Abstract
Purpose
Treatment with regorafenib, which inhibits vascular endothelial growth factor (VEGF) receptor, frequently results in hand-foot skin reaction (HFSR), requiring treatment discontinuation or dose reduction. In our prospective study of regorafenib on patients with metastatic colorectal cancer, 17% of patients developed grade 3 HFSR. Herein, we retrospectively examined genetic polymorphisms associated with regorafenib-induced severe HFSR.
Methods
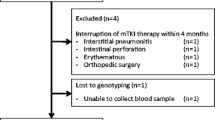
To identify associated polymorphisms, exploratory whole-exome sequencing focusing on factors related to VEGF-mediated signaling pathways was first performed in seven patients each, with grade 3 HFSR and without HFSR. The identified HFSR-associated polymorphisms were analyzed in all the 40 patients.
Results
The genotype frequency of rs3025009 G/A or A/A in the gene encoding VEGF-A (VEGFA) in patients with ≥ grade 2 HFSR was significantly higher than in other patients (P = 0.0257, Pc = 0.0771 [Bonferroni correction]). The frequency of C–C motif of chemokine ligand 4-like 2 (CCL4L2) rs3744596 A/T or T/T in patients with grade 3 HFSR was significantly lower than in others (P = 0.00894, Pc = 0.0268). The combination of the risk genotypes VEGFA rs3025009 G/A or A/A and CCL4L2 rs3744596 A/A was significantly associated with a higher incidence of grade 3 (P = 0.000614, Pc = 0.00246) and a longer median progression-free survival (P = 0.0234) than others.
Conclusions
These VEGF-related polymorphisms were found to be associated with HFSR and the survival benefits of regorafenib treatment.
Trial registration number and date
UMIN000013939, registered on May 12, 2014, when 6 months after the approval by the Institutional Review Board of Showa University.


Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this published article and electronic supplementary material.
Code availability
Not applicable.
References
Roed Skarderud M, Polk A, Kjeldgaard Vistisen K, Larsen FO, Nielsen DL (2018) Efficacy and safety of regorafenib in the treatment of metastatic colorectal cancer: a systematic review. Cancer Treat Rev 62:61–73. https://doi.org/10.1016/j.ctrv.2017.10.011
Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouche O, Mineur L, Barone C, Adenis A, Tabernero J, Yoshino T, Lenz HJ, Goldberg RM, Sargent DJ, Cihon F, Cupit L, Wagner A, Laurent D, Group CS (2013) Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381:303–312. https://doi.org/10.1016/S0140-6736(12)61900-X
Yoshino T, Komatsu Y, Yamada Y, Yamazaki K, Tsuji A, Ura T, Grothey A, Van Cutsem E, Wagner A, Cihon F, Hamada Y, Ohtsu A (2015) Randomized phase III trial of regorafenib in metastatic colorectal cancer: analysis of the CORRECT Japanese and non-Japanese subpopulations. Investig New Drugs 33:740–750. https://doi.org/10.1007/s10637-014-0154-x
Lipworth AD, Robert C, Zhu AX (2009) Hand-foot syndrome (hand-foot skin reaction, palmar-plantar erythrodysesthesia): focus on sorafenib and sunitinib. Oncology 77:257–271. https://doi.org/10.1159/000258880
Lacouture ME, Reilly LM, Gerami P, Guitart J (2008) Hand foot skin reaction in cancer patients treated with the multikinase inhibitors sorafenib and sunitinib. Ann Oncol 19:1955–1961. https://doi.org/10.1093/annonc/mdn389
Kubota Y, Fujita KI, Takahashi T, Sunakawa Y, Ishida H, Hamada K, Ichikawa W, Tsunoda T, Shimada K, Masuo Y, Kato Y, Sasaki Y (2020) Higher systemic exposure to unbound active metabolites of regorafenib is associated with short progression-free survival in colorectal cancer patients. Clin Pharmacol Ther 108:586–595. https://doi.org/10.1002/cpt.1810
Ding F, Liu B, Wang Y (2020) Risk of hand-foot skin reaction associated with vascular endothelial growth factor-tyrosine kinase inhibitors: a meta-analysis of 57 randomized controlled trials involving 24,956 patients. J Am Acad Dermatol 83:788–796. https://doi.org/10.1016/j.jaad.2019.04.021
Azad NS, Aragon-Ching JB, Dahut WL, Gutierrez M, Figg WD, Jain L, Steinberg SM, Turner ML, Kohn EC, Kong HH (2009) Hand-foot skin reaction increases with cumulative sorafenib dose and with combination anti-vascular endothelial growth factor therapy. Clin Cancer Res 15:1411–1416. https://doi.org/10.1158/1078-0432.CCR-08-1141
Hasan Alshammari A, Masuo Y, Fujita KI, Shimada K, Iida N, Wakayama T, Kato Y (2022) Discrimination of hand-foot skin reaction caused by tyrosine kinase inhibitors based on direct keratinocyte toxicity and vascular endothelial growth factor receptor-2 inhibition. Biochem Pharmacol 197:114914. https://doi.org/10.1016/j.bcp.2022.114914
Apte RS, Chen DS, Ferrara N (2019) VEGF in signaling and disease: beyond discovery and development. Cell 176:1248–1264. https://doi.org/10.1016/j.cell.2019.01.021
Gurtner GC, Werner S, Barrandon Y, Longaker MT (2008) Wound repair and regeneration. Nature 453:314–321. https://doi.org/10.1038/nature07039
Singer AJ, Clark RA (1999) Cutaneous wound healing. N Engl J Med 341:738–746. https://doi.org/10.1056/NEJM199909023411006
Suenaga M, Mashima T, Kawata N, Wakatsuki T, Horiike Y, Matsusaka S, Dan S, Shinozaki E, Seimiya H, Mizunuma N, Yamaguchi K, Yamaguchi T (2016) Serum VEGF-A and CCL5 levels as candidate biomarkers for efficacy and toxicity of regorafenib in patients with metastatic colorectal cancer. Oncotarget 7:34811–34823. https://doi.org/10.18632/oncotarget.9187
Wang SW, Liu SC, Sun HL, Huang TY, Chan CH, Yang CY, Yeh HI, Huang YL, Chou WY, Lin YM, Tang CH (2015) CCL5/CCR5 axis induces vascular endothelial growth factor-mediated tumor angiogenesis in human osteosarcoma microenvironment. Carcinogenesis 36:104–114. https://doi.org/10.1093/carcin/bgu218
dbSNP National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/snp/. Accessed 10 Aug 2023
Suenaga M, Schirripa M, Cao S, Zhang W, Yang D, Ning Y, Cremolini C, Antoniotti C, Borelli B, Mashima T, Okazaki S, Berger MD, Miyamoto Y, Gopez R Jr, Barzi A, Lonardi S, Yamaguchi T, Falcone A, Loupakis F, Lenz HJ (2018) Gene polymorphisms in the CCL5/CCR5 pathway as a genetic biomarker for outcome and hand-foot skin reaction in metastatic colorectal cancer patients treated with regorafenib. Clin Colorectal Cancer 17:e395–e414. https://doi.org/10.1016/j.clcc.2018.02.010
Howard OM, Turpin JA, Goldman R, Modi WS (2004) Functional redundancy of the human CCL4 and CCL4L1 chemokine genes. Biochem Biophys Res Commun 320:927–931. https://doi.org/10.1016/j.bbrc.2004.06.039
Colobran R, Adreani P, Ashhab Y, Llano A, Este JA, Dominguez O, Pujol-Borrell R, Juan M (2005) Multiple products derived from two CCL4 loci: high incidence of a new polymorphism in HIV+ patients. J Immunol 174:5655–5664. https://doi.org/10.4049/jimmunol.174.9.5655
Kobayashi K, Kawakami K, Yokokawa T, Aoyama T, Suzuki K, Wakatsuki T, Suenaga M, Sato H, Sugiyama E, Yamaguchi K, Hama T (2019) Association of hand-foot skin reaction with regorafenib efficacy in the treatment of metastatic colorectal cancer. Oncology 96:200–206. https://doi.org/10.1159/000495989
Argiles G, Mulet N, Valladares-Ayerbes M, Vieitez JM, Gravalos C, Garcia-Alfonso P, Santos C, Tobena M, Garcia-Paredes B, Benavides M, Cano MT, Loupakis F, Rodriguez-Garrote M, Rivera F, Goldberg RM, Cremolini C, Bennouna J, Ciardiello F, Tabernero JM, Aranda E, Spanish Cooperative Group for the Treatment of Digestive T, Unicancer GI, The Ri, Principal i, Argiles G, Tabernero J, Steering C, Investigators (2022) A randomised phase 2 study comparing different dose approaches of induction treatment of regorafenib in previously treated metastatic colorectal cancer patients (REARRANGE trial). Eur J Cancer 177:154–163. https://doi.org/10.1016/j.ejca.2022.09.037
Bekaii-Saab TS, Ou FS, Ahn DH, Boland PM, Ciombor KK, Heying EN, Dockter TJ, Jacobs NL, Pasche BC, Cleary JM, Meyers JP, Desnoyers RJ, McCune JS, Pedersen K, Barzi A, Chiorean EG, Sloan J, Lacouture ME, Lenz HJ, Grothey A (2019) Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol 20:1070–1082. https://doi.org/10.1016/S1470-2045(19)30272-4
Funding
This study was supported in part by a JSPS Grant-in-Aid for Scientific Research (C) Grant Number 19K07204 to K.F.
Author information
Authors and Affiliations
Contributions
K.O., R.M., and K.F. wrote the manuscript; K.O., R.M., and K.F. designed the study; K.O., R.M., N.M., Y.K., H.I., and K.F. performed the research; and K.O., R.M., N.M., Y.K., H.I., and K.F. analyzed the data.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was conducted in accordance with the principles of the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board of Showa University. The study was registered with the University Hospital Medical Information Network-Clinical Trials Registry for Japan on May 12, 2014 (UMIN000013939), when 6 months after the approval by the Institutional Review Board of Showa University.
Consent to participate
All patients gave written informed consent for the use of their peripheral blood samples and medical information for research purposes.
Consent to publish
All patients gave written informed consent regarding publishing their data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ono, K., Murase, R., Matsumoto, N. et al. Association of VEGFA and CCL4L2 polymorphisms with hand-foot skin reaction and survival of regorafenib in Japanese patients with colorectal cancer. Cancer Chemother Pharmacol (2024). https://doi.org/10.1007/s00280-024-04649-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00280-024-04649-5