy and safety of natalizumab in multiple sclerosis: interim observational programme results. J Neurol Neurosurg Psychiatry 85(11):1190–1197. https://doi.org/10.1136/jnnp-2013-306936
Butzkueven H, Kappos L, Wiendl H, Trojano M, Spelman T, Chang I, Kasliwal R, Jaitly S, Campbell N, Ho PR, Licata S, Tysabri Observational Program I (2020) Long-term safety and effectiveness of natalizumab treatment in clinical practice: 10 years of real-world data from the Tysabri observational program (TOP). J Neurol Neurosurg Psychiatry. https://doi.org/10.1136/jnnp-2019-322326
Trojano M, Butzkueven H, Kappos L, Wiendl H, Spelman T, Pellegrini F, Chen Y, Dong Q, Koendgen H, Belachew S, Tysabri Observational Program I (2018) Natalizumab treatment shows low cumulative probabilities of confirmed disability worsening to EDSS milestones in the long-term setting. Mult Scler Relat Disord 24:11–19. https://doi.org/10.1016/j.msard.2018.04.020
Kramer J, Tenberge JG, Kleiter I, Gaissmaier W, Ruck T, Heesen C, Meuth SG (2017) Is the risk of progressive multifocal leukoencephalopathy the real reason for natalizumab discontinuation in patients with multiple sclerosis? PLoS ONE 12(4):e0174858. https://doi.org/10.1371/journal.pone.0174858
Putzki N, Yaldizli O, Buhler R, Schwegler G, Curtius D, Tettenborn B (2010) Natalizumab reduces clinical and MRI activity in multiple sclerosis patients with high disease activity: results from a multicenter study in Switzerland. Eur Neurol 63(2):101–106. https://doi.org/10.1159/000276400
Oturai AB, Koch-Henriksen N, Petersen T, Jensen PE, Sellebjerg F, Sorensen PS (2009) Efficacy of natalizumab in multiple sclerosis patients with high disease activity: a Danish nationwide study. Eur J Neurol 16(3):420–423. https://doi.org/10.1111/j.1468-1331.2008.02517.x
Sangalli F, Moiola L, Bucello S, Annovazzi P, Rizzo A, Radaelli M, Vitello G, Grimaldi LM, Ghezzi A, Martinelli V, Comi G (2011) Efficacy and tolerability of natalizumab in relapsing-remitting multiple sclerosis patients: a post-marketing observational study. Neurol Sci 31(Suppl 3):299–302. https://doi.org/10.1007/s10072-010-0344-z
Outteryck O, Ongagna JC, Duhamel A, Zephir H, Collongues N, Lacour A, Fleury MC, Berteloot AS, Blanc F, Giroux M, Vermersch P, de Seze J (2012) Anti-JCV antibody prevalence in a French cohort of MS patients under natalizumab therapy. J Neurol 259(11):2293–2298. https://doi.org/10.1007/s00415-012-6487-5
Prosperini L, Borriello G, Fubelli F, Marinelli F, Pozzilli C (2011) Natalizumab treatment in multiple sclerosis: the experience of S. Andrea MS Centre in Rome. Neurol Sci 31(Suppl 3):303–307. https://doi.org/10.1007/s10072-010-0348-8
Mancardi GL, Tedeschi G, Amato MP, D’Alessandro R, Drago F, Milanese C, Popoli P, Rossi P, Savettieri G, Tola MR, Comi G, Pozzilli C, Bertolotto A, Marrosu MG, Grimaldi LM, Laroni A, Vanacore N, Covezzoli A, De Rosa M, Piccinni C, Montanaro N, Periotto L, Iommelli R, Tomino C, Provinciali L (2011) Three years of experience: the Italian registry and safety data update. Neurol Sci 31(Suppl 3):295–297. https://doi.org/10.1007/s10072-010-0356-8
Putzki N, Yaldizli O, Maurer M, Cursiefen S, Kuckert S, Klawe C, Maschke M, Tettenborn B, Limmroth V (2010) Efficacy of natalizumab in second line therapy of relapsing-remitting multiple sclerosis: results from a multi-center study in German speaking countries. Eur J Neurol 17(1):31–37. https://doi.org/10.1111/j.1468-1331.2009.02728.x
Piehl F, Holmen C, Hillert J, Olsson T (2011) Swedish natalizumab (Tysabri) multiple sclerosis surveillance study. Neurol Sci 31(Suppl 3):289–293. https://doi.org/10.1007/s10072-010-0345-y
Horga A, Castillo J, Rio J, Tintore M, Auger C, Sastre-Garriga J, Edo MC, Perez-Miralles F, Tur C, Nos C, Huerga E, Comabella M, Rovira A, Montalban X (2011) An observational study of the effectiveness and safety of natalizumab in the treatment of multiple sclerosis. Rev Neurol 52(6):321–330
Prosperini L, Annovazzi P, Capobianco M, Capra R, Buttari F, Gasperini C, Galgani S, Solaro C, Centonze D, Bertolotto A, Pozzilli C, Ghezzi A (2015) Natalizumab discontinuation in patients with multiple sclerosis: profiling risk and benefits at therapeutic crossroads. Mult Scler 21(13):1713–1722. https://doi.org/10.1177/1352458515570768
Plavina T, Subramanyam M, Bloomgren G, Richman S, Pace A, Lee S, Schlain B, Campagnolo D, Belachew S, Ticho B (2014) Anti-JC virus antibody levels in serum or plasma further define risk of natalizumab-associated progressive multifocal leukoencephalopathy. Ann Neurol 76(6):802–812. https://doi.org/10.1002/ana.24286
Kartau M, Sipila JO, Auvinen E, Palomaki M, Verkkoniemi-Ahola A (2019) Progressive multifocal leukoencephalopathy: current Insights. Degener Neurol Neuromuscul Dis 9:109–121. https://doi.org/10.2147/DNND.S203405
Sgarlata E, Chisari CG, D’Amico E, Millefiorini E, Patti F (2020) Changes in anti-JCV antibody status in a large population of multiple sclerosis patients treated with natalizumab. CNS Drugs. https://doi.org/10.1007/s40263-020-00716-6
Bozic C, Richman S, Plavina T, Natarajan A, Scanlon JV, Subramanyam M, Sandrock A, Bloomgren G (2011) Anti-John Cunnigham virus antibody prevalence in multiple sclerosis patients: baseline results of STRATIFY-1. Ann Neurol 70(5):742–750. https://doi.org/10.1002/ana.22606
Ho PR, Koendgen H, Campbell N, Haddock B, Richman S, Chang I (2017) Risk of natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: a retrospective analysis of data from four clinical studies. Lancet Neurol 16(11):925–933. https://doi.org/10.1016/S1474-4422(17)30282-X
Sgarlata E, Chisari CG, D’Amico E, Millefiorini E, Patti F (2020) Changes in Anti-JCV antibody status in a large population of multiple sclerosis patients treated with natalizumab. CNS Drugs 34(5):535–543. https://doi.org/10.1007/s40263-020-00716-6
Conway DS, Hersh CM, Harris HC, Hua LH (2020) Duration of natalizumab therapy and reasons for discontinuation in a multiple sclerosis population. Mult Scler J Exp Transl Clin 6(1):2055217320902488. https://doi.org/10.1177/2055217320902488
Tur C, Tintore M, Vidal-Jordana A, Castillo J, Galan I, Rio J, Arrambide G, Comabella M, Arevalo MJ, Horno R, Vicente MJ, Caminero A, Nos C, Sastre-Garriga J, Montalban X (2012) Natalizumab discontinuation after PML risk stratification: outcome from a shared and informed decision. Mult Scler 18(8):1193–1196. https://doi.org/10.1177/1352458512439238
Tugemann B (2019) The risk of PML from natalizumab. Lancet Neurol 18(3):230. https://doi.org/10.1016/S1474-4422(19)30021-3
Vennegoor A, Rispens T, Strijbis EM, Seewann A, Uitdehaag BM, Balk LJ, Barkhof F, Polman CH, Wolbink G, Killestein J (2013) Clinical relevance of serum natalizumab concentration and anti-natalizumab antibodies in multiple sclerosis. Mult Scler 19(5):593–600. https://doi.org/10.1177/1352458512460604
Acknowledgements
Carlo Avolio, Roberto Balgera, Paola Banfi, Paolo Bellantonio, Placido Bramanti, Lorenzo Capone, Guido Cavalletti, Luca Chiveri, Raffaella Clerici, Marinella Clerico, Francesco Corea, Vincenzo Dattola, Francesca De Robertis, Giancarlo Di Battista, Simonetta Galgani, Maurizia Gatto, Maria Grazia Grasso, Lorenzo Lo Russo, Francesco Ottavio Logullo, Renato Mantegazza, Alessandra Protti, Monica Rezzonico, Mariarosa Rottoli, Marco Salvetti, Elio Scarpini, Leonardo Sinisi, Maddalena Sparaco, Daniele Spitaleri, Tiziana Tassinari, Simone Tonietti, Paola Valentino, Franco Valzania, Simonetta Venturi.
Funding
This study was not funded.
Author information
Authors and Affiliations
Consortia
Contributions
CGC: contributed to the data collection, performed the statistical analysis and interpreted data, drafted the work and revised it critically for important intellectual content, approved the final version of the paper. GC: contributed to the data collection, approved the final version of the paper. MF contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. DP contributed to the design of the work and to the data collection, approved the final version of the paper. PI: contributed to the data collection, approved the final version of the paper. MZ: contributed to the data collection, approved the final version of the paper. VBM: contributed to the data collection, approved the final version of the paper. EC: contributed to the data collection, approved the final version of the paper. GAM: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. LMG: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. MI: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. SB: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. AL contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. Giuseppe Salemi: contributed to the data collection, approved the final version of the paper. GDL: contributed to the data collection, approved the final version of the paper. SC: contributed to the data collection, approved the final version of the paper. AC: contributed to the data collection, approved the final version of the paper. PS: contributed to the data collection, approved the final version of the paper. UA: contributed to the data collection, approved the final version of the paper. GTM: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. CG: contributed to the data collection, approved the final version of the paper. MTF: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. IP: contributed to the data collection, approved the final version of the paper. MPA: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. MR: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. CS: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. GL: contributed to the data collection, approved the final version of the paper. DM: contributed to the data collection, approved the final version of the paper. RB: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. FG: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. ADS: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. AB: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. RT: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. MV: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. PC: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. PB: contributed to the data collection, revised it critically for important intellectual content, approved the final version of the paper. VL: contributed to the study design, approved the final version of the paper. FP: contributed to the data collection, performed the statistical analysis and interpreted data, drafted the work and revised it critically for important intellectual content, approved the final version of the paper.
Corresponding author
Ethics declarations
Conflicts of interest
Clara Grazia Chisari has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Giancarlo Comi has served as a Biogen, Merck, Mylan, Novartis, Roche, Sanofi/Genzyme and Teva Advisory Board Member. He received congress and travel/accommodation expense compensations or speaker honoraria from Biogen, Merck, Mylan, Novartis, Sanofi/Genzyme, Teva and Fondazione Italiana Sclerosi Multipla (FISM). Massimo Filippi is Editor-in-Chief of the Journal of Neurology; received compensation for consulting services and/or speaking activities from Biogen Idec, Merck-Serono, Novartis, Teva Pharmaceutical Industries; and receives research support from Biogen Idec, Merck-Serono, Novartis, Teva Pharmaceutical Industries, Roche, Italian Ministry of Health, Fondazione Italiana Sclerosi Multipla, and ARiSLA (Fondazione Italiana di Ricerca per la SLA). Damiano Paolicelli has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Pietro Iaffaldano has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Mauro Zaffaroni has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Vincenzo Brescia Morra: has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Eleonora Cocco has no conflict of interest. Girolama Alessandra Marfia: has served as an advisory board member and received speaker honoraria, congress, travel and accommodation expense compensations from Merck, Teva, Mylan, Bayer, Novartis, Roche, Almirall, Biogen and Sanofi Genzyme. Luigi Maria Grimaldi: has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Matilde Inglese: received honoraria or consultation fees from Roche, Biogen, Merck-Serono, Novartis, Genzyme and research grants from NIH, NMSS, FISM, and Teva Neuroscience. Simona Bonavita: Speaker and/or advisory board fee, and7or travel grant from Novartis, Teva, Sanofi-Genzyme, Roche, Biogen-Idec, Merck-Serono. Alessandra Lugaresi has served as a Biogen, Merck, Mylan, Novartis, Roche, Sanofi/ Genzyme and Teva Advisory Board Member. She received congress and travel/accommodation expense compensations or speaker honoraria from Biogen, Merck, Mylan, Novartis, Sanofi/Genzyme, Teva and Fondazione Italiana Sclerosi Multipla (FISM). Her institutions received research grants from Novartis. Paola Cavalla has served as an advisory board member for Almirall, Biogen, Merck-Serono, Sanofi-Genzyme, Roche, Teva; she has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Giuseppe Salemi: has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Giovanna De Luca has no conflict of interest. Salvatore Cottone: has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Antonella Conte: declares no conflict of interest. Patrizia Sola declares no conflict of interest. Umberto Aguglia has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Giorgia Teresa Maniscalco has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Claudio Gasperini declares no conflict of interest. Maria Teresa Ferrò: has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Ilaria Pesci declares no conflict of interest. Maria Pia Amato has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Marco Rovaris: received travel grants and fees for consulting and public speaking from Almirall, Biogen, Genzyme-Sanofi, Merck Serono, Mylan and Novartis. Claudio Solaro has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Giacomo Lus declares no conflict of interest. Davide Maimone declares no conflict of interest. Roberto Bergamaschi: declares no conflict of interest. Franco Granella has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Alessia Di Sapio received personal compensation for speaking and consulting by Biogen and Novartis and has been reimbursed by Merck, Biogen, Genzyme and Roche for attending several conferences. Antonio Bertolotto has received grants to attend scientific congresses or speaker honoraria from Biogen, Merck-Serono, Novartis, Roche, Sanofi/Genzyme, Teva. Rocco Totaro: declares no conflict of interest. Marika Vianello declares no conflict of interest. Paolo Bellantonio declares no conflict of interest. Vito Lepore declares no conflict of interest. Francesco Patti has received honoraria for speaking activities by Almirall, Bayer Schering, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi Genzyme, and TEVA; he also served as advisory board member the following companies: Bayer Schering, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi Genzyme, and TEVA; he was also funded by Pfizer and FISM for epidemiological studies; he received grants for congress participation from Almirall, Bayer Schering, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi Genzyme, and TEVA.
Ethical approval
This study protocol was approved by the local Ethical Committee of the University of Catania (Catania 1) and by the Ethical Committee of the participating centers.
Informed consent
Each patient participating to the study signed an Informed Consent specifically designed to participate to the Italian MS Register.
Additional information
The members of the Italian MS Register Study Group are listed in acknowledgements.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chisari, C.G., Comi, G., Filippi, M. et al. PML risk is the main factor driving the choice of discontinuing natalizumab in a large multiple sclerosis population: results from an Italian multicenter retrospective study. J Neurol 269, 933–944 (2022). https://doi.org/10.1007/s00415-021-10676-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10676-6